Ketamine and Other NMDA Antagonists: Early Clinical Trials and Possible Mechanisms in Depression
Abstract
Objective:
The authors conducted a systematic review and meta-analysis of ketamine and other N-methyl-d-aspartate (NMDA) receptor antagonists in the treatment of major depression.
Method:
Searches of MEDLINE, PsycINFO, and other databases were conducted for placebo-controlled, double-blind, randomized clinical trials of NMDA antagonists in the treatment of depression. Primary outcomes were rates of treatment response and transient remission of symptoms. Secondary outcomes included change in depression symptom severity and the frequency and severity of dissociative and psychotomimetic effects. Results for each NMDA antagonist were combined in meta-analyses, reporting odds ratios for dichotomous outcomes and standardized mean differences for continuous outcomes.
Results:
Ketamine (seven trials encompassing 147 ketamine-treated participants) produced a rapid, yet transient, antidepressant effect, with odds ratios for response and transient remission of symptoms at 24 hours equaling 9.87 (4.37–22.29) and 14.47 (2.67–78.49), respectively, accompanied by brief psychotomimetic and dissociative effects. Ketamine augmentation of ECT (five trials encompassing 89 ketamine-treated participants) significantly reduced depressive symptoms following an initial treatment (Hedges’ g=0.933) but not at the conclusion of the ECT course. Other NMDA antagonists failed to consistently demonstrate efficacy; however, two partial agonists at the NMDA coagonist site, d-cycloserine and rapastinel, significantly reduced depressive symptoms without psychotomimetic or dissociative effects.
Conclusions:
The antidepressant efficacy of ketamine, and perhaps D-cycloserine and rapastinel, holds promise for future glutamate-modulating strategies; however, the ineffectiveness of other NMDA antagonists suggests that any forthcoming advances will depend on improving our understanding of ketamine’s mechanism of action. The fleeting nature of ketamine’s therapeutic benefit, coupled with its potential for abuse and neurotoxicity, suggest that its use in the clinical setting warrants caution.
The emergence of intravenous ketamine therapy has been celebrated as perhaps “the most important breakthrough in antidepressant treatment in decades” (1). However, concern has been raised that off-label clinical utilization of ketamine as a pharmacotherapeutic agent is outpacing scientific scrutiny and may invite adverse sequelae that will exceed any accrued therapeutic benefit (2–4).
The flurry of interest in the antidepressant utility of ketamine and other N-methyl-d-aspartate (NMDA) receptor antagonists has been driven by a confluence of forces. First are the shortcomings of the current antidepressant armamentarium. The failings of existing antidepressants, which are largely thought to work primarily by enhancing monoamine neurotransmission, as well as the clear deficiencies of the underlying monoamine hypothesis of depression (5), are well-documented. When used to treat depression, currently available antidepressants are hindered by a prolonged delay of onset of action and disappointing remission rates (6). Both weaknesses are likely attributable, at least in part, to the fact that current antidepressants work via indirect mechanism(s) of action. There is considerable evidence that the therapeutic activity of antidepressants is not mediated by their direct synaptic effects, on for example monoamine reuptake, but by the brain’s adaptive response to sustained increases in monoaminergic neurotransmission produced by these agents, in a manner akin to the emergence of tolerance in the context of chronic use of habit-forming substances (7). Whatever the precise mechanism of action of currently available antidepressants truly is, their less than optimal efficacy has now been well established in large-scale clinical trials such as the Sequenced Treatment Alternatives to Relieve Depression Study (8–13) and the International Study to Predict Optimized Treatment in Depression (14).
The concatenation of the unsatisfactory remission rates and the delayed therapeutic response plaguing current antidepressants highlight the important unfilled need for an improved antidepressant pharmacopoeia, especially in view of the mortality (e.g., suicide and risk for heart disease and other major medical disorders) and morbidity associated with unremitted depression (15–18). Such unmet needs may be overcome by identifying interventions that more directly address the underlying pathophysiology of depression. In fact, this assumption has been the driving force behind much of the effort to identify novel antidepressant compounds over the past decade. Unfortunately, the search for novel compounds has been remarkably unsuccessful in recent years, resulting in a stagnant developmental pipeline for new antidepressant agents (19–22). It is understandable, given this otherwise bleak picture, that promising results from clinical and preclinical antidepressant studies of NMDA receptor antagonists would generate considerable excitement.
Nearly 20 years ago, several lines of evidence pointed to aberrant NMDA receptor-mediated glutamate neurotransmission as a viable neurobiological substrate on which to base a novel intervention for depression. Evidence includes alterations in central NMDA receptor binding profiles of rodents exposed to chronic stress, a laboratory animal model of depression, and postmortem tissue from suicide victims, in addition to changes in NMDA receptor activity produced by chronic antidepressant exposure (23). Indeed, the evidence that glutamatergic agents might hold antidepressant efficacy dates as far back as 50 years ago (24–26).
The complex physiology of the NMDA receptor (Figure 1) offers numerous pharmacodynamic targets for intervention. A tetramer, composed of two GluN1 subunits and two GluN2 subunits, encompassing an ion channel that regulates neuronal influx of calcium (Ca++) in addition to sodium (Na+) influx and potassium (K+) efflux, the NMDA receptor is unique in that it possesses both a ligand gate and a voltage gate, each of which must be opened to enable ion flow. Furthermore, the ligand gate is opened only when concurrently activated by two ligand molecules, a receptor agonist, glutamate, and a receptor coagonist, either glycine or d-serine, and the voltage gate is opened only when neuronal depolarization is triggered elsewhere (e.g., via glutamate binding the α–amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid [AMPA] receptors, acetylcholine binding at α7 nicotinic receptors, etc.). Additional complexity is conferred by the presence of NMDA receptors not only within the synapse but at extrasynaptic sites as well. The origin of endogenous ligands at extrasynaptic NMDA receptors, though obscure, may be a variety of sources, including spillover of synaptic glutamate, glial release of glutamate, glycine, and d-serine, neuronal release of glycine and d-serine, and capillary extravasation of serum glycine (27), implying a complex physiological regulation. Moreover, activation of extrasynaptic NMDA receptors has been implicated in neuronal toxicity, whereas synaptic NMDA receptor activation has been credited with promoting neuronal survival (28). The respective roles of synaptic and extrasynaptic NMDA receptors in mediating synaptic plasticity and neuronal toxicity are likely more complex and remain a focus of intense scrutiny (27, 29, 30).
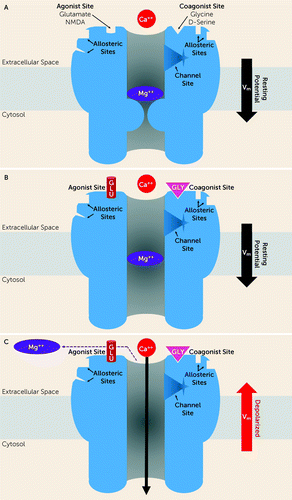
FIGURE 1. N-Methyl-d-Aspartate (NMDA) Receptor Physiology Resting State, Activated State, and Active Statea
a In the NMDA receptor’s A) resting state, both the ligand and voltage gates are closed. The agonist and coagonist sites are unoccupied, and the transmembrane resting potential permits a magnesium (Mg++) ion to block the channel. An array of ligand binding sites have been identified, including the agonist and coagonist sites, which regulate the receptor’s ligand gating, various allosteric sites, and a channel binding site, sometimes called the phencyclidine site, within the receptor’s ion channel. In the B) activated state, glutamate and glycine binding at the agonist and coagonist sites, respectively, has opened the ligand gate; however, ion flow blocked by the Mg++ ion, is held in place within the ion channel by the transmembrane resting potential. In the C) active state, neuronal depolarization has permitted the Mg++ ion to escape the ion channel, thereby opening the NMDA receptor’s voltage gate. Depolarization of the neuron is triggered by glutamate binding to another receptor (i.e., the α–amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor [not shown]). Concurrent opening of both the ligand and voltage gates enables neuronal influx of calcium (Ca++), in addition to sodium (Na+) influx and potassium (K+) efflux, through the receptor ion channel.
There is growing evidence of antidepressant effects of many of the compounds listed in the data supplement accompanying the online version of this article. For example, a series of preclinical antidepressant screening test studies have demonstrated antidepressant-like effects (e.g., decreased immobility time in the forced swim test) for numerous NMDA receptor antagonists, including ketamine (31–35), memantine (36–38), ifenprodil (39), and D-cycloserine (39, 40).
A plethora of open-label trials of ketamine reporting a 25%−78% reduction in depressive symptom severity have been published in recent years (41–72), including those demonstrating particular antidepressant efficacy among those with family histories of alcoholism (73–77), as well as positive open-label studies of amantadine (78) and memantine (79).
The objective of the present review by the APA Council of Research Task Force on Novel Biomarkers and Treatments was to conduct a systematic review and meta-analysis of the randomized clinical trials of ketamine and other NMDA receptor antagonists in the treatment of depression, critically examining findings for both the efficacy and adverse effects of these various agents.
Method
Search
We searched MEDLINE, PsycINFO, the Cochrane Central Register of Controlled Trials, the Cumulative Index to Nursing and Allied Health Literature, and Google Scholar through May 2015 for peer-reviewed articles published in English and addressing treatment of major depression (including major depressive episodes of bipolar disorder) using ketamine, memantine, and other NMDA antagonists. In addition, we searched ClinicalTrials.gov and screened references of included studies and relevant reviews. Only placebo-controlled, double-blind, randomized clinical trials reporting response to treatment utilizing a standardized rating scale for depression were eligible for inclusion.
Data Extraction and Outcomes
All data were extracted by the same reviewer (D.J.N.). The primary outcome measures were treatment response rate, defined as the proportion of patients experiencing a 50% reduction in the total score of the depression rating scale, and rate of transient remission of symptoms, defined as the depression rating scale score falling below a generally accepted threshold value. In addition, we evaluated change in depression symptom severity utilizing each study’s primary depression rating scale. All contributing studies utilized either the Montgomery-Åsberg Depression Rating Scale (MADRS) or the Hamilton Depression Rating Scale (HAM-D). The frequency and/or severity of dissociative, psychotomimetic, and hemodynamic effects were evaluated as secondary outcomes.
Meta-Analytic Calculations
Odds ratios were used for the dichotomous (i.e., treatment response and transient remission of symptom) measures. Adjustment for zero frequency cells was made by adding a fixed value (0.5) to any zero count cells prior to odds ratio calculation. Standardized mean differences were calculated as the mean difference in depressive symptom rating produced by the intervention and the control divided by the pooled standard deviation, with Hedges’ g adjustment for small samples (80). When standard deviations were not directly reported in the article, they were calculated from other available data when possible (e.g., from 95% confidence intervals or t test p values).
The risk of small study effects was assessed through visual inspection of contour-enhanced funnel plots (81) followed by an Egger regression test (82, 83) to formally test for small study effects.
Statistical analyses were performed using Comprehensive Meta Analysis software version 2.2 (Biostat, Frederick, Md.) and SAS software version 9.3 (SAS Institute, Cary, N.C.). All statistical tests were two-tailed with α set at 0.05.
Results
The literature search yielded 581 citations. Most publications were excluded because they reported data from nonrandomized or open-label trials, addressed treatment efficacy for illnesses other than mood disorders, reported the response to treatment of an isolated depressive symptom (i.e., suicidality) rather than the overall syndrome, or were review articles or commentaries on the topic. Twenty-four studies (Table 1 [also see TS2 in the online data supplement]) fulfilled the a priori criteria for meta-analysis inclusion.
| Source | Design | Ketamine Regimen | Control Regimen | Concomitant Therapy | Diagnoses | Sample Size | Depression Scale |
|---|---|---|---|---|---|---|---|
| Ketamine monotherapy | |||||||
| Berman et al. (90) | Cross Over | 0.5 mg/kg intravenous×1 | Placebo | None | Major depressive disorder, bipolar disorder | 8 | 25-Item Hamilton Depression Rating Scale (HAM-D) |
| Lapidus et al. (84) | Cross Over | 0.5 mg intranasal×1 | Placebo | None | Major depressive disorder | 18 | Montgomery-Åsberg Depression Rating Scale (MADRS) |
| Murrough et al. (87) | Parallel | 0.5 mg/kg intravenous×1 | Midazolama | None | Major depressive disorder | 72 | MADRS |
| Zarate et al. (88) | Cross Over | 0.5 mg/kg intravenous×1 | Placebo | None | Major depressive disorder | 17 | 21-Item HAM-D |
| Ketamine augmentation of psychotropic | |||||||
| Diazgranados et al. (85) | Cross Over | 0.5 mg/kg intravenous×1 | Placebo | Lithium or valproic acid | Bipolar disorder | 16 | MADRS |
| Sos et al. (91) | Cross Over | 0.54 mg/kg intravenous×1 | Placebo | Various | Major depressive disorder | 27 | MADRS |
| Zarate et al. (86) | Cross Over | 0.5 mg/kg intravenous×1 | Placebo | Lithium or valproic acid | Bipolar disorder | 14 | MADRS |
| Ketamine augmentation of ECT | |||||||
| Abdallah et al. (95) | Parallelb | 0.5 mg/kg intravenous pre-ECT | Placebo | ECT+thiopental | Major depressive disorder, bipolar disorder | 16 | 25-Item HAM-D |
| Järventausta et al. (93) | Parallelb | 0.4 mg/kg intravenous pre-ECTc | Placebo | ECT+propofol | Major depressive disorder | 49 | MADRS |
| Loo et al. (96) | Parallelb | 0.5 mg/kg intravenous pre-ECT | Placebo | ECT+thiopentone | Major depressive disorder, bipolar disorder | 46 | MADRS |
| Wang et al. (94) | Paralleld | 0.8 mg/kg intravenous pre-ECT | Propofola | ECT | Major depressive disorder | 40 | 17-Item HAM-D |
| Yoosefi et al. (97) | Parallele | 1–2 mg/kg intravenous pre-ECT | Thiopentala | ECT | Major depressive disorder | 29 | HAM-D |
TABLE 1. Characteristics of Included Randomized Clinical Trials of Ketamine
Ketamine Studies
The search identified 12 reports of randomized clinical trials examining ketamine in the treatment of depression (Table 1). The 12 studies encompass ketamine used as a monotherapy, to augment other psychotropic agents, and as an augmentation to ECT. In addition, ketamine was administered in these studies to treat depressive episodes of bipolar disorder, major depressive disorder, or both. It was administered through intravenous infusion in all but one of the studies, which utilized intranasal administration (84). Four of the studies specified that participants had failed a previous or current treatment for depression (84–87).
Efficacy.
A single intravenous infusion of ketamine, excluding its use in conjunction with ECT, which is examined separately, consistently produced a rapid antidepressant response peaking within one day of administration (Table 2). Twenty-four hours after administration, each of the six contributing studies demonstrated a statistically significant odds ratio for treatment response (Figure 2A). The composite odds ratio for therapeutic response at 24 hours postinfusion equaled 9.87 (95% confidence interval [CI]=4.37–22.29, z=5.50, p<0.001). Day 1 response rates remained significant when the data were stratified by diagnosis, separating major depressive disorder (odds ratio=8.42 [95% CI=3.47–20.39] z=4.72, p<0.001) and bipolar disorder (odds ratio=24.05 [95% CI=2.96–195.56] z=2.97, p=0.003). Moreover, the response rate was also significant when limited to patients receiving ketamine as a monotherapy for major depressive disorder (odds ratio=7.55 [95% CI=2.89–19.76] z=4.12, p<0.001).
| Time Posttreatment Initiation | Treatment Response (Percentage) | Transient Symptom Remission (Percentage) | Study | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ketamine | Control | Odds Ratio | 95% CI | p | Ketamine | Control | Odds Ratio | 95% CI | p | ||
| 40 Minutes | 36.9% | 1.5% | 13.2 | 3.2–53.7 | <0.001 | 6.4% | 0.0% | 2.6 | 0.5–13.8 | 0.26 | Diazgranados et al. (85); Lapidus et al. (84); Zarate et al. (86); Zarate et al. (88) |
| 80 Minutes | 51.1% | 2.1% | 24.7 | 5.0–122.5 | <0.001 | 17.0% | 0.0% | 7.3 | 1.4–39.3 | 0.02 | Diazgranados et al. (85); Zarate et al. (86); Zarate et al. (88) |
| 2 Hours | 51.1% | 2.1% | 24.7 | 5.0–122.5 | <0.001 | 23.4% | 0.0% | 10.3 | 1.9–55.8 | 0.007 | Diazgranados (85); Zarate et al. (86); Zarate et al. (88) |
| 4 Hours | 47.7% | 1.5% | 24.4 | 6.0–99.5 | <0.001 | 25.5% | 0.0% | 11.8 | 2.2–64.1 | 0.004 | Diazgranados et al. (85); Lapidus et al. (84); Zarate et al. (86); Zarate et al. (88) |
| 1 Day | 52.6% | 7.0% | 9.9 | 4.4–22.3 | <0.001 | 29.8% | 0.0% | 14.5 | 2.7–78.5 | <0.002 | Diazgranados et al. (85); Lapidus et al. (84); Murrough et al. (87); Sos et al. (91); Zarate et al. (86); Zarate et al. (88) |
| 2 Days | 50.0% | 6.9% | 8.4 | 3.4–20.4 | <0.001 | 21.3% | 0.0% | 8.4 | 1.6–45.0 | <0.01 | Diazgranados et al. (85); Lapidus et al. (84); Murrough et al. (87); Zarate et al. (86); Zarate et al. (88) |
| 3 Days | 46.6% | 8.9% | 7.1 | 3.3–14.9 | <0.001 | 19.1% | 2.1% | 5.6 | 1.2–27.1 | <0.03 | Berman et al. (90); Diazgranados et al. (85); Lapidus et al. (84); Murrough et al. (87); Sos et al. (91); Zarate et al. (86); Zarate et al. (88) |
| 7 Days | 31.4% | 7.0% | 4.6 | 2.1–10.2 | <0.001 | 14.9% | 2.1% | 3.1 | 0.6–15.4 | <0.17 | Diazgranados et al. (85); Lapidus et al. (84); Murrough et al. (87); Sos et al. (91); Zarate et al. (86); Zarate et al. (88) |
| 14 Days | 10.9% | 0.0% | 4.4 | 1.0–18.8 | 0.05 | 2.6% | 0.0% | 1.5 | 0.3–7.9 | <0.65 | Berman et al. (90); Diazgranados et al. (85); Zarate et al. (86); Zarate et al. (88) |
TABLE 2. Results of Meta-Analyses of Ketamine Response and Transient Symptom Remission Rates
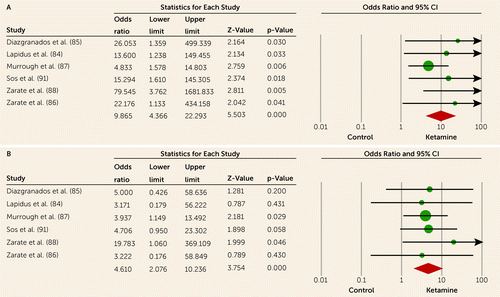
FIGURE 2. Forest Plots of Therapeutic Response Rates One Day and One Week After Initiation of Ketaminea
a The A) top plot shows results one day after initiation of ketamine (heterogeneity: χ2=4.27, df=4, p=0.51, I2=0%). The B) bottom plot shows results one week after initiation of ketamine (heterogeneity: χ2=1.14, df=5, p=0.95, I2=0%).
The odds ratio for treatment response declined steadily but remained statistically significant as long as 2 weeks following ketamine infusion (Table 2). Although only two of the six studies contributing data at day 7 demonstrated a statistically significant odds ratio for treatment response (Figure 2B), the composite odds ratio for response was nevertheless statistically significant (odds ratio=4.61 [95% CI=2.08–10.24], z=3.75, p<0.001). After excluding the intranasal ketamine study, which narrowly missed statistical significance (odds ratio=4.71 [95% CI=0.95–23.30], z=1.90, p=0.058), the five remaining intravenous infusion studies produced a statistically significant composite odds ratio for therapeutic response (odds ratio=4.58 [95% CI=1.82–11.49], z=3.24, p=0.001). Stratified by diagnosis, the odds ratio for treatment response at day 7 was statistically significant for major depressive disorder (odds ratio=4.72 [95% CI=1.95–11.38], z=3.45, p=0.001) but not bipolar disorder (odds ratio=4.16 [95% CI=0.64–27.22], z=1.49, p=0.137).
Odds ratios for transient remission of symptoms followed a similar temporal pattern, though remission rates failed to achieve the same magnitude or to sustain statistical significance beyond day 3 (Table 2). The composite odds ratio for transient symptom remission was statistically significant on day 1 (odds ratio=14.47 [95% CI=2.67–78.49] z=3.10, p=0.002) but not day 7 (odds ratio=3.08 [95% CI=0.61–15.43] z=1.37, p=0.172). Stratifying by diagnosis, major depressive disorder transient remission data were provided by a single study (88), demonstrating identical results on day 1 and day 7 (odds ratio=15.40 [95% CI=0.83–284.53], z=1.83, p=0.066). Studies of bipolar disorder patients, all utilizing ketamine to augment therapy with a conventional mood stabilizer, demonstrated a statistically significant rate of symptom remission on day 1 (odds ratio=14.01 [95% CI=1.73–111.70], z=2.49, p=0.013) but not day 7 (odds ratio=1.51 [95% CI=0.22–10.49], z=0.42, p=0.674).
Of note, ketamine infusion has also been reported to significantly outperform placebo in rapidly reducing suicidal ideation among patients with treatment-resistant depression (89).
Psychotomimetic and dissociative side effects.
Several of the contributing studies administered the positive symptom subscale of the Brief Psychiatric Rating Scale (BPRS) and/or the Clinician-Administered Dissociative States Scale to determine whether ketamine therapy produced psychotomimetic or dissociative symptoms, respectively. The BPRS data (85–88, 90) indicate that psychotomimetic effects were, in fact, associated with ketamine therapy with mean BPRS positive subscale scores 0.74 (95% CI=0.46–1.01) points higher (Hedges’ g=0.82, z=5.73, p<0.001). Similarly, the Clinician-Administered Dissociative States Scale data (85–87) reveal that ketamine therapy was associated with the emergence of transient dissociative symptoms, with mean scores on this scale 23.75 (95% CI=22.13–25.37) points higher (Hedges’ g=1.78, z=7.31, p<0.001) among those receiving ketamine. Some studies (84, 88, 91), though not all (85, 86, 90), reported a statistically significant inverse association between the severity of dissociative or psychotomimetic side effects and subsequent reduction in depressive symptoms.
Hemodynamic side effects.
Despite ketamine’s long-recognized sympathomimetic properties (92), the hemodynamic effects of ketamine were systematically reported in only two of the seven ketamine trials (84, 87), with those studies reporting mean systolic blood pressure increases of 7.6 mm Hg and 19.0 mm Hg, respectively, 40 minutes after infusion. Both studies reported that blood pressure measures had returned to baseline within 4 hours of infusion. Each of the six studies to report adverse event findings (84–88, 91) noted transient blood pressure increases following ketamine administration. Blood pressure changes warranting discontinuation of ketamine infusion (N=2) were observed in only one of the studies.
Ketamine Augmentation of ECT
Efficacy.
The literature search identified five studies examining ketamine use in conjunction with ECT (Table 1). In these studies, ketamine was used either in addition to or in lieu of another agent to induce anesthesia prior to ECT administration. Meta-analysis indicated that ketamine augmentation was associated with a significantly greater reduction in depressive symptoms after an initial ECT session (see Figure S1A in the online data supplement) but not at the conclusion of the complete course of ECT (see Figure S1B in the online data supplement). Moreover, ketamine augmentation did not improve therapeutic response (odds ratio=0.78 [95% CI=0.36–1.68], z=–0.64, p=0.52) at the conclusion of the course of ECT. None of the contributing studies reported symptom remission rates.
Psychotomimetic and dissociative side effects.
None of the ketamine-ECT studies utilized the BPRS or Clinician-Administered Dissociative States Scale measures to evaluate treatment-emergent psychotomimetic or dissociative symptoms. However, one study reported that post-ECT disorientation and restlessness were twice as common among those receiving ketamine (93), and another reported that ketamine was associated with significantly higher rates of post-ECT delirium and fear upon waking due to psychotic symptoms (94).
Hemodynamic side effects.
Cardiovascular effects of ECT augmentation with ketamine were not reported at all in two of the five studies (95, 96) and systematically evaluated in only two of the studies (94, 97). Wang and colleagues (94) reported higher rates of blood pressure elevation among subjects receiving ketamine anesthesia than among those receiving propofol (67% versus 25%, p=0.023), with five (42%) of the ketamine-treated subjects requiring intravenous administration of urapidil to treat the adverse effect; however, coadministration of propofol with ketamine eliminated ketamine’s adverse hemodynamic effects. Yoosefi and colleagues (97) also observed higher blood pressure measures in subjects assigned to ketamine therapy but reported no serious adverse cardiovascular events.
Seizure duration.
Inclusion of ketamine in the ECT anesthetic regimen was associated with longer seizure duration, prolonged by 11.49 seconds (95% CI=8.63–14.34) (Hedges’ g=0.68, z=4.21, p<0.001).
Memantine Studies
Like ketamine, memantine acts as an antagonist by binding to the NMDA receptor at a site within the receptor ion channel. The search identified three reports of randomized clinical trials examining memantine in the treatment of depression (see Table S2 in the online data supplement), including its use as a monotherapy for major depressive disorder (98) and as an augmentation agent either for major depressive disorder (99) or bipolar disorder (100). (Response rate data for use as augmentation for major depressive disorder were provided by Dr. E.G. Smith via personal communication, November 2014 [also see reference 99].) In all three studies, memantine was administered at or about a daily dose of 20 mg during an 8-week trial.
Efficacy.
Meta-analysis of these three studies indicates that memantine did not outperform placebo in achieving a therapeutic response at any biweekly interval during the 8-week trials (Table 3 [also see Figure S2A in the online data supplement]) or in reducing depressive symptom severity (see Figure S2B in the online data supplement). Stratified by diagnosis, the odds ratio for treatment response at week 8 was statistically significant for neither major depressive disorder (odds ratio=0.80 [95% CI=0.19–3.35], z=–0.30, p=0.77) nor bipolar disorder (odds ratio=3.67 [95% CI=0.77–17.43], z=1.63, p=0.10).
| Time Posttreatment Initiation | Treatment Response (Percentage) | Study | ||||
|---|---|---|---|---|---|---|
| Memantine | Control | Odds Ratio | 95% CI | p | ||
| 2 Weeks | 27.6% | 9.7% | 3.8 | 0.9–16.5 | <0.08 | Anand et al. (100); Smith et al. (99) |
| 4 Weeks | 35.5% | 25.8% | 1.7 | 0.5–5.5 | <0.37 | Anand et al. (100); Smith et al. (99) |
| 6 Weeks | 38.7% | 22.6% | 2.7 | 0.9–9.1 | <0.10 | Anand et al. (100); Smith et al. (99) |
| 8 Weeks | 26.7% | 14.9% | 1.6 | 0.6–4.6 | <0.38 | Anand et al. (100); Smith et al. (99); Zarate et al. (98) |
TABLE 3. Results of Meta-Analyses of Memantine Therapeutic Response Rates
Symptom remission rates, reported in only one of the studies (100), were not lower among the memantine-treated group.
Psychotomimetic and dissociative side effects.
The memantine studies did not utilize formal scales such as the BPRS or Clinician-Administered Dissociative States Scale to evaluate adverse effects. However, one of the studies specifically reported having observed no group differences in rates of dissociation or confusion (99), and another reported no differences in “central nervous system side (CNS) effects” (100).
Lanicemine (AZD6765) Studies
Lanicemine, formerly known as AZD6765, also binds to the NMDA receptor at a site within the ion channel. Like ketamine, lanicemine is administered through intravenous infusion. The literature search identified two published reports encompassing three randomized clinical trials examining the antidepressant utility of lanicemine (see Table S2 in the data supplement). These studies, all addressing the treatment of depressive episodes of major depressive disorder, include two trials of single intravenous infusion of lanicemine (101, 102) and an additional trial of serial intravenous infusions administered over 3 weeks at two different doses (102). There are no published randomized clinical trials evaluating lanicemine treatment of depressive episodes of bipolar disorder.
Efficacy.
Whereas one of the two single infusion studies reported a transient statistically significant reduction in depressive symptom severity at 80 and 110 minutes following infusion (101), meta-analysis demonstrated that lanicemine failed to produce a statistically significant reduction in depressive symptoms at 1 day or 3 days following infusion (see Figures S3A and S3B in the data supplement). In the lone study to report rates of response and transient symptom remission (101), no significant differences were observed at any time point (data not shown).
In contrast to the single infusion lanicemine results, serial administration of lanicemine over 3 weeks demonstrated significant improvement in rates of treatment response (odds ratio=2.62 [95% CI=1.33–5.15], z=2.80, p=0.005) and symptom remission (odds ratio=2.33 [95% CI=1.04–5.25], z=2.05, p=0.04) (102).
Psychotomimetic and dissociative side effects.
BPRS data, reported for both single infusion studies (101, 102), indicate that lanicemine therapy was not associated with psychotomimetic side effects because mean scores on the BPRS positive symptom subscale did not differ between the lanicemine and control groups (Hedges’ g=0.04, z=0.17, p=0.87). Similarly, mean Clinician-Administered Dissociative States Scale scores did not differ between groups (Hedges’ g=0.12, z=0.96, p=0.34), suggesting that lanicemine therapy was not associated with dissociative side effects.
Nitrous Oxide Study
Nitrous oxide (N20) is yet another NMDA antagonist that binds to the receptor at a site within the ion channel. In the lone randomized clinical trial published to date (see Table S2 in the data supplement), N20 was administered via inhalation for 1 hour in a 50% N20/50% oxygen mixture. The placebo control was a mixture of 50% nitrogen/50% oxygen. Treatment response was assessed 2 hours and 24 hours after the inhalation (103).
Efficacy.
In this study, N20 was associated with a significantly greater reduction in HAM-D scores than placebo at both 2 hours (−4.8 points versus –2.3 points, p<0.001) and 24 hours (−5.5 points versus –2.8 points, p<0.001) after inhalation. At 24 hours postinhalation, N20 was also associated with higher rates of treatment response (odds ratio=4.0 [95% CI=0.45–35.79]) and transient remission of symptoms (odds ratio=3.0 [95% CI=0.31–28.8]), though neither achieved statistical significance.
Psychotomimetic and dissociative side effects.
The BPRS and Clinician-Administered Dissociative States Scale were not administered in the existing N20 study. However, the authors report evaluating the participants for “the presence of euphoria and psychosis at each time point” and later remark that the intervention “appeared to be devoid of psychotomimetic side effects” (103).
Traxoprodil (CP-101,606) Study
In contrast to the agents reviewed thus far, traxoprodil does not bind to the NMDA receptor at a channel site, binding instead to an allosteric site outside the receptor ion channel on the GluN2B subunit. The lone traxoprodil randomized clinical trial to date (see Table S2 in the data supplement) administered the agent through intravenous infusion to augment the antidepressant paroxetine in a sample who had failed to respond to a 6-week open-label trial of paroxetine and had received a single blind intravenous placebo infusion. Treatment effects were reported using single-sided statistical tests to evaluate treatment effects at 2, 5, 8, 12, and 15 days postinfusion (104).
Efficacy.
When results from this study were analyzed using more conservative two-sided testing, traxoprodil was associated with a significantly greater reduction in MADRS scores at only one of the five postinfusion intervals, 5 days postinfusion (8.9-point difference in means, p=0.01). Because the study reported rates of response and transient symptom remission for those receiving traxoprodil but not those randomly assigned to placebo, odds ratios could not be calculated.
Psychotomimetic and dissociative effects.
Structured assessments, such as the BPRS and Clinician-Administered Dissociative States Scale, were not utilized. Of note, treatment-emergent dissociative symptoms were reported by six (40%) subjects receiving traxoprodil versus two (13%) of those randomly assigned to receive placebo.
MK-0657 (CERC-301) Study
MK-0657 also acts as an NMDA antagonist by binding to the receptor at an allosteric site on the GluN2B subunit. The lone published randomized clinical trial of MK-0657 for depression (see Table S2 in the data supplement) was prematurely terminated when the manufacturer discontinued the program. Thus, only five participants completed the cross-over trial (105).
Efficacy.
In this study, MK-0657 failed to demonstrate a significantly greater reduction of depressive symptoms as measured by the MADRS (p=0.27).
Psychotomimetic and dissociative side effects.
Although the BPRS and Clinician-Administered Dissociative States Scale were administered, results were not reported. The authors did report, however, “no significant difference . . . in the emergence of dissociative or psychotomimetic side effects” (105).
D-Cycloserine Studies
Distinct from the agents previously reviewed, D-cycloserine binds to the glycine coagonist binding site on the NMDA receptor, where it has been shown to act as a partial agonist (106–108). However, additional evidence suggests a more complex pharmacology, confirming that D-cycloserine acts as a partial agonist at glycine binding sites present on GluN2A, GluN2B, and GluN2D subunits of the NMDA receptor, but as a full agonist at glycine binding sites on GluN2C subunits (109, 110).
The literature search identified two randomized clinical trials of D-cycloserine treatment of depression (see Table S2 in the data supplement), both published by the same group (111, 112). The two studies used widely divergent oral D-cycloserine doses, 250 mg (111) versus 1,000 mg (112) per day. Because a dose disparity of this magnitude could dramatically alter the bioactivity of a partial agonist, the two studies are considered independently.
Efficacy.
The earlier lower-dose D-cycloserine trial (111) offered no evidence indicative of efficacy in the treatment of depression. At the 250-mg daily dose, D-cycloserine was not associated with a greater reduction in 21-item HAM-D-measured depressive symptoms than placebo (4.4-point versus 3.1-point reduction, p=0.51).
Conversely, endpoint assessments at the conclusion of the 6-week higher-dose study (112) revealed a significantly greater reduction in depressive symptoms per the 21-item HAM-D (12.0-point versus 3.9-point reduction, p=0.005) in association with D-cycloserine therapy. Moreover, D-cycloserine treatment was associated with a significantly greater likelihood of therapeutic response (54% versus 15%, χ2=4.24, p=0.04); however, the difference in rate of symptom remission (38% versus 15%) did not achieve statistical significance (χ2=1.76, p=0.19).
Interestingly, a subset analysis limited to participants with baseline serum glycine levels exceeding 300 μM demonstrated an especially robust treatment effect, reducing depressive symptoms per the 21-item HAM-D (13.6-point versus 0.1-point reduction, p<0.001). Preclinical studies indicate that D-cycloserine reliably functions as an NMDA antagonist in the context of higher glycine concentrations (106–108), suggesting the same may be true in this clinical trial.
Psychotomimetic and dissociative side effects.
The Positive and Negative Syndrome Scale (PANSS) was used in the lower-dose D-cycloserine study as a proxy to measure psychotomimetic side effects. There were no group differences on the PANSS positive subscale, PANSS negative subscale, or PANSS general scale result.
In the higher-dose D-cycloserine study, the paranoia and depersonalization/derealization items on the HAM-D were used as indices of psychotomimetic and dissociative side effects, respectively. D-cycloserine was not associated with elevation of the HAM-D paranoia item (0.2 [SD=0.4] versus 0.1 [SD=0.2], p=0.31) or depersonalization/derealization item (0.0 [SD=0.0] versus 0.0 [SD=0.0], p=0.72).
Rapastinel (GLYX-13) Study
Like D-cycloserine, rapastinel is a partial agonist at NMDA receptor glycine binding sites (113–115). Whether rapastinel, like D-cycloserine, possesses pharmacodynamic specificity that varies by the NMDA receptor subunit on which the glycine binding site resides is unknown. The literature search identified one randomized clinical trial of rapastinel (116, 117) (see Table S2 in the data supplement), evaluating the antidepressant efficacy of a single intravenous infusion at four doses (1, 5, 10, and 30 mg/kg) with treatment effects assessed at 2, 4, 8, and 12 hours and 1, 3, 7, and 14 days postinfusion.
Efficacy.
No statistically significant differences were observed in rates of treatment response or symptom remission associated with placebo (64% and 42%, respectively) versus rapastinel at any dose (up to 70% and 53%, respectively). However, statistically significant differences in the reduction of the 17-item HAM-D scores were observed for the 5-mg/kg dose at all intervals except day 14 (peak 17-item HAM-D reduction 3.1 points greater than placebo) and the 10-mg/kg dose at day 1 and day 3 (peak 17-item HAM-D reduction 4.3 points greater than placebo). Neither the low (1 mg/kg) nor high (30 mg/kg) rapastinel doses were associated with significant greater 17-item HAM-D score reduction than placebo, leading the authors to posit an inverted U-shape dose response curve.
Psychotomimetic and dissociative side effects.
The BPRS positive symptom subscale was administered in this trial with no differences observed between the placebo group and any of the four rapastinel treatment groups. The Clinician-Administered Dissociative States Scale was not administered in this study.
Discussion
To date, published results of randomized clinical trials examining the antidepressant utility of NMDA antagonists include four ion channel blockers (ketamine, memantine, lanicemine, N20), two antagonists that bind to allosteric sites (traxoprodil, MK-0657), and two partial agonists (D-cycloserine, rapastinel) that bind to the receptor’s glycine coagonist site. Ketamine is not only the most extensively studied NMDA antagonist, with 12 published randomized clinical trials (Table 1), followed by lanicemine (four randomized clinical trials), memantine (three randomized clinical trials), and D-cycloserine (two randomized clinical trials) as the only agents with more than one published clinical trial (see Table S2 in the online data supplement), but is the only NMDA antagonist to date consistently demonstrating antidepressant efficacy across multiple trials.
Current data provide compelling evidence that the antidepressant effects of ketamine infusion are both rapid and robust, albeit transient. For example, the odds ratio for transient symptom remission peaks on postinfusion day 1 at 14.5; however, by day 7, the odds ratio for symptom remission is no longer statistically significant and the odds ratio for treatment response, though statistically significant, has declined to 4.6 from a peak of 24.7. Surprisingly, other NMDA antagonists, including the other ion channel blockers (lanicemine, memantine, and N2O), which bind to the receptor at the same site as ketamine, did not exhibit the same consistent evidence for antidepressant efficacy. It may be noteworthy that ketamine is also distinguished from the other NMDA antagonists by the frequency of psychotomimetic and dissociative side effects. Moreover, although ketamine-associated side effects were transient, reported only on the day of infusion, their occurrence was predictive of improvement of depressive symptoms in some of the ketamine trials (84, 88, 91).
It may be argued that ketamine’s prominent side effects compromise efforts to blind study participants and investigators to treatment assignment, thereby leading to biased results. While plausible, it is difficult to sustain this argument given the uniform evidence of rapid antidepressant efficacy for ketamine across nearly all studies (see Figure 2A [also see Figure S1A in the data supplement]), including those in which therapeutic response was not predicted by the occurrence of psychotomimetic and dissociative side effects (85, 86, 90). A randomized controlled trial comparing ketamine with other psychotomimetic agents previously described to possess anxiolytic and antidepressant properties with a dramatically different mechanism of action, such as psilocybin (118), may be worthwhile to address this potential bias. Alternatively, some have speculated that the psychotomimetic and dissociative side effects of ketamine may be necessary for its antidepressant efficacy (91, 119), suggesting that ketamine may produce its antidepressant, psychotomimetic, and dissociative effects via the same mechanism. However, findings of antidepressant efficacy for other NMDA receptor antagonists in the absence of prominent psychotomimetic or dissociative side effects (102, 112, 116, 117) argue against this contention.
Notably absent in the literature are studies demonstrating that ketamine’s antidepressant effects can be sustained with serial infusions or transition to an alternative maintenance pharmacotherapy. Three small open-label studies of a series of four to six ketamine infusions administered over a 2-week interval reported relapse rates of 55%−89% in the month following treatment (41, 63, 68). Similarly, maintenance therapy with riluzole, a glutamatergic modulator, failed to outperform placebo in sustaining the therapeutic response to a single ketamine infusion in two randomized clinical trials, with relapse rates of 67%–80% among those randomly assigned to riluzole (51, 120). Of note, the literature includes a single case report of a woman who received 41 ketamine infusions yet remained significantly depressed and was ultimately referred for deep brain stimulation (121).
The following concatenation of findings emerges from the meta-analysis of existing NMDA antagonist studies: 1) intravenous infusion of subanesthetic doses of ketamine reliably produces a rapid antidepressant effect; 2) ketamine infusion also commonly produces prominent dissociative and psychotomimetic side effects; 3) ketamine’s therapeutic benefit quickly dissipates; and 4) other NMDA antagonists have failed to match ketamine’s consistent evidence of antidepressant efficacy across multiple randomized clinical trials. These findings, in turn, invite a series of seminal questions: 1) What conclusions regarding ketamine’s antidepressant mechanism of action can be gleaned from the existing clinical trial data? 2) To what extent is ketamine’s emerging use as an antidepressant in the clinical setting supported by the current data? 3) What lies ahead for future research regarding the role of NMDA antagonists in the treatment of depression?
Ketamine’s Mechanism of Action
Exploring the discrepant findings between ketamine and other NMDA antagonists, particularly the other NMDA ion channel blockers whose pharmacodynamic activities most closely resemble ketamine, may help elucidate ketamine’s antidepressant mechanism of action. That other NMDA channel blockers have yet to replicate ketamine’s rapid antidepressant effects has led to speculation that ketamine’s antidepressant properties may not be mediated via the NMDA receptor at all (122). Indeed, ketamine possesses a rich pharmacology, including activity at sigma receptors (123, 124). Moreover, actions within dopaminergic (125–127) or serotonergic (128–132) systems have also been postulated as alternate mechanisms for ketamine’s antidepressant effects. Conversely, ketamine’s unique antidepressant properties may be attributable to distinctions in its pharmacodynamic activity within the NMDA receptor (34, 122).
Comparing ketamine and memantine may be particularly illustrative. Despite the fact that both agents bind to the NMDA receptor at the channel binding site, memantine’s absence of antidepressant efficacy is in stark contrast to the positive ketamine results. Numerous studies, both clinical and preclinical, comparing the pharmacodynamic profiles of ketamine and memantine may explain the apparent inconsistency (see Table S3 in the data supplement). For example, whereas both agents reduce postsynaptic currents in in vitro neuronal cultures, only ketamine does so when magnesium is added to the culture to mimic physiologic conditions (34). This pivotal distinction suggests that ketamine, but not memantine, readily exceeds the physiologic capacity of the NMDA receptor’s magnesium-dependent voltage gating (Figures 1B and 1C) to impede ion flow through the receptor channel. Ketamine’s superior capacity for blocking ion flow is unlikely to be a consequence of differential affinity for the NMDA channel binding site (133) but instead ketamine’s greater propensity, relative to memantine, for becoming trapped, once bound, within the channel (134).
Additional differences have been observed in the capacities for ketamine and memantine to activate intracellular signaling pathways linked to synaptic plasticity. Indeed, some have postulated that ketamine-induced synaptogenesis is crucial to its antidepressant effects (122, 135–137). Ketamine has been more reliably associated with a cascade of increased phosphorylation of eukaryotic elongation factor 2 (eEF2), increased synthesis of brain-derived neurotrophic factor (BDNF), and heightened activation of mammalian target of rapamycin (mTOR) than memantine (see Table S3 in the data supplement). BDNF’s role in ketamine’s mechanism of action is further suggested by the elimination of its antidepressant-like effects in a BDNF knockout model (32). In addition, both a clinical and preclinical study (138, 139) have reported altered antidepressant effects of ketamine in association with a BDNF functional polymorphism. Similarly, pretreatment with rapamycin, an mTOR inhibitor, extinguished ketamine-induced synaptogenesis and antidepressant-like effects in two rodent studies (140, 141).
Activation of these synaptic signaling proteins may not, however, be an immediate consequence of ketamine’s antagonism of the NMDA receptor. Additional evidence indicates that activation of glutamatergic AMPA receptors is necessary for ketamine’s antidepressant effects. Specifically, coadministration of an AMPA receptor antagonist has been shown to block ketamine’s antidepressant-like behavioral effects (32, 142, 143) and ketamine’s induction of synaptogenesis by synaptic signaling proteins (e.g., mTOR and BDNF) (35, 144).
The mechanism whereby ketamine infusion produces glutamatergic activation of AMPA receptors remains obscure. Some studies (145–147), though not all (148, 149), suggest that NMDA antagonists trigger presynaptic release of glutamate, which, in turn, binds to AMPA receptors. No matter what the underlying mechanism, the apparent linkage between NMDA antagonism and AMPA receptor activation may help clarify how ketamine can be neuroprotective (150, 151) in some contexts but potentially neurotoxic (151–153) in others. The necessity of AMPA activation implies that ketamine induces synaptogenesis by increasing glutamate signaling rather than by protecting neurons from glutamate excitotoxicity. Thus, excessive or ill-timed NMDA antagonism by an agent such as ketamine may leave neurons vulnerable to glutamate excitotoxicity. Ketamine’s potential to be either neuroprotective or neurotoxic must be considered when contemplating ketamine therapy in the clinical setting.
Clinical Use of Ketamine
That the encouraging results from published ketamine trials would generate excitement is certainly understandable. Yet, this enthusiasm should be tempered with the realization that ketamine’s clinical trial data, although positive, remain limited and demonstrate only a transient benefit.
Current efficacy data suggest that ketamine infusion provides a rapid therapeutic response for many patients suffering with treatment-resistant depression. It is perhaps understandable that infusion centers, employing ketamine as an alternative to ECT (154), have rapidly appeared across the nation. Upon closer inspection, however, the available data do not support ketamine infusion as an ECT alternative for acute treatment of depression. Whereas relapse rates approach 50% in the 6 months following ECT (155), relapse rates range up to nearly 90% only 4 weeks following serial ketamine infusions (41, 63, 68). As with ECT (156), achieving sustained remission of illness with ketamine may require continuation or maintenance treatment. However, there are currently no data regarding the efficacy and safety of continuation or maintenance phase therapy with ketamine delivered intravenously, intranasally, or via other routes.
Despite the attractiveness of such options as ketamine infusion centers or intranasal ketamine for home use, serious safety concerns remain. First, ketamine, in some contexts, could be neurotoxic. Soriano describes three risk factors for ketamine-induced neurotoxicity: 1) administration in early development during peak synaptogenesis, 2) administration at high doses, and 3) extended exposure (157).
In preclinical studies, ketamine has been associated with neuronal apoptosis in the developing CNS of both rodents (151, 158) and rhesus monkeys (159, 160). Additional evidence suggests that repeated ketamine administration to human infants may adversely affect neurodevelopment (152). However, other data support a neuroprotective role for ketamine in the developing brain by inhibiting inflammation in the context of a noxious stimulus such as pain (151). In addition, ketamine related neuronal apoptosis has been demonstrated in adult rodents (161). It is unclear if ketamine would have similar effects in adult humans.
Reliance upon low, subanesthetic doses in ketamine therapy for depression arguably safeguards against neurotoxic effects. For example, an in vitro study exposing human dopaminergic neurons to varying ketamine concentrations demonstrated apoptosis at high (500 μM) concentrations and evidence of oxidative stress at concentrations consistent with clinical use during anesthesia induction (100 μM); however, lower (20 μM) concentrations that would be typical of subanesthetic administration produced no evidence of neuronal injury (162).
As part of a general anesthetic regimen, ketamine is seldom administered repeatedly over days and weeks. Conversely, used as an intervention for depression, repeated administration of ketamine could potentially have a very different safety profile, including the risk for neurotoxicity. For example, in adult rodents, weekly serial ketamine administration has been observed to lead to the development of locomotor sensitization (163). Unfortunately, to date there is no substantive evidence to clarify whether there exists a duration of exposure safety threshold for ketamine neurotoxicity in adult humans.
An additional concern is the addictive/abuse liability of ketamine (4). Ketamine abuse is a widely recognized social problem in several countries in Europe and Asia, as well as in the United States (164). When abused, the intoxicating effects of ketamine are produced at doses (1 mg/kg–2 mg/kg) that are only marginally higher than the doses used in existing randomized clinical trials of ketamine. Widespread dissemination in the outpatient setting could readily produce physiological and psychological dependence on ketamine. Furthermore, diversion of prescribed ketamine for illicit use could rival, or even exceed, problems currently encountered with prescription opiates and sedative-hypnotics. Indeed, clinicians should be wary of the slippery slope posed by off-label use of ketamine (3). It should be noted that the history of pharmacology is replete with examples of new drug development with the promise of major therapeutic advances leading instead to disastrous public health consequences (e.g., heroin as a less addictive and more effective analgesic than morphine) (165).
Future Research Directions
Forthcoming ketamine research should continue to examine three central concerns: 1) elucidating ketamine’s mechanism of action; 2) understanding the administration profile necessary to provide a sustained therapeutic benefit; and 3) examining ketamine’s safety profile, particularly with repeated and likely low-dose administration.
Among the other NMDA antagonists studied to date, most intriguing are the recent studies of high-dose D-cycloserine (112) and rapastinel (116, 117). Because the therapeutic effect of D-cycloserine is most prominent at a higher dose in the context of high glycine concentrations, the binding site’s endogenous ligand, the partial agonist can be reliably assumed to be functioning as a relative antagonist. Yet, both preclinical (166, 167) and clinical (168–170) studies reveal an inverted U-shape dose-response curve for D-cycloserine, which is not typical for classic partial agonists. Interestingly, the rapastinel randomized clinical trial for depression suggested a similar dose-response curve (116, 117). One plausible explanation is that these agents behave as classic partial agonists within a low (weak agonist activity) to moderate (relative antagonist activity) dose range but at especially high doses exhibit full agonist activity via GluN2C glycine binding sites activation. These agents are certainly worthy of further scrutiny.
Future studies may not be limited to NMDA receptor antagonists. Additional targets within the glutamatergic system include other ionotropic receptors (AMPA, kainate) (171, 172), metabotropic receptors (173), and glutamate transporters.
1 : http://www.nimh.nih.gov/about/director/2014/ketamine.shtmlGoogle Scholar
2 : The ketamine challenge: when practice leaps ahead of science. http://psychnews.psychiatryonline.org/doi/full/10.1176/appi.pn.2015.2a26Google Scholar
3 : A word to the wise about ketamine. Am J Psychiatry 2014; 171:262–264Link, Google Scholar
4 : Proceed with caution: off-label ketamine treatment for major depressive disorder. Curr Psychiatry Rep 2014; 16:527Crossref, Medline, Google Scholar
5 : History and evolution of the monoamine hypothesis of depression. J Clin Psychiatry 2000; 61(Suppl 6):4–6Crossref, Medline, Google Scholar
6 : NIMH-funded pragmatic trials: moving on. Neuropsychopharmacology 2010; 35:2489–2490Crossref, Medline, Google Scholar
7 : Initiation and adaptation: a paradigm for understanding psychotropic drug action. Am J Psychiatry 1996; 153:151–162Link, Google Scholar
8 : A comparison of mirtazapine and nortriptyline following two consecutive failed medication treatments for depressed outpatients: a STAR*D report. Am J Psychiatry 2006; 163:1161–1172Link, Google Scholar
9 : Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry 2006; 163:1531–1541, quiz 1666Link, Google Scholar
10 : A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry 2006; 163:1519–1530, quiz 1665Link, Google Scholar
11 : Bupropion-SR, sertraline, or venlafaxine-XR after failure of SSRIs for depression. N Engl J Med 2006; 354:1231–1242Crossref, Medline, Google Scholar
12 : Medication augmentation after the failure of SSRIs for depression. N Engl J Med 2006; 354:1243–1252Crossref, Medline, Google Scholar
13 : Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry 2006; 163:28–40Link, Google Scholar
14 : The International Study to Predict Optimized Treatment in Depression (iSPOT-D): outcomes from the acute phase of antidepressant treatment. J Psychiatr Res 2015; 61:1–12Crossref, Medline, Google Scholar
15 : Nonresponse, partial response, and failure to achieve remission: humanistic and cost burden in major depressive disorder. Depress Anxiety 2009; 26:83–97Crossref, Medline, Google Scholar
16 : Completeness of response and quality of life in mood and anxiety disorders. Depress Anxiety 2000; 12(Suppl 1):95–101Crossref, Medline, Google Scholar
17 : Update on partial response in depression. J Clin Psychiatry 2009; 70(Suppl 6):4–9Crossref, Medline, Google Scholar
18 : Clinical and functional outcomes of patients who experience partial response to citalopram: secondary analysis of STAR*D. J Psychiatr Pract 2014; 20:178–187Crossref, Medline, Google Scholar
19 Friedman RA: A dry pipeline for psychiatric drugs. New York Times. Aug 19, 2013Google Scholar
20 : The failure of the antidepressant drug discovery process is systemic. J Psychopharmacol 2013; 27:407–413, discussion 413–416Crossref, Medline, Google Scholar
21 : No new meds: with drug firms in retreat, the pipeline for new psychiatric medications dries up. Sci News 2013; 183:26–29Crossref, Google Scholar
22 Yan J: Pipeline for new antidepressants flowing slowly. Psychiatr News. March 2, 2012Google Scholar
23 : Glutamate-based antidepressants: preclinical psychopharmacology. Biol Psychiatry 2013; 73:1125–1132Crossref, Medline, Google Scholar
24 : Cyloserine as an antidepressant agent. Am J Psychiatry 1959; 115:1025–1026Link, Google Scholar
25 : The psychotropic effect of cycloserine: A new use of an antibiotic. Compr Psychiatry 1961; 2:51–59Crossref, Google Scholar
26 : Amantadine in depression. Lancet 1971; 2:437Crossref, Medline, Google Scholar
27 : Organization, control and function of extrasynaptic NMDA receptors. Philos Trans R Soc Lond B Biol Sci 2014; 369:20130601Crossref, Medline, Google Scholar
28 : Extrasynaptic NMDARs oppose synaptic NMDARs by triggering CREB shut-off and cell death pathways. Nat Neurosci 2002; 5:405–414Crossref, Medline, Google Scholar
29 : Extrasynaptic NMDA receptor in excitotoxicity: function revisited. Neuroscientist 2015; 21:337–344Crossref, Medline, Google Scholar
30 : Extrasynaptic glutamate NMDA receptors: key players in striatal function. Neuropharmacology 2015; 89:54–63Crossref, Medline, Google Scholar
31 : Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog Neuropsychopharmacol Biol Psychiatry 2008; 32:140–144Crossref, Medline, Google Scholar
32 : NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011; 475:91–95Crossref, Medline, Google Scholar
33 : Acute suppression of spontaneous neurotransmission drives synaptic potentiation. J Neurosci 2013; 33:6990–7002Crossref, Medline, Google Scholar
34 : Mechanisms underlying differential effectiveness of memantine and ketamine in rapid antidepressant responses. Proc Natl Acad Sci USA 2014; 111:8649–8654Crossref, Medline, Google Scholar
35 : Ketamine-induced antidepressant effects are associated with AMPA receptors-mediated upregulation of mTOR and BDNF in rat hippocampus and prefrontal cortex. Eur Psychiatry 2014; 29:419–423Crossref, Medline, Google Scholar
36 : Potential antidepressive properties of amantadine, memantine and bifemelane. Pharmacol Toxicol 1993; 72:394–397Crossref, Medline, Google Scholar
37 : Synergistic effect of uncompetitive NMDA receptor antagonists and antidepressant drugs in the forced swimming test in rats. Neuropharmacology 2002; 42:1024–1030Crossref, Medline, Google Scholar
38 : Neurochemical and behavioural effects of acute and chronic memantine administration in rats: Further support for NMDA as a new pharmacological target for the treatment of depression? Brain Res Bull 2010; 81:585–589Crossref, Medline, Google Scholar
39 : Effects of ifenprodil on the antidepressant-like activity of NMDA ligands in the forced swim test in mice. Prog Neuropsychopharmacol Biol Psychiatry 2013; 46:29–35Crossref, Medline, Google Scholar
40 : [D-cycloserine is active in the adult mouse and inactive in the aged mouse, in the forced swim test]. Ann Pharm Fr 1998; 56:209–212Medline, Google Scholar
41 : Safety and efficacy of repeated-dose intravenous ketamine for treatment-resistant depression. Biol Psychiatry 2010; 67:139–145Crossref, Medline, Google Scholar
42 : Improvement in suicidal ideation after ketamine infusion: relationship to reductions in depression and anxiety. J Psychiatr Res 2014; 58:161–166Crossref, Medline, Google Scholar
43 : Neural correlates of rapid antidepressant response to ketamine in treatment-resistant unipolar depression: a preliminary positron emission tomography study. Biol Psychiatry 2013; 73:1213–1221Crossref, Medline, Google Scholar
44 : Acute antidepressant effects of intramuscular versus intravenous ketamine. Indian J Psychol Med 2014; 36:71–76Crossref, Medline, Google Scholar
45 : Two case studies of patients with major depressive disorder given low-dose (subanesthetic) ketamine infusions. Pain Med 2006; 7:92–95Crossref, Medline, Google Scholar
46 : Monitoring ketamine treatment response in a depressed patient via peripheral mammalian target of rapamycin activation. Am J Psychiatry 2011; 168:751–752Link, Google Scholar
47 : Rapid resolution of suicidal ideation after a single infusion of an N-methyl-D-aspartate antagonist in patients with treatment-resistant major depressive disorder. J Clin Psychiatry 2010; 71:1605–1611Crossref, Medline, Google Scholar
48 : Baseline delta sleep ratio predicts acute ketamine mood response in major depressive disorder. J Affect Disord 2013; 145:115–119Crossref, Medline, Google Scholar
49 : Rapid relief of severe major depressive disorder by use of preoperative ketamine and electroconvulsive therapy. J ECT 2007; 23:23–25Crossref, Medline, Google Scholar
50 : Rapid decrease in depressive symptoms with an N-methyl-d-aspartate antagonist in ECT-resistant major depression. Prog Neuropsychopharmacol Biol Psychiatry 2011; 35:1155–1159Crossref, Medline, Google Scholar
51 : Course of improvement in depressive symptoms to a single intravenous infusion of ketamine vs add-on riluzole: results from a 4-week, double-blind, placebo-controlled study. Neuropsychopharmacology 2012b; 37:1526–1533Crossref, Medline, Google Scholar
52 : Oral ketamine for the rapid treatment of depression and anxiety in patients receiving hospice care. J Palliat Med 2010; 13:903–908Crossref, Medline, Google Scholar
53 : Clinically favourable effects of ketamine as an anaesthetic for electroconvulsive therapy: a retrospective study. Eur Arch Psychiatry Clin Neurosci 2011; 261:575–582Crossref, Medline, Google Scholar
54 : A preliminary naturalistic study of low-dose ketamine for depression and suicide ideation in the emergency department. Int J Neuropsychopharmacol 2011; 14:1127–1131Crossref, Medline, Google Scholar
55 : Repeated intravenous ketamine therapy in a patient with treatment-resistant major depression. World J Biol Psychiatry 2009; 10:640–643Crossref, Medline, Google Scholar
56 : Brain-derived neurotrophic factor and initial antidepressant response to an N-methyl-D-aspartate antagonist. J Clin Psychiatry 2009; 70:1662–1666Crossref, Medline, Google Scholar
57 : The use of a series of ketamine infusions in two patients with treatment-resistant depression. J Neuropsychiatry Clin Neurosci 2010; 22:442–444Crossref, Medline, Google Scholar
58 : Rapid and longer-term antidepressant effects of repeated ketamine infusions in treatment-resistant major depression. Biol Psychiatry 2013b; 74:250–256Crossref, Medline, Google Scholar
59 : Rapid antidepressant effect of ketamine anesthesia during electroconvulsive therapy of treatment-resistant depression: comparing ketamine and propofol anesthesia. J ECT 2010; 26:223–227Crossref, Medline, Google Scholar
60 : Oral administration of the NMDA receptor antagonist S-ketamine as add-on therapy of depression: a case series. Pharmacopsychiatry 2010; 43:33–35Crossref, Medline, Google Scholar
61 : Comparison of racemic ketamine and S-ketamine in treatment-resistant major depression: report of two cases. World J Biol Psychiatry 2009; 10:241–244Crossref, Medline, Google Scholar
62 : Effects of intravenous ketamine on explicit and implicit measures of suicidality in treatment-resistant depression. Biol Psychiatry 2009; 66:522–526Crossref, Medline, Google Scholar
63 : Serial infusions of low-dose ketamine for major depression. J Psychopharmacol 2013; 27:444–450Crossref, Medline, Google Scholar
64 : Increased anterior cingulate cortical activity in response to fearful faces: a neurophysiological biomarker that predicts rapid antidepressant response to ketamine. Biol Psychiatry 2009; 65:289–295Crossref, Medline, Google Scholar
65 : Anterior cingulate desynchronization and functional connectivity with the amygdala during a working memory task predict rapid antidepressant response to ketamine. Neuropsychopharmacology 2010; 35:1415–1422Crossref, Medline, Google Scholar
66 : An investigation of amino-acid neurotransmitters as potential predictors of clinical improvement to ketamine in depression. Int J Neuropsychopharmacol 2012; 15:1063–1072Crossref, Medline, Google Scholar
67 : Repeated S-ketamine infusions in therapy resistant depression: a case series. J Clin Pharmacol 2013; 53:996–998Crossref, Medline, Google Scholar
68 : Augmentation of response and remission to serial intravenous subanesthetic ketamine in treatment resistant depression. J Affect Disord 2014; 155:123–129Crossref, Medline, Google Scholar
69 : Intravenous ketamine “burst” for refractory depression in a patient with advanced cancer. J Palliat Med 2008; 11:1268–1271Crossref, Medline, Google Scholar
70 : A 12-month naturalistic observation of three patients receiving repeat intravenous ketamine infusions for their treatment-resistant depression. J Affect Disord 2013; 147:416–420Crossref, Medline, Google Scholar
71 : Rapid response with ketamine on suicidal cognition in resistant depression. Indian J Psychol Med 2012; 34:170–175Crossref, Medline, Google Scholar
72 : The antidepressant effect of ketamine is not associated with changes in occipital amino acid neurotransmitter content as measured by [(1)H]-MRS. Psychiatry Res 2011; 191:122–127Crossref, Medline, Google Scholar
73 : Altered NMDA glutamate receptor antagonist response in individuals with a family vulnerability to alcoholism. Am J Psychiatry 2004; 161:1776–1782Link, Google Scholar
74 : Family history of alcohol dependence and initial antidepressant response to an N-methyl-D-aspartate antagonist. Biol Psychiatry 2009; 65:181–184Crossref, Medline, Google Scholar
75 : Family history of alcohol dependence and antidepressant response to an N-methyl-D-aspartate antagonist in bipolar depression. Bipolar Disord 2012; 14:880–887Crossref, Medline, Google Scholar
76 : Ketamine’s antidepressant efficacy is extended for at least four weeks in subjects with a family history of an alcohol use disorder. Int J Neuropsychopharmacol 2014; 18:pyu039. Available at doi:
77 : Clinical predictors of ketamine response in treatment-resistant major depression. J Clin Psychiatry 2014; 75:e417–e423Crossref, Medline, Google Scholar
78 : Amantadine in depressive patients with Borna disease virus (BDV) infection: an open trial. Bipolar Disord 2000; 2:65–70Crossref, Medline, Google Scholar
79 : An open-label, flexible-dose study of memantine in major depressive disorder. Clin Neuropharmacol 2007; 30:136–144Crossref, Medline, Google Scholar
80 : Systematic Reviews in Healthcare: Meta Analysis in Context. London, BMJ Publishing, 2000Google Scholar
81 : Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J Clin Epidemiol 2008; 61:991–996Crossref, Medline, Google Scholar
82 : Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315:629–634Crossref, Medline, Google Scholar
83 : Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol 2000; 53:1119–1129Crossref, Medline, Google Scholar
84 : A randomized controlled trial of intranasal ketamine in major depressive disorder. Biol Psychiatry 2014; 76:970–976. Available at doi:
85 : A randomized add-on trial of an N-methyl-D-aspartate antagonist in treatment-resistant bipolar depression. Arch Gen Psychiatry 2010; 67:793–802Crossref, Medline, Google Scholar
86 : Replication of ketamine’s antidepressant efficacy in bipolar depression: a randomized controlled add-on trial. Biol Psychiatry 2012; 71:939–946Crossref, Medline, Google Scholar
87 : Antidepressant efficacy of ketamine in treatment-resistant major depression: a two-site randomized controlled trial. Am J Psychiatry 2013; 170:1134–1142Link, Google Scholar
88 : A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry 2006; 63:856–864Crossref, Medline, Google Scholar
89 : Effects of ketamine on explicit and implicit suicidal cognition: a randomized controlled trial in treatment-resistant depression. Depress Anxiety 2014; 31:335–343Crossref, Medline, Google Scholar
90 : Antidepressant effects of ketamine in depressed patients. Biol Psychiatry 2000; 47:351–354Crossref, Medline, Google Scholar
91 : Relationship of ketamine’s antidepressant and psychotomimetic effects in unipolar depression. Neuroendocrinol Lett 2013; 34:287–293Medline, Google Scholar
92 : The cardiovascular effects of ketamine in man. Anaesthesia 1976; 31:873–882Crossref, Medline, Google Scholar
93 : Effects of S-ketamine as an anesthetic adjuvant to propofol on treatment response to electroconvulsive therapy in treatment-resistant depression: a randomized pilot study. J ECT 2013; 29:158–161Crossref, Medline, Google Scholar
94 : Effects of propofol and ketamine as combined anesthesia for electroconvulsive therapy in patients with depressive disorder. J ECT 2012; 28:128–132Crossref, Medline, Google Scholar
95 : Rapid antidepressant effect of ketamine in the electroconvulsive therapy setting. J ECT 2012; 28:157–161Crossref, Medline, Google Scholar
96 : Neuropsychological and mood effects of ketamine in electroconvulsive therapy: a randomised controlled trial. J Affect Disord 2012; 142:233–240Crossref, Medline, Google Scholar
97 : Comparing effects of ketamine and thiopental administration during electroconvulsive therapy in patients with major depressive disorder: a randomized, double-blind study. J ECT 2014; 30:15–21Crossref, Medline, Google Scholar
98 : A double-blind, placebo-controlled study of memantine in the treatment of major depression. Am J Psychiatry 2006; 163:153–155Link, Google Scholar
99 : Antidepressant augmentation using the N-methyl-D-aspartate antagonist memantine: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry 2013; 74:966–973Crossref, Medline, Google Scholar
100 : Early antidepressant effect of memantine during augmentation of lamotrigine inadequate response in bipolar depression: a double-blind, randomized, placebo-controlled trial. Bipolar Disord 2012; 14:64–70Crossref, Medline, Google Scholar
101 : A randomized trial of a low-trapping nonselective N-methyl-D-aspartate channel blocker in major depression. Biol Psychiatry 2013; 74:257–264Crossref, Medline, Google Scholar
102 : Lanicemine: a low-trapping NMDA channel blocker produces sustained antidepressant efficacy with minimal psychotomimetic adverse effects. Mol Psychiatry 2014; 19:978–985Crossref, Medline, Google Scholar
103 : Nitrous oxide for treatment-resistant major depression: a proof-of-concept trial. Biol Psychiatry 2015; 78:10–18 Crossref, Medline, Google Scholar
104 : An innovative design to establish proof of concept of the antidepressant effects of the NR2B subunit selective N-methyl-D-aspartate antagonist, CP-101,606, in patients with treatment-refractory major depressive disorder. J Clin Psychopharmacol 2008; 28:631–637Crossref, Medline, Google Scholar
105 : A Randomized, placebo-controlled, crossover pilot trial of the oral selective NR2B antagonist MK-0657 in patients with treatment-resistant major depressive disorder. J Clin Psychopharmacol 2012; 32:551–557Crossref, Medline, Google Scholar
106 : Actions of D-cycloserine at the N-methyl-D-aspartate-associated glycine receptor site in vivo. Neuropharmacology 1991; 30:1167–1171Crossref, Medline, Google Scholar
107 : D-cycloserine: a ligand for the N-methyl-D-aspartate coupled glycine receptor has partial agonist characteristics. Neurosci Lett 1989; 98:91–95Crossref, Medline, Google Scholar
108 : D-cycloserine acts as a partial agonist at the glycine modulatory site of the NMDA receptor expressed in Xenopus oocytes. Brain Res 1990; 510:158–160Crossref, Medline, Google Scholar
109 : Subunit specificity and mechanism of action of NMDA partial agonist D-cycloserine. Neuropharmacology 2001; 41:151–158Crossref, Medline, Google Scholar
110 : Structural determinants of D-cycloserine efficacy at the NR1/NR2C NMDA receptors. J Neurosci 2010; 30:2741–2754Crossref, Medline, Google Scholar
111 : Controlled trial of D-cycloserine adjuvant therapy for treatment-resistant major depressive disorder. J Affect Disord 2006; 93:239–243Crossref, Medline, Google Scholar
112 : A randomized add-on trial of high-dose D-cycloserine for treatment-resistant depression. Int J Neuropsychopharmacol 2013; 16:501–506Crossref, Medline, Google Scholar
113 : A NMDA receptor glycine site partial agonist, GLYX-13, simultaneously enhances LTP and reduces LTD at Schaffer collateral-CA1 synapses in hippocampus. Neuropharmacology 2008; 55:1238–1250Crossref, Medline, Google Scholar
114 : GLYX-13: a monoclonal antibody-derived peptide that acts as an N-methyl-D-aspartate receptor modulator. Neuropharmacology 2005; 49:1077–1087Crossref, Medline, Google Scholar
115 : GLYX-13, a NMDA receptor glycine-site functional partial agonist, induces antidepressant-like effects without ketamine-like side effects. Neuropsychopharmacology 2013; 38:729–742Crossref, Medline, Google Scholar
116 : Randomized proof of concept trial of GLYX-13, an N-methyl-D-aspartate receptor glycine site partial agonist, in major depressive disorder nonresponsive to a previous antidepressant agent. J Psychiatr Pract 2015; 21:140–149Crossref, Medline, Google Scholar
117 : GLYX-13, an NMDA receptor glycine site functional partial agonist enhances cognition and produces antidepressant effects without the psychotomimetic side effects of NMDA receptor antagonists. Expert Opin Investig Drugs 2014; 23:243–254Crossref, Medline, Google Scholar
118 : Single treatments that have lasting effects: some thoughts on the antidepressant effects of ketamine and botulinum toxin and the anxiolytic effect of psilocybin. J Psychiatry Neurosci 2013; 38:78–83Crossref, Medline, Google Scholar
119 : Do the dissociative side effects of ketamine mediate its antidepressant effects? J Affect Disord 2014; 159:56–61Crossref, Medline, Google Scholar
120 : Riluzole for relapse prevention following intravenous ketamine in treatment-resistant depression: a pilot randomized, placebo-controlled continuation trial. Int J Neuropsychopharmacol 2010; 13:71–82Crossref, Medline, Google Scholar
121 : On the safety and benefits of repeated intravenous injections of ketamine for depression. Biol Psychiatry 2012; 72:e11–e12Crossref, Medline, Google Scholar
122 : Ketamine: promising path or false prophecy in the development of novel therapeutics for mood disorders? Neuropsychopharmacology 2015; 40:259–267Crossref, Medline, Google Scholar
123 : Discriminative stimulus effects of hallucinogenic drugs: a possible relation to reinforcing and aversive effects. J Pharmacol Sci 2012; 120:70–76Crossref, Medline, Google Scholar
124 : Evaluation of sigma (σ) receptors in the antidepressant-like effects of ketamine in vitro and in vivo. Eur Neuropsychopharmacol 2012; 22:308–317Crossref, Medline, Google Scholar
125 : Restoring mood balance in depression: ketamine reverses deficit in dopamine-dependent synaptic plasticity. Biol Psychiatry 2014; 76:927–936Crossref, Medline, Google Scholar
126 : Chronic ketamine administration modulates midbrain dopamine system in mice. PLoS One 2012; 7:e43947Crossref, Medline, Google Scholar
127 : Dopamine D2/D3 but not dopamine D1 receptors are involved in the rapid antidepressant-like effects of ketamine in the forced swim test. Behav Brain Res 2015; 279:100–105Crossref, Medline, Google Scholar
128 : Chronic fluoxetine treatment induces structural plasticity and selective changes in glutamate receptor subunits in the rat cerebral cortex. Neuroscience 2010; 169:98–108Crossref, Medline, Google Scholar
129 : Ketamine-enhanced immobility in forced swim test: a possible animal model for the negative symptoms of schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry 2012; 38:310–316Crossref, Medline, Google Scholar
130 : Ketamine elicits sustained antidepressant-like activity via a serotonin-dependent mechanism. Psychopharmacology (Berl) 2013; 228:157–166Crossref, Medline, Google Scholar
131 : Raphe AMPA receptors and nicotinic acetylcholine receptors mediate ketamine-induced serotonin release in the rat prefrontal cortex. Int J Neuropsychopharmacol 2014; 17:1321–1326Crossref, Medline, Google Scholar
132 : Subanesthetic doses of ketamine transiently decrease serotonin transporter activity: a PET study in conscious monkeys. Neuropsychopharmacology 2013; 38:2666–2674Crossref, Medline, Google Scholar
133 : Comparative patch-clamp studies with freshly dissociated rat hippocampal and striatal neurons on the NMDA receptor antagonistic effects of amantadine and memantine. Eur J Neurosci 1996; 8:446–454Crossref, Medline, Google Scholar
134 : Differences in degree of trapping of low-affinity uncompetitive N-methyl-D-aspartic acid receptor antagonists with similar kinetics of block. J Pharmacol Exp Ther 1999; 288:204–210Medline, Google Scholar
135 : Targeting the glutamatergic system to treat major depressive disorder: rationale and progress to date. Drugs 2012; 72:1313–1333Crossref, Medline, Google Scholar
136 : Ketamine as a novel antidepressant: from synapse to behavior. Clin Pharmacol Ther 2012; 91:303–309Crossref, Medline, Google Scholar
137 : A review of ketamine in affective disorders: current evidence of clinical efficacy, limitations of use and pre-clinical evidence on proposed mechanisms of action. J Affect Disord 2014; 156:24–35Crossref, Medline, Google Scholar
138 : Brain-derived neurotrophic factor Val66Met polymorphism and antidepressant efficacy of ketamine in depressed patients. Biol Psychiatry 2012; 72:e27–e28Crossref, Medline, Google Scholar
139 : Brain-derived neurotrophic factor Val66Met allele impairs basal and ketamine-stimulated synaptogenesis in prefrontal cortex. Biol Psychiatry 2012; 71:996–1005Crossref, Medline, Google Scholar
140 : mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 2010; 329:959–964Crossref, Medline, Google Scholar
141 : Glutamate N-methyl-D-aspartate receptor antagonists rapidly reverse behavioral and synaptic deficits caused by chronic stress exposure. Biol Psychiatry 2011; 69:754–761Crossref, Medline, Google Scholar
142 : Cellular mechanisms underlying the antidepressant effects of ketamine: role of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol Psychiatry 2008; 63:349–352Crossref, Medline, Google Scholar
143 : Requirement of AMPA receptor stimulation for the sustained antidepressant activity of ketamine and LY341495 during the forced swim test in rats. Behav Brain Res 2014; 271:111–115Crossref, Medline, Google Scholar
144 : Antidepressant effects of AMPA and ketamine combination: role of hippocampal BDNF, synapsin, and mTOR. Psychopharmacology (Berl) 2013; 230:291–298Crossref, Medline, Google Scholar
145 : Activation of glutamatergic neurotransmission by ketamine: a novel step in the pathway from NMDA receptor blockade to dopaminergic and cognitive disruptions associated with the prefrontal cortex. J Neurosci 1997; 17:2921–2927Crossref, Medline, Google Scholar
146 : Reversal of phencyclidine effects by a group II metabotropic glutamate receptor agonist in rats. Science 1998; 281:1349–1352Crossref, Medline, Google Scholar
147 : Effect of clozapine, haloperidol, or M100907 on phencyclidine-activated glutamate efflux in the prefrontal cortex. Biol Psychiatry 2001; 50:750–757Crossref, Medline, Google Scholar
148 : Effect of phencyclidine on endogenous excitatory amino acid release from the rat anterior cingulate cortex--an in vivo microdialysis study. J Neural Transm 1993; 94:235–240Crossref, Google Scholar
149 : Immobilization-induced glutamate efflux in medial prefrontal cortex: blockade by (+)-Mk-801, a selective NMDA receptor antagonist. Stress 2000; 3:195–199Crossref, Medline, Google Scholar
150 : Effect of the noncompetitive NMDA antagonists MK-801 and ketamine on the spastic Han-Wistar mutant: a rat model of excitotoxicity. Dev Neurosci 2001; 23:31–40Crossref, Medline, Google Scholar
151 : Dual effects of ketamine: neurotoxicity versus neuroprotection in anesthesia for the developing brain. J Neurosurg Anesthesiol 2014; 26:155–160Crossref, Medline, Google Scholar
152 : Repeated exposure to anesthetic ketamine can negatively impact neurodevelopment in infants: a prospective preliminary clinical study. J Child Neurol 2014; 29:1333–1338Crossref, Medline, Google Scholar
153 : Existence of glia mitigated ketamine-induced neurotoxicity in neuron-glia mixed cultures of neonatal rat cortex and the glia-mediated protective effect of 2-PMPA. Neurotoxicology 2014; 44:218–230Crossref, Medline, Google Scholar
154 : www.foxnews.com/health/2014/02/24/antidepressant-ketamine-may-be-more-effective-than-electroconvulsive-therapy/Google Scholar
155 : The impact of medication resistance and continuation pharmacotherapy on relapse following response to electroconvulsive therapy in major depression. J Clin Psychopharmacol 1990; 10:96–104Crossref, Medline, Google Scholar
156 : Continuation and maintenance electroconvulsive therapy for mood disorders: review of the literature. Neuropsychobiology 2011; 64:129–140Crossref, Medline, Google Scholar
157 : Neurotoxicity of ketamine: known unknowns. Crit Care Med 2012; 40:2518–2519Crossref, Medline, Google Scholar
158 : Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science 1999; 283:70–74Crossref, Medline, Google Scholar
159 : Ketamine-induced neuroapoptosis in the fetal and neonatal rhesus macaque brain. Anesthesiology 2012; 116:372–384Crossref, Medline, Google Scholar
160 : Ketamine-induced neuronal cell death in the perinatal rhesus monkey. Toxicol Sci 2007; 98:145–158Crossref, Medline, Google Scholar
161 : Ketamine exposure in adult mice leads to increased cell death in C3H, DBA2 and FVB inbred mouse strains. Drug Alcohol Depend 2008; 92:217–227Crossref, Medline, Google Scholar
162 : Ketamine causes mitochondrial dysfunction in human induced pluripotent stem cell-derived neurons. PLoS One 2015; 10:e0128445Crossref, Medline, Google Scholar
163 : Increased response to ketamine following treatment at long intervals: implications for intermittent use. Biol Psychiatry 2008; 63:178–183Crossref, Medline, Google Scholar
164 UNODC (United Nations Office on Drugs and Crime): World Drug Report 2011. United Nations PublicationGoogle Scholar
165 Askwith R: How aspirin turned hero. Sunday Times Sept 13, 1998Google Scholar
166 : Actions of D-cycloserine at the N-methyl-D-aspartate-associated glycine receptor site in vivo. Neuropharmacology 1991; 30:1167–1171Crossref, Medline, Google Scholar
167 : D-cycloserine, a partial agonist at the glycine site coupled to N-methyl-D-aspartate receptors, improves visual recognition memory in rhesus monkeys. J Pharmacol Exp Ther 1996; 278:891–897Medline, Google Scholar
168 : Dose-finding trial of D-cycloserine added to neuroleptics for negative symptoms in schizophrenia. Am J Psychiatry 1995; 152:1213–1215Link, Google Scholar
169 : D-cycloserine increases positive symptoms in chronic schizophrenic patients when administered in addition to antipsychotics: a double-blind, parallel, placebo-controlled study. Neuropsychopharmacology 1999; 21:203–210Crossref, Medline, Google Scholar
170 : Characterization of the interactive effects of glycine and D-cycloserine in men: further evidence for enhanced NMDA receptor function associated with human alcohol dependence. Neuropsychopharmacology 2011; 36:701–710Crossref, Medline, Google Scholar
171 : Examination of Org 26576, an AMPA receptor positive allosteric modulator, in patients diagnosed with major depressive disorder: an exploratory, randomized, double-blind, placebo-controlled trial. J Psychopharmacol 2012; 26:1525–1539Crossref, Medline, Google Scholar
172 : Maximum tolerated dose evaluation of the AMPA modulator Org 26576 in healthy volunteers and depressed patients: a summary and method analysis of bridging research in support of phase II dose selection. Drugs R D 2012; 12:127–139Crossref, Medline, Google Scholar
173 : Metabotropic glutamate receptor 5 negative modulation in phase I clinical trial: potential impact of circadian rhythm on the neuropsychiatric adverse reactions-do hallucinations matter? ISRN Psychiatry 2014; 2014:652750. Available at doi:
174 : Multiple mechanisms of ketamine blockade of N-methyl-D-aspartate receptors. Anesthesiology 1997; 86:903–917Crossref, Medline, Google Scholar
175 : The synergistic effect of selective sigma receptor agonists and uncompetitive NMDA receptor antagonists in the forced swim test in rats. J Physiol Pharmacol 2006; 57:217–229Medline, Google Scholar
176 : Indistinguishable synaptic pharmacodynamics of the NMDAR channel blockers memantine and ketamine. Mol Pharmacol 2013; 84:935–947Crossref, Medline, Google Scholar
177 : Mg2+ imparts NMDA receptor subtype selectivity to the Alzheimer’s drug memantine. J Neurosci 2009; 29:2774–2779Crossref, Medline, Google Scholar
178 : Expression of mRNA of neurotrophic factors and their receptors are significantly altered after subchronic ketamine treatment. Med Chem 2008; 4:256–263Crossref, Medline, Google Scholar
179 : Acute administration of ketamine in rats increases hippocampal BDNF and mTOR levels during forced swimming test. Ups J Med Sci 2013; 118:3–8Crossref, Medline, Google Scholar
180 : Memantine treatment reverses anhedonia, normalizes corticosterone levels and increases BDNF levels in the prefrontal cortex induced by chronic mild stress in rats. Metab Brain Dis 2012; 27:175–182Crossref, Medline, Google Scholar
181 : Ketamine alters behavior and decreases BDNF levels in the rat brain as a function of time after drug administration. Rev Bras Psiquiatr 2013; 35:262–266Crossref, Medline, Google Scholar
182 : Acute increases in plasma mammalian target of rapamycin, glycogen synthase kinase-3β, and eukaryotic elongation factor 2 phosphorylation after ketamine treatment in three depressed patients. Biol Psychiatry 2013; 73:e35–e36Crossref, Medline, Google Scholar
183 : A targeted multiplexed proteomic investigation identifies ketamine-induced changes in immune markers in rat serum and expression changes in protein kinases/phosphatases in rat brain. J Proteome Res 2015; 14:411–421 Crossref, Medline, Google Scholar



