Attenuation of Antidepressant Effects of Ketamine by Opioid Receptor Antagonism
Abstract
Objective:
In addition to N-methyl-d-aspartate receptor antagonism, ketamine produces opioid system activation. The objective of this study was to determine whether opioid receptor antagonism prior to administration of intravenous ketamine attenuates its acute antidepressant or dissociative effects.
Method:
In a proposed double-blind crossover study of 30 adults with treatment-resistant depression, the authors performed a planned interim analysis after studying 14 participants, 12 of whom completed both conditions in randomized order: placebo or 50 mg of naltrexone preceding intravenous infusion of 0.5 mg/kg of ketamine. Response was defined as a reduction ≥50% in score on the 17-item Hamilton Depression Rating Scale (HAM-D) score on postinfusion day 1.
Results:
In the interim analysis, seven of 12 adults with treatment-resistant depression met the response criterion during the ketamine plus placebo condition. Reductions in 6-item and 17-item HAM-D scores among participants in the ketamine plus naltrexone condition were significantly lower than those of participants in the ketamine plus placebo condition on postinfusion days 1 and 3. Secondary analysis of all participants who completed the placebo and naltrexone conditions, regardless of the robustness of response to ketamine, showed similar results. There were no differences in ketamine-induced dissociation between conditions. Because naltrexone dramatically blocked the antidepressant but not the dissociative effects of ketamine, the trial was halted at the interim analysis.
Conclusions:
The findings suggest that ketamine’s acute antidepressant effect requires opioid system activation. The dissociative effects of ketamine are not mediated by the opioid system, and they do not appear sufficient without the opioid effect to produce the acute antidepressant effects of ketamine in adults with treatment-resistant depression.
Depression is the leading cause of disability worldwide (1), yet novel antidepressant development has stalled (2). While traditional antidepressant medications remain the staple for treating major depressive disorder, a significant proportion of patients fail to achieve clinical response with standard treatments (3) and require interventional approaches such as ECT, repetitive transcranial magnetic stimulation, and intravenous ketamine infusion (4). With 40%−60% of patients meeting clinical criteria for an antidepressant response after infusion, ketamine has demonstrated impressive efficacy in patients who have failed to respond to traditional antidepressant therapies (5).
Although the specific mechanisms of action responsible for the acute antidepressant effects of ketamine have yet to be determined (5), they have generally been conceptualized to be due to N-methyl-d-aspartate (NMDA) receptor antagonism (5, 6). However, other candidate NMDA receptor antagonists have not been proven to be effective antidepressants (5). More recently, a preclinical study reported that antidepressant effects of ketamine are independent of NMDA receptor antagonism and are due to modulation at other receptors, such as the AMPA receptor (7).
Beyond the glutamate system, ketamine interacts with several additional neurotransmitter systems, including mu, delta, and kappa opioid receptors, and it is currently used as an antinociceptive agent for acute and chronic pain (8). Ketamine’s analgesic effects are blocked by mu and delta opioid receptor antagonists but not by kappa opioid receptor antagonists, indicating a mu or delta opioid mechanism in ketamine’s antinociceptive effects (9). We and others have hypothesized that ketamine’s antidepressant mechanism of action may in fact be related to intrinsic opioid receptor properties of ketamine (10) and have proposed that coadministration of an opioid receptor antagonist with ketamine could be employed to test this hypothesis (11). Yet, no study to date has probed the role that ketamine’s opioid properties play in its antidepressant effects (12).
As a dissociative anesthetic (13, 14), ketamine is capable of producing dramatic psychotomimetic effects (15–17), and these effects have been correlated to its antidepressant efficacy (18). Here too, the specific receptor system or systems responsible for mediating dissociative effects of ketamine are unknown. Some but not all NMDA receptor antagonists cause dissociation (19). The pure kappa opioid receptor agonist salvinorin A does produce dissociative effects similar to those of ketamine (20, 21). A low dose (25 mg) of the opioid receptor antagonist naltrexone can augment the psychoactive effects of lower subanesthetic doses (∼0.4 mg/kg per hour) of ketamine, but not higher subanesthetic doses (∼0.6 mg/kg per hour) in healthy subjects (22). However, opioid receptor antagonists have not been previously used to probe the role opioid receptors play in ketamine’s dissociative effects in adults with treatment-resistant depression, and the 25-mg dose of naltrexone does not completely block opioid receptors (23).
The objective of this study was not to assess ketamine’s antidepressant efficacy but rather to determine the role of the opioid system in ketamine’s antidepressant and dissociative effects in adults with treatment-resistant depression. We conducted a randomized double-blind crossover trial in which intravenous ketamine was infused once each across two conditions, with participants receiving pretreatment with naltrexone before one of their ketamine infusions (the ketamine plus naltrexone condition) and placebo before the other ketamine infusion (the ketamine plus placebo condition) in a counterbalanced manner. Through this mechanistic clinical trial design, we tested whether pretreatment with an opioid receptor antagonist is able to attenuate the acute antidepressant or dissociative effects of ketamine.
Method
Participants
Potential study participants were brought into the clinic for a screening visit to determine eligibility. All study participants were outpatients. Inclusion criteria included a current diagnosis of a nonpsychotic, nonatypical major depressive episode as part of either major depressive disorder or bipolar II disorder, defined by DSM-5 criteria (24). For the initial enrollment, all participants were required to have a score ≥20 on the 17-item Hamilton Depression Rating Scale (HAM-D) (25). Each participant was also required not to have benefited sufficiently from trials of at least four different antidepressant medications or other somatic treatments as defined by the Massachusetts General Hospital Antidepressant Treatment History Questionnaire criteria (26).
All eligible participants provided fully informed written consent. The study protocol was approved by the Stanford University Institutional Review Board. Participants were required to avoid taking certain medications for a period before ketamine administration: any stimulant medications documented during the screening phase were to be withheld during the 24 hours prior to ketamine administration, and any benzodiazepine for the 8 hours prior to (or any hypnotic drugs the night prior to) ketamine administration; these medications could be resumed on postinfusion day 1 after completion of the ratings. Furthermore, any medical marijuana use was to be withheld for 2 weeks in order to allow for proper washout prior to the baseline/randomization visit (i.e., at least five half-lives of the drug). We excluded individuals on opioids in order to avoid naltrexone precipitating opioid withdrawal, along with eliminating the confounding of ketamine-opioid interactions. If a washout period was necessary prior to study participation, the study physician maintained ongoing contact with the participant to ensure safety during this period. Antidepressant medications deemed not likely to interact with ketamine (selective serotonin reuptake inhibitors, selective norepinephrine reuptake inhibitors, monoamine oxidase inhibitors, tricyclic antidepressants, and bupropion) and some adjunctive medications (antipsychotics, antiepileptics, and thyroid hormone) were maintained at a constant dosage for at least 4 weeks. After eligibility was confirmed, participants’ demographic and medical data were collected.
Sixteen patients consented to participate in the study. Two of the 16 were withdrawn—one who was found to be positive for methamphetamine and one who was found to have an unreported medical illness. The patient flow through the study is summarized in Figure S1 in the online supplement. Fourteen participants received at least one ketamine infusion; two of the 14 discontinued, one because of adverse events and the other because of a need for an increase in care. Thus, 12 participants completed both infusions. Table 1 summarizes the sample characteristics. The mean age was 41.3 years (SD=11.8). Of the 12 participants who completed both conditions, all were diagnosed with recurrent major depressive disorder. Six were women, five were unemployed, and two were receiving disability benefits.
| Characteristic or Measure | Overall Sample (N=14) | Responders (N=7) | Nonresponders (N=5) | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| Age (years) | 41.3 | 11.8 | 39.8 | 8.2 | 44.4 | 18.2 |
| Age at illness onset (years) | 17.3 | 4.3 | 16.3 | 3.2 | 17.8 | 5.8 |
| Duration of illness (years) | 24.1 | 10.6 | 23.5 | 9.2 | 26.6 | 14.6 |
| Duration of current episode (years) | 8.6 | 7.4 | 7.7 | 8.3 | 10.2 | 6.8 |
| Antidepressants and adjunctive agents used in current episode | 5.7 | 5.8 | 4.0 | 3.3 | 5.0 | 1.6 |
| Antidepressant failures in current episode | 3.8 | 3.0 | 2.9 | 2.4 | 3.6 | 1.1 |
| Total number of antidepressants, lifetime | 6.9 | 3.5 | 7.0 | 35.0 | 5.5 | 3.4 |
| N | % | N | % | N | % | |
| Female | 6 | 42.9 | 4 | 57.1 | 1 | 20.0 |
| Diagnosis of recurrent major depression | 12 | 85.7 | 7 | 100.0 | 4 | 80.0 |
| Past ECT or transcranial magnetic stimulation | 6 | 42.9 | 2 | 28.6 | 2 | 40.0 |
| Past psychotherapy | 11 | 78.6 | 6 | 85.7 | 3 | 60.0 |
| Family history of depression | 5 | 35.7 | 3 | 42.9 | 2 | 40.0 |
| Mean | SD | Mean | SD | Mean | SD | |
| Hamilton Depression Rating Scale (17 item) | 25.9 | 4.6 | 26.0 | 4.3 | 26.6 | 5.8 |
| Montgomery-Åsberg Depression Rating Scale | 35.3 | 4.9 | 35.1 | 3.8 | 35.6 | 4.8 |
| Clinical Global Impressions Scale, severity score | 5.1 | 0.5 | 5.1 | 0.4 | 5.0 | 0.7 |
| Beck Depression Inventory–II | 30.1 | 10.5 | 29.1 | 9.5 | 28.8 | 9.8 |
TABLE 1. Baseline Demographic and Clinical Characteristics of Patients in a Study of Ketamine’s Antidepressant Effect After Pretreatment With Naltrexone or Placeboa
The average length of depressive illness was 24.1 years (SD=10.6), and the average length of the current depressive episode was 8.6 years (SD=7.4). Participants reported a lifetime mean of 9.8 (SD=6.5, mode=8) unsuccessful antidepressant treatments (primary, adjunctive, somatic, and psychotherapy) and a lifetime mean of 6.9 (SD=3.5, mode=3) primary antidepressant medication trials. Participants reported using a mean total of 5.7 (SD=5.8, mode=3) antidepressant agents (primary and adjunctive) during the current episode and a mean of 3.8 (SD=3.0, mode=3) primary antidepressant treatments during the current episode. Several participants (N=6) had a history of failing to respond to repetitive transcranial magnetic stimulation, and one had failed to respond to ECT.
Study Design
The study employed a crossover design comprising two treatment conditions: oral placebo or oral naltrexone (50 mg) preceding a 0.5 mg/kg intravenous infusion of ketamine. Placebo and naltrexone pills were identical in appearance (the naltrexone pill was overencapsulated). The order of the treatments was randomized, and investigators and participants were blind to the order. Placebo or naltrexone was administered 45 minutes before the initiation of the ketamine infusion in order to achieve peak naltrexone levels at the initiation of the ketamine infusion (27). Ketamine was then administered intravenously over 40 minutes. Participants were monitored with continuous three-lead ECG, pulse oximetry, end-tidal capnography, and noninvasive blood pressure measurement every 5 minutes during the infusion. Participants were monitored by a study physician and study staff throughout the course of the infusion.
Ratings of depression were assessed on the 6-item and 17-item HAM-D at baseline and at postinfusion days 1, 3, 5, 7, and 14. The primary outcome measure was reduction of depressive symptoms at postinfusion day 1 among participants who met the response criterion during the ketamine plus placebo condition. Ketamine response was defined as a reduction ≥50% in score on the 17-item HAM-D at postinfusion day 1, as has been done in a number of previous ketamine studies (27). The secondary outcome measure was the rating on the Clinician-Administered Dissociative States Scale (CADSS) (28). We collected data at multiple time points to assess for prolonged effects of ketamine and/or naltrexone, including whether naltrexone blocked or delayed the antidepressant effects of ketamine, since naltrexone produces a 96-hour blockade of opioid receptors in the brain (23). The CADSS was administered prior to infusion and again at multiple points up to 180 minutes after infusion. Raters were blind to treatment condition for all assessments.
After completing their first treatment condition, participants were assessed 28 days later to evaluate for relapse (defined as having a score within 20% of their baseline score on the 17-item HAM-D) and to determine eligibility for entering the second treatment condition. We selected an interval of 28 days between infusions to minimize carryover effects. If the participant had no response at any of the assessment points in the first 14 days, they could enter the second treatment condition after day 14. If there was a sustained antidepressant response from the first treatment, the participant was seen every 2 weeks until relapse occurred, up to 120 days. Once relapse was determined, participants crossed over to the second treatment condition.
Data Analysis
In this two-condition crossover study, we estimated a priori that a sample of 30 participants would be needed to yield 15 ketamine responders, as defined by a reduction ≥50% in baseline 17-item HAM-D score on postinfusion day 1 in the ketamine plus placebo condition (25). A power calculation indicated that analysis of 15 participants in a crossover model would be fully powered to detect statistical significance, assuming moderate to large effect sizes and an alpha of 0.05 (two-tailed). An interim analysis was planned for the midway point.
The primary endpoint evaluated the antidepressant response to ketamine plus naltrexone relative to the response to ketamine plus placebo in participants identified as ketamine plus placebo responders. A fixed-effects repeated-measures model was used to compare mean changes on the 17-item HAM-D and the 6-item subscale scores for two time points (preinfusion day 0 and postinfusion day 1) for the two conditions. There were no missing data on the primary endpoints (i.e., the 17-item and 6-item HAM-D). Statistical comparisons at time points after day 1 were conditional on the primary endpoint being statistically significant (29). Paired comparisons were conducted for the HAM-D scores measured at postinfusion days 3, 5, 7, and 14. There were five missing HAM-D scores across the 14-day study (three at day 5, one at day 7, and one at day 14). The secondary endpoint compared participants’ peak levels of dissociation in the two conditions, as measured by change in CADSS score at the end of the 40-minute infusion.
To more fully describe the relative effects of ketamine plus either placebo or naltrexone, we applied analytical methods similar to those reported by Zarate (30). After testing the primary mechanistic hypothesis among ketamine responders, two sets of analyses were used to understand more fully the effect of ketamine plus placebo and of ketamine plus naltrexone on the 17-item and 6-item HAM-D. The first set included analyses of all 12 participants who completed the crossover and received both treatment conditions (i.e., both responders and nonresponders). For these participants, a general linear model for repeated measurements tested within-subject effects of the two treatment conditions on change in HAM-D scores from day 0 to day 1.
Effect sizes were also calculated using standardized mean differences between conditions for the primary endpoints (i.e., 17-item and 6-item HAM-D before and after infusion). Potential carryover effects were tested using a fixed-effects model with treatment order as a between-subject factor and the HAM-D baseline score for each phase as the dependent variable. An alpha of 0.05 (two-tailed) was used to determine statistical significance.
Results
Fourteen participants received at least one infusion, and 12 participants completed the crossover and underwent both the ketamine plus placebo and the ketamine plus naltrexone conditions. The interval between ending the first condition and starting the second ranged from 14 to 63 days (mean=33, SD=14.8). After unblinding, analyses indicated that seven of the 12 participants who completed both treatment conditions met the prespecified response criterion, defined as a reduction ≥50% from baseline to day 1 in 17-item HAM-D score in the ketamine plus placebo condition (see the patient flow chart in Figure S1 in the online supplement).
Depression
There were no significant differences in mean baseline 17-item HAM-D score for the ketamine plus placebo condition (mean=26.7, SD=5.4) and the ketamine plus naltrexone condition (mean=28.1, SD=5.4). A robust reduction in mean 17-item HAM-D score was observed at postinfusion day 1 in the ketamine plus placebo condition (mean=−22.3, SD=3.2; F=106, p<0.001). A significant reduction from baseline was also observed in the ketamine plus naltrexone condition (mean=−5.6, SD=5.7; F=6.8, p=0.04), although the ketamine-induced reductions in depressive symptoms were significantly attenuated (mean difference=−16.7, SD=6.7; F=43.6, p<0.001; effect size, d=2.5). Significant differences between the ketamine plus placebo and ketamine plus naltrexone conditions were still evident at day 3, but not at days 5, 7, and 14 (see Figure 1A).
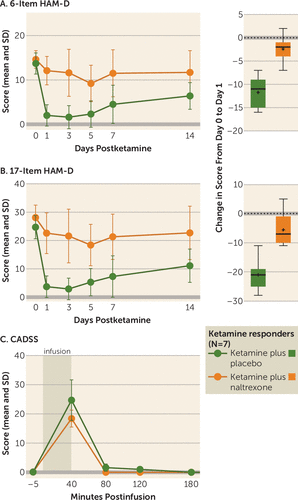
FIGURE 1. Time Course of Primary and Secondary Outcome Measures for Ketamine-Responsive Patients With Treatment-Resistant Depression (N=7) in Two Conditions in a Crossover Study of Ketamine’s Antidepressant Effect After Pretreatment With Naltrexone or Placeboa
a Treatments were administered on day 0, after the first administration of the questionnaires. In panel A, the graph on the left plots the time course of mean scores on the 6-item Hamilton Depression Rating Scale (HAM-D); analysis of between-group differences on postinfusion day 1 showed that scores for the ketamine plus naltrexone condition were significantly higher than those for the ketamine plus placebo condition, with participants in the latter condition demonstrating expected postinfusion HAM-D score reduction. The box and whisker plot on the right illustrates the distribution of the change in score from day 0 to day 1 for each treatment condition; the horizontal line indicates the median, the plus sign indicates the mean, the box indicates the 25th–75th percentile range, and the whiskers indicate the minimum and maximum scores. In panel B, the graphs show similar results for the 17-item HAM-D. Panel C plots the time course of the secondary outcome measure, score on the Clinician-Administered Dissociative States Scale (CADSS), before and after infusion of ketamine for both conditions. Peak CADSS scores immediately after infusion (at 40 minutes) did not differ significantly between conditions.
On postinfusion day 1 for the ketamine plus placebo condition, five of seven responders met criteria for remission (a score ≤7 on the 17-item HAM-D) (31). In contrast, on postinfusion day 1 for the ketamine plus naltrexone condition, none of the seven ketamine plus placebo responders met the response criterion (a reduction ≥50% in 17-item HAM-D score).
Similar results were observed with the 6-item HAM-D, which assesses the core symptoms of depression. The baseline mean score was 14.17 (SD=2.17) for the ketamine plus placebo condition and 14.29 (SD=2.33) for the ketamine plus naltrexone condition. In the ketamine plus placebo condition, a statistically significant reduction from baseline in mean 6-item HAM-D score was observed at postinfusion day 1 (mean=−11.7, SD=3.1; F=93.8, p<0.001). In the ketamine plus naltrexone condition, a change from baseline was observed at day 1, although the reduction was not statistically significant (mean=−2.4, SD=2.8; F=5.4, p=0.059). Comparison of reductions between conditions indicated that the reduction in 6-item HAM-D score observed in the ketamine plus naltrexone condition was significantly lower than that observed in the ketamine plus placebo condition (mean difference=9.3, SD=4; F=29.8, p=0.002; d=2.3; see Figure 1B).
After testing the mechanistic hypothesis by assessing attenuation of response for patients who responded to ketamine plus placebo (N=7), similar analyses were conducted for all participants who underwent both treatment conditions (N=12), regardless of whether they met the response criterion during the ketamine plus placebo condition. These data, including mean scores on the 6-item and 17-item HAM-D, are summarized in Figure 2A and 2B, respectively. On postinfusion day 1, statistically significant reductions in mean 17-item HAM-D scores were observed for both the ketamine plus placebo condition (mean=−14.2, SD=10.7; F=19.3, p<0.001) and the ketamine plus naltrexone condition (mean=−4.9, SD=6.8; F=8.7, p=0.013), with a significantly smaller reduction for the ketamine plus naltrexone condition (mean difference=−8.4, SD=12.6; condition-by-time interaction, F=5.4, p=0.041; d=0.7). A statistically significant reduction was also observed in 6-item HAM-D score in the ketamine plus placebo condition (mean=−7.5, SD=5.8, F=20.3, p<0.001). In the ketamine plus naltrexone condition, the mean reduction in 6-item HAM-D score was not statistically significant (mean=−2.0, SD=3.9; F=3.0 p=0.11), and the reduction was significantly attenuated compared with the ketamine plus placebo condition (mean difference=5.5, SD=6.9; F=7.7, p=0.018; d=0.8).
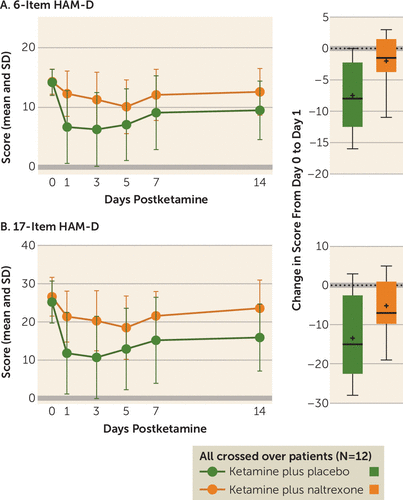
FIGURE 2. Time Course of Primary Outcome Measures for Ketamine-Responsive and Ketamine-Nonresponsive Patients With Treatment-Resistant Depression (N=12) in Two Conditions in a Crossover Study of Ketamine’s Antidepressant Effect After Pretreatment With Naltrexone or Placeboa
a In contrast to Figure 1, this figure presents data for all patients who received both infusions in the crossover design (N=12), including five patients who did not meet the ketamine response criterion required for inclusion in our primary outcome analysis. Treatments were administered on day 0, after the first administration of the questionnaires. In panel A, the graph on the left plots the time course of mean scores on the 6-item Hamilton Depression Rating Scale (HAM-D). The difference between scores for participants in the ketamine plus naltrexone and ketamine plus placebo conditions on postinfusion day 1 is maintained when ketamine nonresponders are included in the crossover analysis. The box and whisker plot on the right illustrates the distribution of the change in score from day 0 to day 1 for each treatment condition; the horizontal line indicates the median, the plus sign indicates the mean, the box indicates the 25th–75th percentile range, and the whiskers indicate the minimum and maximum scores. In panel B, the graphs show similar results for the 17-item HAM-D.
Dissociation
Among the ketamine responders (N=7), the mean CADSS score significantly increased from before infusion to 40 minutes after infusion in both conditions (ketamine plus placebo: median=23, mean=24.7, SD=18.3; ketamine plus naltrexone: median=21, mean=18.2, SD=7.6), as shown in Figure 1C. However, there was no significant difference between the ketamine plus placebo and ketamine plus naltrexone conditions in average levels of dissociation (Wilcoxon test, p=0.45). Among study completers (N=12), which included responders and nonresponders to ketamine plus placebo, CADSS scores increased in both conditions, albeit to a lesser extent in the ketamine plus naltrexone condition (ketamine plus placebo: median=17.5, mean=19.1, SD=16.3; ketamine plus naltrexone: median=14.5, mean=13.8, SD=8.8). After 40 minutes, CADSS scores normalized, with only three patients having scores ≥1 at 80 minutes.
Evaluation of Blind and Side Effects
Data were collected on a range of visual analog scales that address a variety of potential psychoactive side effects 45 minutes after ingestion of naltrexone or placebo, immediately before initiation of the ketamine infusion. There were no differences on these scales between participants receiving naltrexone or placebo (see Figure S4 in the online supplement), as has been demonstrated previously for naltrexone (23, 32–37). No other direct assessment of blind integrity was performed. Among the 12 participants who completed the crossover, seven participants in the naltrexone condition experienced nausea after the ketamine infusion, in contrast to three who experienced nausea in the ketamine plus placebo condition; two of these participants in each condition also experienced vomiting.
Termination of Study
At the interim analysis, given the finding that the combination of ketamine and naltrexone was not only ineffective but also noxious for many participants, we decided to stop enrolling patients in the study.
Discussion
Ketamine has well-established rapid-onset antidepressant effects. The majority of preclinical studies investigating the mechanism of this effect have focused on NMDA receptor antagonism, and several clinical trials have attempted to replicate this rapid antidepressant effect with other NMDA receptor antagonists, with limited success (5). We now present the first evidence in humans that opioid receptors are necessary for ketamine’s acute antidepressant effect. In ketamine-responsive patients with treatment-resistant depression, pretreatment with naltrexone profoundly attenuated ketamine’s antidepressant effect, with none of the ketamine responders meeting the response criterion at day 1. We observed concordant effects on related measures of depression, including clinician-administered scales (the 6-item HAM-D and the Montgomery-Åsberg Depression Rating Scale [MADRS]) and a self-report instrument (the Beck Depression Inventory–II) (see the online supplement), which strengthens our conclusion that ketamine’s antidepressant effects require opioid system activation. Of note, we observed a statistically significant difference from baseline at postinfusion day 1 in 17-item HAM-D score for the ketamine plus naltrexone condition, but not on the MADRS or the 6-item HAM-D, both of which are scales thought to reflect core depressive symptoms.
The endogenous opioid system has been reported to play a central role in the pathophysiology and treatment of affective disorders (38–43). A robust nonhuman primate literature supports the idea that opioids are important in mediating emotions associated with depression (44, 45). Depressive disorders have been associated with dysregulation of the endogenous opioid system, particularly mu and kappa opioid receptors’ tone (39, 40). Moreover, buprenorphine, a mu opioid receptor partial agonist and a kappa opioid receptor antagonist, has been shown to produce antidepressant effects (41, 42), even in individuals who have failed to benefit from ECT (43). In obsessive-compulsive disorder, single infusions of ketamine have been reported to produce a multiday benefit (46), as has a single oral dose of morphine, which is a mu opioid receptor agonist (47). These data suggest that mu opioid receptor agonists with additional NMDA receptor antagonist properties may have therapeutic potential as intermittently dosed therapies for mood or anxiety disorders.
The kappa opioid receptor is also emerging as a regulator of mood and motivation (48–50), with increased kappa opioid receptor activity being associated with depression (51). Because naltrexone does not have substantial selectivity for the mu opioid receptor over the kappa opioid receptor (52, 53), the 50-mg dose of naltrexone used in this study saturated the mu opioid receptors and likely equally saturated the kappa receptors (23, 54). Thus, our data cannot distinguish between the respective roles of mu and kappa opioid receptors in mediating ketamine’s antidepressant effects. Nonetheless, given the available data implicating mu opioid receptor–based mechanisms of antidepressant efficacy, inconsistent findings regarding kappa opioid receptor antagonists in depression (55, 56), and ketamine’s putative kappa agonist mechanism (21, 57), we favor the interpretation that ketamine produces its acute antidepressant response primarily through direct and/or indirect actions at the mu opioid receptors. Naltrexone, when chronically administered alone in healthy subjects as well as in individuals with mood and substance use disorders, has been demonstrated across several placebo-controlled trials either to act as an antidepressant or to be mood neutral (23, 32–37), which suggests that naltrexone is not simply acting as a depressogenic agent in this case but rather providing selective blockade of the antidepressant effects produced by ketamine.
How do we reconcile these data with the large body of evidence implicating glutamate receptors in ketamine’s primary antidepressant mechanism? The majority of studies to date have focused on ketamine’s antidepressant mechanism of action as a noncompetitive antagonist of the NMDA receptor and subsequent activation of AMPA receptors. Recently, a preclinical study reported that a metabolite of ketamine, 2R,6R-hydroxynorketamine, has antidepressant efficacy through stimulation of the AMPA receptor independently of NMDA receptor antagonism (7). This mechanism of action has been replicated by some (58, 59) but not all (60) groups. In addition, preclinical studies demonstrate that glutamate receptor modulation triggers downstream modulation of synthesis and release of brain-derived neurotrophic factor and enhances synaptic plasticity via activation of molecular targets such as mammalian target of rapamycin and eukaryotic elongation factor 2 (58, 59, 61–63). These glutamate system effects may in fact drive the transient maintenance of the antidepressant response through modulation of brain plasticity (64) rather than producing the actual acute antidepressant effects.
No studies to date have directly addressed the role of opioid receptors in ketamine’s antidepressant effect. However, our demonstration of an opioid system activation requirement for ketamine’s acute antidepressant effect mirrors a long-standing literature investigating the opioid mechanism of action of ketamine’s analgesic properties. On the MADRS and the 6-item HAM-D (which reflects the core depressive symptoms [65]), our data demonstrated that the effect of ketamine was actually ablated by naltrexone. Meta-analyses have consistently shown that ketamine has a clinically significant opioid-sparing effect, in which coadministration of ketamine allows for lower doses of traditional opioids to be used to achieve similar antinociceptive effects (66). In addition to ketamine’s combined naloxone-sensitive and naloxone-insensitive analgesic effects (67), human and preclinical studies have found that ketamine 1) substantially potentiates the analgesic effect of opioids (68), 2) produces opioid receptor–dependent analgesia (9, 69, 70), 3) reduces opioid tolerance and opioid-induced hyperalgesia to opioids (71), and 4) produces mu opioid receptor–dependent respiratory depression (70).
With the proviso that the scope of ketamine’s pharmacology is continually expanding (72), the available evidence suggests that ketamine-mediated analgesia involves either a direct action at mu opioid receptors (57, 73–75) or an interaction between NMDA receptor antagonists and mu opioid receptors (76–78). The hypothesis that NMDA receptor antagonists and mu opioid receptors share subcellular co-localization and may exist as a functional complex in a crucial nociceptive brain area (the periaqueductal gray) (77) forms a particularly compelling explanation for apparently conflicting findings in the context of ketamine-mediated analgesia. Notably, naltrexone pretreatment did not significantly affect ketamine-induced dissociation, as measured by the CADSS, nor did CADSS score correlate with ketamine’s antidepressant efficacy. Previous work has attributed ketamine’s dissociative and hypnotic properties to NMDA receptor antagonism and hyperpolarization-activated cyclic nucleotide-gated cation channel 1 blockade (72) as well as activation of kappa opioid receptors (21). Our finding that the dissociative effects of ketamine persist despite naltrexone antagonism of opioid receptors suggests that opioid receptors do not play a major role in mediating ketamine’s dissociative effects.
The public health significance of ketamine’s opioid properties needs to be studied. Depression and opioid dependence are currently the two most significant public health problems facing the United States and have become leading causes of disability and death worldwide (1, 79, 80). While opioids have a history of use as antidepressants (43, 81), they pose a significant risk if used chronically (82). Half of patients who receive prescriptions for opioids have a mental health diagnosis (50, 83–85), and over half of individuals with opioid use disorders have a primary diagnosis of depression (86). There is also a significant ketamine abuse problem worldwide (87–89), and ketamine ranks high on the list of commonly abused substances (90–94). Moreover, ketamine abusers have high rates of depression (77) and experience significant brain dysfunction (95). While these risks have not been demonstrated in serial infusions for depressed patients (96), short-interval repetitive dosing strategies may pose greater risks (97), and there have been case reports of apparent tolerance after chronic administration (98, 99). Ketamine tolerance has been observed in pain/anesthesia indications (100–107) as well as in animal models (108–110). The route of administration (111) and the patient’s access to the medication may play a role in the risk (112). Thus, the abuse and dependence potential of frequent ketamine treatment in major depression needs further study, and our results provide strong justification for further caution against widespread and repeated use of ketamine before further mechanistic testing has been performed (99, 113, 114).
Our study has a number of strengths and weaknesses. A crossover design was the optimal method for testing the study’s mechanistic hypothesis, since it can clearly identify ketamine responders post hoc and establish, in the individual study participant, that ketamine’s antidepressant effects are mediated via the opioid system. We did not employ an alternative design in which responders would first be identified by open-label treatment with ketamine, which could produce an expectancy bias, with participants expecting that they would have a similar response in the randomized treatment. Moreover, the crossover design provides significantly greater statistical power to detect group differences with fewer subjects. Limitations of a crossover study include potential carryover effects (115). However, because ketamine’s effects are transient, our washout period was sufficient for participants’ 17-item HAM-D scores to return to within 20% of their baseline scores, and thus any medication-related carryover effects were limited. While we cannot completely rule out the presence of carryover effects in our primary analysis (115), in an alternative analysis involving only the first randomized infusion (prior to crossover), we did observe a significant difference in response between the ketamine plus placebo and ketamine plus naltrexone conditions (see the online supplement), further demonstrating that naltrexone blocks the antidepressant effects of ketamine.
We assessed the integrity of the blind post hoc with visual analog scale assessments made 45 minutes after taking naltrexone or placebo, just prior to ketamine infusion. We could identify no item or group of items (see Figure S4 in the online supplement), or side effect, that participants could have reasonably used to infer their blinded condition. In any longitudinal study, regression to the mean is a possible issue. Data from Murrough et al. (12) indicate that initial response to ketamine is replicated by reinfusing ketamine three times per week for 2 weeks with repeated treatments. One weakness in our study was the final sample size in the interim analysis, and our findings do need to be replicated in other studies. Still, we found the same qualitative block of ketamine’s effect regardless of the depression instrument used, and with several alternative statistical analyses. We decided to stop the study because our results were both statistically and clinically significant and we were concerned about the ethics of exposing more people to a clearly ineffective and noxious combination treatment.
Further studies are needed to expand our understanding of the opioid effects of ketamine, including studies seeking to determine which opioid receptors are involved in mediating ketamine’s antidepressant effects, using more selective opioid receptor antagonists (116), surrogate markers (117), and functional neuroimaging capable of discerning those selective effects (54). The findings presented here challenge our understanding of the mechanisms of action of ketamine that underlie its potent antidepressant properties (118, 119).
1 : Depression is the leading cause of disability around the world. JAMA 2017; 317:1517Google Scholar
2 : Revolution stalled. Sci Transl Med 2012; 4:155cm11Crossref, Medline, Google Scholar
3 : Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry 2006; 163:1905–1917Link, Google Scholar
4 : Interventional psychiatry: why now? J Clin Psychiatry 2014; 75:895–897Crossref, Medline, Google Scholar
5 : NMDA antagonist treatment of depression. Curr Opin Neurobiol 2016; 36:112–117Crossref, Medline, Google Scholar
6 : NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011; 475:91–95Crossref, Medline, Google Scholar
7 : NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature 2016; 533:481–486Crossref, Medline, Google Scholar
8 : Ketamine for chronic pain: risks and benefits. Br J Clin Pharmacol 2014; 77:357–367Crossref, Medline, Google Scholar
9 : Central antinociception induced by ketamine is mediated by endogenous opioids and μ- and δ-opioid receptors. Brain Res 2014; 1562:69–75Crossref, Medline, Google Scholar
10 : A word to the wise about ketamine. Am J Psychiatry 2014; 171:262–264Link, Google Scholar
11 : Ketamine: promising path or false prophecy in the development of novel therapeutics for mood disorders? Neuropsychopharmacology 2015; 40:1307Crossref, Medline, Google Scholar
12 : Rapid and longer-term antidepressant effects of repeated ketamine infusions in treatment-resistant major depression. Biol Psychiatry 2013; 74:250–256Crossref, Medline, Google Scholar
13 : Ketamine: its pharmacology and therapeutic uses. Anesthesiology 1982; 56:119–136Crossref, Medline, Google Scholar
14 : Pharmacologic effects of Ci-581, a new dissociative anesthetic, in man. Clin Pharmacol Ther 1965; 6:279–291Crossref, Medline, Google Scholar
15 : Differential psychopathology and patterns of cerebral glucose utilisation produced by (S)- and (R)-ketamine in healthy volunteers using positron emission tomography (PET). Eur Neuropsychopharmacol 1997; 7:25–38Crossref, Medline, Google Scholar
16 : Effects of (S)-ketamine on striatal dopamine: a [11C]raclopride PET study of a model psychosis in humans. J Psychiatr Res 2000; 34:35–43Crossref, Medline, Google Scholar
17 : Therapeutic infusions of ketamine: do the psychoactive effects matter? Drug Alcohol Depend 2014; 136:153–157Crossref, Medline, Google Scholar
18 : Do the dissociative side effects of ketamine mediate its antidepressant effects? J Affect Disord 2014; 159:56–61Crossref, Medline, Google Scholar
19 : NMDA agonists and antagonists as probes of glutamatergic dysfunction and pharmacotherapies in neuropsychiatric disorders. Harv Rev Psychiatry 1999; 7:125–143Crossref, Medline, Google Scholar
20 : Human psychopharmacology and dose-effects of salvinorin A, a kappa opioid agonist hallucinogen present in the plant Salvia divinorum. Drug Alcohol Depend 2011; 115:150–155Crossref, Medline, Google Scholar
21 : Role of kappa-opioid receptors in the effects of salvinorin A and ketamine on attention in rats. Psychopharmacology (Berl) 2010; 210:263–274Crossref, Medline, Google Scholar
22 : Potentiation of low dose ketamine effects by naltrexone: potential implications for the pharmacotherapy of alcoholism. Neuropsychopharmacology 2006; 31:1793–1800Crossref, Medline, Google Scholar
23 : Duration of occupancy of opiate receptors by naltrexone. J Nucl Med 1988; 29:1207–1211Medline, Google Scholar
24 : Diagnostic and Statistical Manual of Mental Disorders, 5th edition: DSM-5. Washington, DC, American Psychiatric Association, 2013Crossref, Google Scholar
25 : A rating scale for depression. J Neurol Neurosurg Psychiatry 1960; 23:56–62Crossref, Medline, Google Scholar
26 : RESEARCH: Validation of the Massachusetts General Hospital Antidepressant Treatment History Questionnaire (ATRQ). CNS Neurosci Ther 2010; 16:322–325Crossref, Medline, Google Scholar
27 : Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry 2015; 172:950–966Link, Google Scholar
28 : Measurement of dissociative states with the Clinician-Administered Dissociative States Scale (CADSS). J Trauma Stress 1998; 11:125–136Crossref, Medline, Google Scholar
29 : Clinical Trials: A Methodologic Perspective. Hoboken, NJ, John Wiley & Sons, 2005Crossref, Google Scholar
30 : A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry 2006; 63:856–864Crossref, Medline, Google Scholar
31 : Conceptualization and rationale for consensus definitions of terms in major depressive disorder: remission, recovery, relapse, and recurrence. Arch Gen Psychiatry 1991; 48:851–855Crossref, Medline, Google Scholar
32 : Naltrexone modulates dopamine release following chronic, but not acute amphetamine administration: a translational study. Transl Psychiatry 2017; 7:e1104Crossref, Medline, Google Scholar
33 : A double-blind, placebo-controlled trial combining sertraline and naltrexone for treating co-occurring depression and alcohol dependence. Am J Psychiatry 2010; 167:668–675Link, Google Scholar
34 : A randomized trial of combined citalopram and naltrexone for nonabstinent outpatients with co-occurring alcohol dependence and major depression. J Clin Psychopharmacol 2015; 35:143–149Crossref, Medline, Google Scholar
35 : Naltrexone and dysphoria: a double-blind placebo controlled trial. Biol Psychiatry 1987; 22:710–716Crossref, Medline, Google Scholar
36 : Naltrexone in bipolar disorder with depression: a double-blind, placebo-controlled study. J Clin Psychopharmacol 2014; 34:749–751Crossref, Medline, Google Scholar
37 : Naltrexone and dysphoria: fact or myth? Am J Addict 2002; 11:151–160Crossref, Medline, Google Scholar
38 : The behavioral effects of the antidepressant tianeptine require the mu-opioid receptor. Neuropsychopharmacology 2017; 42:2052–2063Crossref, Medline, Google Scholar
39 : Current and historical concepts of opiate treatment in psychiatric disorders. Int Clin Psychopharmacol 1988; 3:255–266Crossref, Medline, Google Scholar
40 : Opioid receptors: distinct roles in mood disorders. Trends Neurosci 2013; 36:195–206Crossref, Medline, Google Scholar
41 : Opioid modulation with buprenorphine/samidorphan as adjunctive treatment for inadequate response to antidepressants: a randomized double-blind placebo-controlled trial. Am J Psychiatry 2016; 173:499–508Link, Google Scholar
42 : Ultra-low-dose buprenorphine as a time-limited treatment for severe suicidal ideation: a randomized controlled trial. Am J Psychiatry 2016; 173:491–498Link, Google Scholar
43 : Buprenorphine treatment of refractory depression. J Clin Psychopharmacol 1995; 15:49–57Crossref, Medline, Google Scholar
44 : Defensive behaviors in infant rhesus monkeys: environmental cues and neurochemical regulation. Science 1989; 243:1718–1721Crossref, Medline, Google Scholar
45 : Opiate modulation of separation-induced distress in non-human primates. Brain Res 1988; 440:285–292Crossref, Medline, Google Scholar
46 : Randomized controlled crossover trial of ketamine in obsessive-compulsive disorder: proof-of-concept. Neuropsychopharmacology 2013; 38:2475–2483Crossref, Medline, Google Scholar
47 : Double-blind treatment with oral morphine in treatment-resistant obsessive-compulsive disorder. J Clin Psychiatry 2005; 66:353–359Crossref, Medline, Google Scholar
48 : Asymmetrical distribution of beta-endorphin in cerebral hemispheres of suicides: preliminary data. Psychiatry Res 1990; 32:159–166Crossref, Medline, Google Scholar
49 : Regionally selective increases in mu opioid receptor density in the brains of suicide victims. Brain Res 1990; 530:312–316Crossref, Medline, Google Scholar
50 : Increased density of mu-opioid receptors in the postmortem brain of suicide victims. Brain Res 1995; 682:245–250Crossref, Medline, Google Scholar
51 : Kappa-opioid ligands in the study and treatment of mood disorders. Pharmacol Ther 2009; 123:334–343Crossref, Medline, Google Scholar
52 : Serotonin and norepinephrine uptake inhibiting activity of centrally acting analgesics: structural determinants and role in antinociception. J Pharmacol Exp Ther 1995; 274:1263–1270Medline, Google Scholar
53 : New opioid receptor antagonist: naltrexone-14-O-sulfate synthesis and pharmacology. Eur J Pharmacol 2017; 809:111–121Crossref, Medline, Google Scholar
54 : Differences in delta- and mu-opioid receptor blockade measured by positron emission tomography in naltrexone-treated recently abstinent alcohol-dependent subjects. Neuropsychopharmacology 2008; 33:653–665Crossref, Medline, Google Scholar
55 : Depressive-like effects of the kappa opioid receptor agonist salvinorin A are associated with decreased phasic dopamine release in the nucleus accumbens. Psychopharmacology (Berl) 2010; 210:241–252Crossref, Medline, Google Scholar
56 : Kappa opioids, salvinorin A, and major depressive disorder. Curr Neuropharmacol 2016; 14:165–176Crossref, Medline, Google Scholar
57 : Stereoselective interaction of ketamine with recombinant mu, kappa, and delta opioid receptors expressed in Chinese hamster ovary cells. Anesthesiology 1999; 90:174–182Crossref, Medline, Google Scholar
58 : Ketamine and its metabolite (2R,6R)-hydroxynorketamine induce lasting alterations in glutamatergic synaptic plasticity in the mesolimbic circuit. Mol Psychiatry (Epub ahead of print, Nov 21, 2017)Google Scholar
59 : Ketamine enhances structural plasticity in mouse mesencephalic and human iPSC-derived dopaminergic neurons via AMPAR-driven BDNF and mTOR signaling. Mol Psychiatry 2017; 23:812–823Crossref, Medline, Google Scholar
60 : Effects of a ketamine metabolite on synaptic NMDAR function. Nature 2017; 546:E1–E3Crossref, Medline, Google Scholar
61 : mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010; 329:959–964Crossref, Medline, Google Scholar
62 : The role of eukaryotic elongation factor 2 kinase in rapid antidepressant action of ketamine. Biol Psychiatry 2013; 73:1199–1203Crossref, Medline, Google Scholar
63 : Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med 2016; 22:238–249Crossref, Medline, Google Scholar
64 : Increase in PAS-induced neuroplasticity after a treatment course of intranasal ketamine for depression: report of three cases from a placebo-controlled trial. Compr Psychiatry 2017; 73:31–34Crossref, Medline, Google Scholar
65 : A systematic review of the clinimetric properties of the 6-item version of the Hamilton Depression Rating Scale (HAM-D6). Psychother Psychosom 2017; 86:141–149Crossref, Medline, Google Scholar
66 : Ketamine for pain. F1000 Res 2017; 6(F1000 Faculty Rev):1711 (doi: 10.12688/f1000research.11372.1)Crossref, Medline, Google Scholar
67 : Interaction of the chiral forms of ketamine with opioid, phencyclidine, sigma and muscarinic receptors. Pharmacol Toxicol 1995; 77:355–359Crossref, Medline, Google Scholar
68 : Dextromethorphan and ketamine potentiate the antinociceptive effects of mu- but not delta- or kappa-opioid agonists in a mouse model of acute pain. Pharmacol Biochem Behav 2002; 74:73–86Crossref, Medline, Google Scholar
69 : Comparative pharmacology of the optical isomers of ketamine in mice. Eur J Pharmacol 1978; 49:15–23Crossref, Medline, Google Scholar
70 : The involvement of the mu-opioid receptor in ketamine-induced respiratory depression and antinociception. Anesth Analg 2001; 93:1495–1500Crossref, Medline, Google Scholar
71 : Differential modulation of remifentanil-induced analgesia and postinfusion hyperalgesia by S-ketamine and clonidine in humans. Anesthesiology 2003; 99:152–159Crossref, Medline, Google Scholar
72 . Ketamine: more mechanisms of action than just NMDA blockade. Trends in anaesthesia and critical care. 2014;4(2):76-81.Crossref, Google Scholar
73 : Opiate receptor mediation of ketamine analgesia. Anesthesiology 1982; 56:291–297Crossref, Medline, Google Scholar
74 : Properties of the interaction between ketamine and opiate binding sites in vivo and in vitro. Neuropharmacology 1987; 26:1253–1260Crossref, Medline, Google Scholar
75 : Antidepressant-like effects of ketamine, norketamine, and dehydronorketamine in forced swim test: role of activity at NMDA receptor. Neuropharmacology 2015; 99:301–307Crossref, Medline, Google Scholar
76 : Frequent colocalization of mu opioid and NMDA-type glutamate receptors at postsynaptic sites in periaqueductal gray neurons. J Comp Neurol 1999; 408:549–559Crossref, Medline, Google Scholar
77 : The mu-opioid receptor and the NMDA receptor associate in PAG neurons: implications in pain control. Neuropsychopharmacology 2012; 37:338–349Crossref, Medline, Google Scholar
78 : It’s MORe exciting than mu: crosstalk between mu opioid receptors and glutamatergic transmission in the mesolimbic dopamine system. Front Pharmacol 2014; 5:116Crossref, Medline, Google Scholar
79 : The role of science in addressing the opioid crisis. N Engl J Med 2017; 377:391–394Crossref, Medline, Google Scholar
80 : Increase in suicide in the United States, 1999–2014. NCHS Data Brief, Apr 2016; 241:1–8Google Scholar
81 : Safety, tolerability, and clinical effect of low-dose buprenorphine for treatment-resistant depression in midlife and older adults. J Clin Psychiatry 2014; 75:e785–e793Crossref, Medline, Google Scholar
82 : The influence of prescription opioid use duration and dose on development of treatment resistant depression. Prev Med 2016; 91:110–116Crossref, Medline, Google Scholar
83 : Buprenorphine is a potent kappa-opioid receptor antagonist in pigeons and mice. Eur J Pharmacol 1988; 151:457–461Crossref, Medline, Google Scholar
84 : Buprenorphine has potent kappa opioid receptor antagonist activity. Neuropharmacology 1987; 26:1445–1447Crossref, Medline, Google Scholar
85 : Opioid agonist and antagonist behavioural effects of buprenorphine. Br J Pharmacol 1983; 78:607–615Crossref, Medline, Google Scholar
86 : Prescription opioid use among adults with mental health disorders in the United States. J Am Board Fam Med 2017; 30:407–417Crossref, Medline, Google Scholar
87 : Development of a checklist of short-term and long-term psychological symptoms associated with ketamine use. Shanghai Jingshen Yixue 2015; 27:186–194Google Scholar
88 : The epidemiology and patterns of acute and chronic toxicity associated with recreational ketamine use. Emerg Health Threats J 2011; 4:7107Crossref, Medline, Google Scholar
89 : Emergency department presentation of ketamine abusers in Hong Kong: a review of 233 cases. Hong Kong Med J 2010; 16:6–11Medline, Google Scholar
90 : Frontal white matter abnormalities following chronic ketamine use: a diffusion tensor imaging study. Brain 2010; 133:2115–2122Crossref, Medline, Google Scholar
91 : A review of ketamine abuse and diversion. Depress Anxiety 2016; 33:718–727Crossref, Medline, Google Scholar
92 : Recent trends in drug abuse in China. Acta Pharmacol Sin 2006; 27:140–144Crossref, Medline, Google Scholar
93 : Abuse of ketamine. Br J Psychiatry 1980; 137:303Crossref, Medline, Google Scholar
94 : Chronic ketamine impairs fear conditioning and produces long-lasting reductions in auditory evoked potentials. Neurobiol Dis 2009; 35:311–317Crossref, Medline, Google Scholar
95 : Long-term heavy ketamine use is associated with spatial memory impairment and altered hippocampal activation. Front Psychiatry 2014; 5:149Crossref, Medline, Google Scholar
96 : Neurocognitive performance and serial intravenous subanesthetic ketamine in treatment-resistant depression. Int J Neuropsychopharmacol 2014; 17:1805–1813Crossref, Medline, Google Scholar
97 : Differential effects of single and repeated ketamine administration on dopamine, serotonin, and GABA transmission in rat medial prefrontal cortex. Brain Res 1997; 759:205–212Crossref, Medline, Google Scholar
98 : Long-term ketamine self-injections in major depressive disorder: focus on tolerance in ketamine’s antidepressant response and the development of ketamine addiction. J Psychoactive Drugs 2015; 47:276–285Crossref, Medline, Google Scholar
99 : Relapse on ketamine followed by severe and prolonged withdrawal: a cautionary case and review of potential medical therapies. J Nat Sci 2017; 3:e450Google Scholar
100 : Development of tolerance to ketamine in an infant undergoing repeated anesthesia. Anesthesiology 1981; 54:255–256Crossref, Medline, Google Scholar
101 : The development of an acute tolerance to ketamine. Anesth Analg 1976; 55:788–791Crossref, Medline, Google Scholar
102 : Tolerance to rectal ketamine in paediatric anaesthesia. Anaesthesia 1981; 36:1089–1093Crossref, Medline, Google Scholar
103 : [A case of ketamine dependence]. Psychiatr Pol 2009; 43:593–599 (Polish)Medline, Google Scholar
104 : A case of ketamine dependence with discontinuation symptoms. Addiction 2006; 101:1212–1213Crossref, Medline, Google Scholar
105 : [A case of ketamine dependence]. Psychiatr Pol 1987; 21:434–435 (Polish)Medline, Google Scholar
106 : Ketamine as a preferred substance of abuse. Am J Addict 2002; 11:332–334Crossref, Medline, Google Scholar
107 : Ketamine associated psychedelic effects and dependence. Singapore Med J 2003; 44:31–34Medline, Google Scholar
108 : Rapid development of tolerance to sub-anaesthetic dose of ketamine: an oculomotor study in macaque monkeys. Psychopharmacology (Berl) 2010; 209:313–318Crossref, Medline, Google Scholar
109 : The development of tolerance to ketamine in rats and the significance of hepatic metabolism. Br J Pharmacol 1978; 64:63–69Crossref, Medline, Google Scholar
110 : Side-effects associated with ketamine use in depression: a systematic review. Lancet Psychiatry 2018; 5:65–78Crossref, Medline, Google Scholar
111 : Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry 2018; 75:139–148Crossref, Medline, Google Scholar
112 : The practicalities and ethics of ketamine for depression. Lancet Psychiatry 2017; 4:354–355Crossref, Medline, Google Scholar
113 : Ketamine treatment for depression: opportunities for clinical innovation and ethical foresight. Lancet Psychiatry 2017; 4:419–426Crossref, Medline, Google Scholar
114 : Potential risks of poorly monitored ketamine use in depression treatment. Am J Psychiatry 2016; 173:215–218Link, Google Scholar
115 : How Many Subjects? Statistical Power Analysis in Research, 2nd ed. Los Angeles, Sage Publications, 2016Crossref, Google Scholar
116 : Effects of the mu-opioid receptor antagonist GSK1521498 on hedonic and consummatory eating behaviour: a proof of mechanism study in binge-eating obese subjects. Mol Psychiatry 2013; 18:1287–1293Crossref, Medline, Google Scholar
117 : Ketamine-induced reduction in mGluR5 availability is associated with an antidepressant response: an [11C]ABP688 and PET imaging study in depression. Mol Psychiatry 2018; 23:824–832Crossref, Medline, Google Scholar
118 : The neurobiology of depression: revisiting the serotonin hypothesis, II: genetic, epigenetic, and clinical studies. Philos Trans R Soc Lond B Biol Sci 2013; 368:20120535Crossref, Medline, Google Scholar
119 : The neurobiology of depression: revisiting the serotonin hypothesis, I: cellular and molecular mechanisms. Philos Trans R Soc Lond B Biol Sci 2012; 367:2378–2381Crossref, Medline, Google Scholar



