Attention, Memory, and Motor Skills as Childhood Predictors of Schizophrenia-Related Psychoses: The New York High-Risk Project
Abstract
OBJECTIVE: Childhood neurobehavioral deficits in offspring of schizophrenic, affectively ill, and psychiatrically normal parents were evaluated as predictors of schizophrenia-related psychoses in adulthood. METHOD: The offspring were tested with neurobehavioral measures at 7–12 years of age and assessed in mid-adulthood for axis I diagnoses. The relationships of childhood deficits in attention, verbal memory, and gross motor skills to adulthood schizophrenia-related psychoses were examined in separate path analyses by using logistic regression equations. Sensitivity and specificity were determined for each of the childhood dysfunctions. RESULTS: For the offspring of schizophrenic parents, childhood deficits in verbal memory, gross motor skills, and attention identified 83%, 75%, and 58%, respectively, of the subjects with schizophrenia-related psychoses; 50% were identified by all three variables combined. False positive rates in subjects who did not develop schizophrenia-related psychoses ranged from 18% for those with deficits in attention during childhood to 28% for those with deficits in memory. The three variables had low deficit rates in the offspring of the other two parental groups and were not associated with other psychiatric disorders in any group. CONCLUSIONS: Schizophrenia-related psychoses in adulthood are distinguished in subjects at risk for schizophrenia by childhood deficits in verbal memory, gross motor skills, and attention. The findings suggest that deficits in these variables are relatively specific to schizophrenia risk and may be indicators of the genetic liability to schizophrenia.
Most contemporary high-risk and family studies of schizophrenia rest on cumulative evidence from earlier research that has demonstrated important genetic contributions to this illness (1). The more recent studies search for phenotypic anomalies with higher prevalence in relatives of schizophrenic probands and in other at-risk individuals under the hypothesis that such anomalies represent indicators of susceptibility genes that make up the genetic liability to schizophrenia. Identification of these dysfunctions, it is hypothesized, may provide keys to understanding the pathophysiology of the illness and to parsing clinical heterogeneity (2–4).
Nevertheless, the two types of studies have different emphases. Family studies usually center on adult nonpsychotic relatives and delineation of the schizophrenia genotype’s range of nonclinical expression (5, 6). Phenotypic indicators identified in these studies may be useful as tools in differentiating clinically unaffected family members into probable carriers and noncarriers of schizophrenia susceptibility genes in gene search studies (6, 7).
While high-risk research shares the goal of identifying phenotypic indicators, it is chiefly a longitudinal quest for variables that successfully predict future cases of schizophrenia or its spectrum disorders. Reliable predictor variables are seen as valuable aids for development of early intervention strategies, since they would both highlight problems that require remediation and flag the individuals most in need of receiving such intervention (8).
Numerous deficits in neurobehavioral domains have been noted as being promising schizophrenia liability indicators in high-risk and family studies (9–14). A comparative evaluation of several such variables (6), however, suggested that not all of them are equally useful as potential liability indicators for genetic search studies.
Some of the same neurobehavioral deficits have also been suggested in high-risk research to be predictors of future cases of schizophrenia or its related disorders (15–17), as have other variables that probably reflect environmental more than genetic components of etiology (18, 19). Reported associations between these variables and later schizophrenia-related disorders are often relatively modest, however, and the predictive accuracy of the different variables has not been compared. Furthermore, specificity with respect to risk for schizophrenia, compared with risk for other psychiatric disorders, has not been established for most dysfunctions proposed either as schizophrenia liability indicators or as predictors of future schizophrenia-related disorders.
This report concerns predictive relationships between neurobehavioral variables examined in mid-childhood and later schizophrenia-related psychoses in offspring of schizophrenic, affectively ill, and normal parents in the New York High-Risk Project (20, 21). Accuracy (i.e., sensitivity and specificity) in correctly discriminating subjects who developed schizophrenia-related psychoses from those who did not was evaluated for three neurobehavioral variables. One was a global measure of attention deviance, which we have previously reported to be associated with poor adjustment and schizophrenia spectrum features in adolescence (17, 22). The other two measures were verbal memory and gross motor skills.
Prediction parameters were also compared between the groups with schizophrenic and affectively ill parents to assess unique risk for offspring of parents with schizophrenia and between schizophrenia-related psychoses and other axis I outcomes to assess uniqueness to schizophrenia-related psychoses.
Method
Subject Selection and Diagnostic Procedures
The New York High-Risk Project recruited offspring of schizophrenic, affectively ill, and psychiatrically normal parents in 1971–1972 and in 1977–1979. Mentally ill parents were identified from admissions to state psychiatric hospitals in lower New York State, interviewed with the Schedule for Affective Disorders and Schizophrenia—Lifetime Version (SADS-L) (23), and diagnosed according to Research Diagnostic Criteria (RDC) (24). Normal comparison parents were free of major mental disorders and prior psychiatric treatment history (20).
The offspring were white, English-speaking, a mean age of 9.29 years (SD=1.76), and free of mental retardation, major psychiatric disorders, or treatment for emotional problems at recruitment. Subjects received six rounds of evaluations approximately 3 years apart.
Written informed consent was obtained from the parents for themselves and their minor children at the first contact with the family and from each individual participant at all subsequent examinations.
SADS-L interviews were administered during the fourth through sixth evaluations, at which the subjects were a mean age of 19.70 years (SD=1.83), 23.20 (SD=2.12), and 27.30 (SD=2.16), respectively. Lifetime axis I diagnoses and estimates of onset age were based on the interviews and clinical records. For subjects with new onsets since the sixth evaluation, assessments were updated to autumn 1995, at which point the subjects were a mean age of 30.70 years (SD=3.26). Details concerning interview procedures and reliability have been previously described (20, 21).
For this report, adulthood axis I disorders were hierarchically classified into three categories: 1) schizophrenia-related psychoses (schizophrenia, unspecified psychosis, and “mainly schizophrenic” schizoaffective disorder [per the RDC distinction]), 2) major affective disorders (“mainly affective” schizoaffective disorder [per the RDC distinction], bipolar I and bipolar II disorder, and major depression); and 3) other major axis I disorders (hypomania, intermittent depression, anxiety disorders, and substance abuse disorders). Subjects were counted only once, regardless of comorbidity, in the hierarchal order.
Only those offspring with complete childhood neurobehavioral data and diagnostic assessments in adulthood were included in this report. Of the original 358, 34 had died or could not be traced to adulthood. Of the 324 followed subjects, 55 had incomplete neurobehavioral data because of equipment failure or administration of different tests. Thus, this report concerns 269 subjects. Adulthood axis I diagnoses among the 79 offspring of schizophrenic parents included schizophrenia-related psychoses (15.2%, N=12), major affective disorders (35.4%, N=28), and other major axis I disorders (16.5%, N=13); 32.9% (N=26) had no disorder. Among the 57 offspring of affectively ill parents, 7.0% (N=4) had schizophrenia-related psychoses, 45.6% (N=26) had major affective disorders, and 21.1% (N=12) had other major axis I disorders in adulthood; 26.3% (N=15) had no disorder. Finally, schizophrenia-related psychoses, major affective disorders, and other major axis I disorders affected 0.8% (N=1), 30.1% (N=40), and 27.8% (N=37) of the 133 offspring of normal parents, respectively; 41.4% (N=55) had no disorder.
Childhood Neurobehavioral Performance
The cognitive batteries have been described in detail (22). Both groups (i.e., those recruited in 1971–1972 and in 1977–1979) were given variants of the Continuous Performance Test, a test of visual sustained attention (25) widely used in schizophrenia research.
Other cognitive tasks employed were the Attention Span Task, a measure of short-term memory storage capacity based on procedures reported in studies of schizophrenic patients (26) and the digits forward and backward components of the Digit Span subtest from the Wechsler Intelligence Scale for Children (WISC or WISC-R). (For offspring recruited in 1977–1979, the Visual Aural Digit Span Test [27] was substituted for the Attention Span Task and Digit Span tasks to test immediate auditory and visual recall.)
Neuromotor assessment was based on a 31-item version of the Lincoln-Oseretsky Motor Development Scale (28).
Two types of summary measures were developed: the Attention Deviance Index, a measure of global attention as described previously (17, 22), and composite variables selected by using hypotheses that were based on the structure of the tests and entered into factor analyses with varimax rotation (29). The factor analyses were carried out separately for the two recruitment groups, and the factor structures were identical. The resulting five factors were errors and correct responses (both from Continuous Performance Test variables), memory (from the Digit Span, Attention Span Task, and Visual Aural Digit Span Test), and fine motor and gross motor skills (from the Lincoln-Oseretsky Motor Development Scale).
Statistical Analyses
We developed theoretical models to be tested separately for each of six mediating variables (i.e., performance on the Attention Deviance Index and the aforementioned five factors). The model for each mediating variable generated two equations for which explanatory variables (parental psychiatric status) and control variables (gender, recruitment sample [1971–1972 versus 1977–1979], and age at testing) were included as background variables. The first equation, a linear regression, related parental psychiatric status and control variables to childhood neurobehavioral performance on the mediating variable. The second equation, a logistic regression, related parental psychiatric status, control variables, and performance on the mediating variable to the psychiatric outcome. Although schizophrenia-related psychoses was the main psychiatric outcome of interest, separate analyses were also run for major affective disorders and for other major axis I disorders.
In the theoretical model (Figure 1), two paths of main interest were 1) the effect of being an offspring of a schizophrenic parent versus being an offspring of a psychiatrically normal parent in relation to childhood neurobehavioral performance on the mediating variable (path A) and 2) the relationship of performance on the mediating variable to psychiatric outcome (path B). Paths A and B were assessed in the linear and logistic regressions, respectively. Another critical pathway assessed in the logistic regression analysis was the direct effect of having a schizophrenic parent versus a normal parent in relation to the outcome (path C). Paths D and E (analogous to paths A and C, respectively, but with the effect of being an offspring of an affectively ill parent replacing that of being an offspring of a schizophrenic parent) were also examined through linear and logistic regression equations.
The strongest genetic model would be one showing that 1) being an offspring of a schizophrenic parent significantly affects performance on the mediating variable (path A)—thereby suggesting that this variable could be a phenotypic indicator of the genetic liability to schizophrenia—and 2) performance on the mediating variable and offspring-of-schizophrenic-parents status significantly predict future development of schizophrenia-related psychoses (paths B and C, respectively). Such a model would be consistent with the fact that some, but not all, of the phenotypic indicators have been identified and, therefore, that familial background (path C) remains as a significant direct effect on the development of schizophrenia-related psychoses. Significant findings for paths A and B only present a weaker case for the genetic model, since the schizophrenic-parent effect has no explanatory power for the development of schizophrenia-related psychoses. A model with significant paths A and C only would suggest that, although the mediating variable might be a liability indicator, it is not useful as a predictor of a schizophrenia-related psychosis outcome. A model with significant paths B and C only might suggest that, whereas both performance on the mediating variable and the schizophrenic-parent effect predict the outcome, the mediating variable is not an indicator of genetic liability. This model might be found for environmental factors that interact with a genetic liability in the development of schizophrenia.
Our goal was to compare the sensitivity and specificity of models in which regression coefficients for both paths A and B were significant as predictors of the onset of schizophrenia-related psychoses (i.e., the models in which the mediating variable represents a potential endophenotype as well as predictor of later illness.) On the basis of predictive probabilities yielded by the logistic regression analyses, receiver operating characteristic curves were plotted. From these curves we derived cutting scores to maximize the correct classification rates for schizophrenia-related psychoses, i.e., scores that maximized sensitivity and specificity of the model in predicting schizophrenia-related psychoses for offspring of schizophrenic parents. Sensitivity represents the percentage of subjects with schizophrenia-related psychoses correctly classified by the cutting score, and specificity represents the percentage of subjects without schizophrenia-related psychoses who were correctly classified as not developing schizophrenia-related psychoses. Note that since 1–specificity equals the false positive rate (i.e., the proportion of subjects falsely predicted to develop schizophrenia-related psychoses), maximizing specificity ensures that the false positive rate is minimized.
Results
Mean performance on each of the six mediating variables was significantly worse in the offspring of schizophrenic parents than in the other groups. However, the models for errors, correct responses, and fine motor skills were not significantly related to onset of schizophrenia-related psychoses. These variables were therefore dropped. (No mediating variable was significantly related to onset of major affective disorders or other major axis I disorders.)
The contrast between offspring of schizophrenic parents and offspring of normal parents (path A) was significant in the models involving the three remaining neurobehavioral variables (Attention Deviance Index: t=4.87, df=273, p<0.001; memory: t=–2.02, df=264, p=0.05; gross motor skills: t=–4.22, df=310, p<0.001). For all three models, a significant direct relationship with schizophrenia-related psychoses was seen for both performance on the mediating variable (path B) and the contrast between offspring of schizophrenic parents and offspring of normal parents (path C): (Attention Deviance Index model, path B: Wald χ2=3.70, df=1, p=0.054; path C: Wald χ2=7.83, df=1, p=0.005; odds ratio=20:1. Memory model, path B: Wald χ2=3.66, df=1, p=0.056; path C: Wald χ2=9.05, df=1, p=0.003, odds ratio=25:1. Gross motor skills model, path B: Wald χ2=7.48, df=1, p=0.006; path C: Wald χ2=7.97, df=1, p=0.005, odds ratio=20:1). The combination of significant regression coefficients for paths A, B, and C is consistent with the genetic model and suggests that the mediating neurobehavioral variables may be phenotypic indicators of the genetic liability to schizophrenia-related psychoses and that the variables together with offspring-of-schizophrenic-parents status predict the future development of these disorders.
The contrast between offspring of affectively ill parents and offspring of normal parents (path D) was significant only for gross motor skills (t=–1.98, df=310, p=0.05), but path E—the direct effect of an affectively ill parent on development of schizophrenia-related psychoses—was not significant, which indicated a weak genetic model.
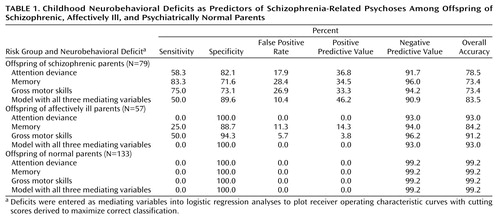
Table 1 presents sensitivity, specificity, and false positive rates for prediction of the development of schizophrenia-related psychoses, derived from the aforementioned receiver operating characteristic curves. Positive and negative predictive values (i.e., the percentages of subjects with impairment on the mediating variable who developed schizophrenia-related psychoses and of subjects without impairment who did not develop such disorders, respectively) and overall accuracy rates (true positive plus true negative subjects) are also presented. For offspring of schizophrenic parents, sensitivity in identifying future development of schizophrenia-related psychoses was highest for the regression model with memory (83.3%) as the mediating variable and lowest for the model with Attention Deviance Index performance (58.3%). Conversely, false positive rates in the group of offspring of schizophrenic parents were lowest for the Attention Deviance Index model and not as good for the models with memory and gross motor skills as mediating variables. Positive predictive values and overall accuracy rates were nearly the same for offspring of schizophrenic parents with all three mediating variables.
Low sensitivity and false positive rates for prediction of schizophrenia-related psychoses in the offspring of affectively ill and psychiatrically normal parents suggest that deficits in each mediating variable representing neurobehavioral performance are comparatively unique to risk for schizophrenia. For example, the total prevalence of impairment in the offspring of schizophrenic parents compared with offspring of affectively ill parents was 24% versus 0%, respectively, for attention, 37% versus 12% for memory, and 34% versus 9% for motor skills.
We also examined agreement across the three prediction models. In the group of offspring of schizophrenic parents, 91.7% (N=11 of 12) of those with schizophrenia-related psychoses were correctly classified by at least one model. As Table 1 shows, all three models together identified half of the subjects with schizophrenia-related psychoses (50% sensitivity, 46% positive predictive value) but also tagged 10% of the subjects without schizophrenia-related psychoses as false positive. Among the offspring of affectively ill parents, no subjects were predicted to develop schizophrenia-related psychoses by all three models, and only one was predicted by two models.
Discussion
Deficits in attention, memory, and neuromotor functions were noted in early formal descriptions of schizophrenia (30) and subsequently documented in experimental studies of patients and their relatives (10, 11, 15). This report suggests that such dysfunctions clearly meet at least three of the criteria that have been established to qualify a variable as a phenotypic indicator (2–4). In this study, the deficits were 1) present before clinical symptoms and thus independent of the illness state; 2) substantially more prevalent in relatives of schizophrenic patients than in relatives of comparison subjects; and 3) comparatively specific to risk for schizophrenia versus risk for affective disorders, in that they exhibited greater prevalence among offspring of schizophrenic parents than among offspring of affectively ill parents. The neurobehavioral variables were also specifically related to schizophrenia-related psychoses as the psychiatric outcome rather than to other outcomes.
A fourth criterion—longitudinal stability and persistence of impairment—was seen for two of the variables as verbal memory was assessed and age-appropriate versions of the Attention Deviance Index (31) were administered over several evaluations. Gross motor skill was tested only in childhood, but other studies (32) suggest that neuromotor deficits continue to older ages.
The chief focus here, however, was on the effectiveness of the three measures. Each showed comparatively good accuracy of classification, with rates at the upper end of the range previously reported in high-risk research, but the sensitivity-specificity balance differed among the variables. Sensitivity, in correctly predicting schizophrenia-related psychoses, was unusually high for verbal memory (83%) and gross motor skills (75%). Comparatively, the Attention Deviance Index had a weaker sensitivity rate but fewer false positive classifications among offspring of schizophrenic parents who did not develop schizophrenia-related psychoses. Thus, implications for use in genetic or intervention research differ for these variables.
Because gene search studies are especially vulnerable to false positive classification of relatives as susceptibility gene carriers (6), the Attention Deviance Index may be the most useful of the three variables for such studies. Indeed, a “diagnostic accuracy” analysis (6) that compared a number of personality disorders and neurobehavioral variables (not including memory or gross motor skills) that had been proposed as indicators of the schizophrenia genotype suggested that the Attention Deviance Index might prove the most useful for increasing informativeness of linkage data. Furthermore, in the present analyses, impairment rates were greater for memory and gross motor skills than for the Attention Deviance Index in offspring of affectively ill parents, thus suggesting better discrimination by the Attention Deviance Index between schizophrenia pedigree members who are at risk for affective disorders and those who are carriers of the schizophrenia genotype. This is important because, given the high population base rate for affective disorders, individuals at risk for these disorders are often included by chance in schizophrenia pedigrees.
Intervention, the long-term but often controversial goal of high-risk research, poses other questions with respect to the level of tolerance for predictive misclassifications. Strategies, such as cognitive intervention therapies, that have little danger of deleterious effects can obviously tolerate higher false positive and lower sensitivity rates (or positive predictive values) than strategies, such as pharmacological interventions, that are potentially more powerful but could produce harmful effects, especially in children or adolescents. On the whole, predictive performance of our three measures, or of most measures reported in high-risk studies, does not seem to support use of the latter types of intervention in young high-risk subjects.
A limitation of the present analyses is that they cannot be directly extrapolated to detection of preschizophrenic individuals in the general population. Since only about 15% of future schizophrenic patients have a schizophrenic first-degree relative, identification of the majority of preschizophrenic individuals for any type of intervention program would need to be based on general population screening. Given the low population base rate of schizophrenia-related psychoses, however, normal control groups in this and other high-risk studies are too small to test an indicator’s capacity as an effective preschizophrenia screen and predictor of schizophrenia-related psychoses in the general population. Thus, this study provides no information about choosing individuals from the general population, even for “benign” interventions.
The most conservative approach to detection either of probable genotypes in schizophrenia pedigrees or of individuals considered for intervention in high-risk research would be one requiring evidence of impairment on multiple “endophenotypic” (1) measures. In the present analyses, the combination of impairment on all three variables together achieved better classification among the offspring of schizophrenic parents with respect to false positive rate (10%), positive predictive value (46%), and overall accuracy (83%) than any of the variables individually.
The nonpsychotic offspring of schizophrenic parents who were among the 10% falsely classified when all three variables were combined are of interest because they appear to be carriers of some of the susceptibility genes for schizophrenia and may yield information, by default, about other factors that are needed for development of the overt illness. It is possible, of course, that some of these subjects will express the clinical illness in the future. However, they were not younger than the mean age at onset of the subjects with schizophrenia-related psychoses and had not met criteria for probable cluster A disorders, which most subjects with schizophrenia-related psychoses exhibited before onset of psychoses.
A likely possibility—expected from the multifactorial nature of schizophrenia (1)—is that deficits in attention, memory, and gross motor skills reflect only some of the phenotypic indicators of a large complex of susceptibility genes, as indicated by the significant effect of path C from family background to schizophrenia-related psychoses when the influence of the mediating variables is controlled. Thus, subjects with schizophrenia-related psychoses may exhibit additional as-yet-unidentified phenotypic impairments than the subjects who were false positive. Although several neurobehavioral variables examined during the childhood testing did not relate to schizophrenia-related psychoses, other measures, not yet available at that time (e.g., functional brain imaging, more complete and refined neuropsychological test batteries, etc.) might have been better predictors. Alternatively, false positive subjects may have fewer negative “polygenic potentiators” (33) or may have experienced less exposure to environmental factors that interact with susceptibility genes for full clinical expression of the illness. Ongoing analyses in the New York High-Risk Project are exploring these possibilities by comparing a number of phenotypic and historical variables among offspring of schizophrenic parents 1) with schizophrenia-related psychoses, 2) without schizophrenia-related psychoses and correctly classified as such (true negative), and 3) without schizophrenia-related psychoses but falsely classified as such.
Childhood manifestation of neurobehavioral deficits by preschizophrenic subjects is consistent with current neurodevelopmental hypotheses (34, 35), which suggest that early brain damage—resulting from environmental insults, faulty genetic programming, or their interaction—may remain unexpressed as clinical symptoms for many years but may appear as earlier neurobehavioral or other deficits. Cognitive deficits, notably including memory and attentional impairments, with possible roots in early developmental damage are considered hallmark characteristics of schizophrenia. Nevertheless, as demonstrated here, motoric dysfunctions must also be considered in explanations of the pathophysiology of the illness.
It has been proposed that dysfunctional frontal-cerebellar-thalamic circuitry, which appears to underlie memory impairment in schizophrenic patients, could also account for a broad range of other cognitive and motor disturbances (36, 37). The hypothesized disruption in a central timing process associated with this circuitry is of interest in connection with the present cognitive and motor findings, especially when taken together with the fact that offspring of schizophrenic parents in this study displayed clear temporal deficits on a perceptual duration task (unpublished 1999 manuscript of T. Penney et al.).
On the whole, the present analyses indicate the importance of prospective follow-up of offspring of schizophrenic and other mentally ill parents for identifying early neurobehavioral dysfunctions that are probably endophenotypic indicators of schizophrenia susceptibility genes, strong predictors of schizophrenia-related psychoses in the offspring, and comparatively specific to the liability for schizophrenia contrasted with other psychiatric disorders.
 |
Received Nov. 2, 1999; revisions received March 2 and March 27, 2000; accepted April 3, 2000. From the Department of Medical Genetics, New York State Psychiatric Institute. Address reprint requests to Dr. Erlenmeyer-Kimling, Department of Medical Genetics, New York State Psychiatric Institute, 1051 Riverside Dr., Unit 6, New York, NY 10032; [email protected] (e-mail). Supported in part by NIMH grant MH-19560 to Dr. Erlenmeyer-Kimling and by the Office of Mental Health of the State of New York. The authors thank the testers, the current and previous social work staff for maintaining continuing contact with the research participants, the interviewers who conducted adulthood diagnostic interviews, Dr. Jean Endicott and members of the Department of Research Assessment and Training at the New York State Psychiatric Institute for diagnostic reviews, and the many members of the group who have taken part in other aspects of this study. The authors would also like to thank the research participants and their families.
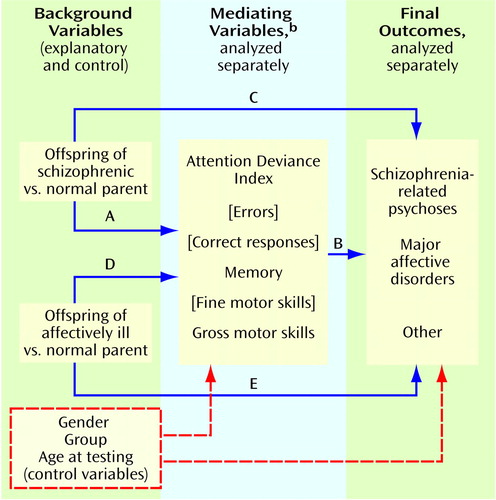
Figure 1. Models to Determine the Relationship of Parental Psychiatric Status, Offspring Neurobehavioral Deficits in Childhood, and Offspring Psychiatric Outcome in Adulthooda
aPaths A and D represent the effect of the explanatory variable (parental psychiatric status) on mediating variables (childhood neurobehavioral performance)Paths C and E represent the direct effect of parental psychiatric status on final outcome. Path B represents the relationship of neurobehavioral performance to final outcome.
bMediating variables shown in brackets were not significant and were dropped from the analyses.
1. Gottesman II, McGuffin P, Farmer AE: Clinical genetics as clues to the “real” genetics of schizophrenia (a decade of modest gains while playing for time). Schizophr Bull 1987; 13:23–47Crossref, Medline, Google Scholar
2. Erlenmeyer-Kimling L: Biological markers for the liability to schizophrenia, in Biological Perspectives of Schizophrenia, the Dahlem Konferenzen. Edited by Helmchen H, Henn FA. Chichester, UK, John Wiley & Sons, 1987, pp 33–56Google Scholar
3. Holzman PS: Behavioral markers of schizophrenia useful for genetic studies. J Psychiatr Res 1992; 26:427–445Crossref, Medline, Google Scholar
4. Moldin SO, Erlenmeyer-Kimling L: Measuring liability to schizophrenia: progress report 1994: editors’ preface. Schizophr Bull 1994; 20:25–30Crossref, Medline, Google Scholar
5. Erlenmeyer-Kimling L: A look at the evolution of developmental models of schizophrenia, in Psychopathology: The Evolving Science of Mental Disorder. Edited by Matthysse S, Levy D, Kagan J, Benes FM. Cambridge, UK, Cambridge University Press, 1996, pp 229–252Google Scholar
6. Faraone SV, Kremen WS, Lyons MJ, Pepple JR, Seidman LJ, Tsuang MT: Diagnostic accuracy and linkage analysis: how useful are schizophrenia spectrum phenotypes? Am J Psychiatry 1995; 152:1286–1290Google Scholar
7. Risch N: Genetic linkage and complex diseases, with special reference to psychiatric disorders. Genet Epidemiol 1990; 7:3–16Crossref, Medline, Google Scholar
8. Green MF: Interventions for neurocognitive deficits: editor’s introduction. Schizophr Bull 1999; 25:197–200Crossref, Medline, Google Scholar
9. Levy DL, Holzman PS, Matthysse S, Mendell NR: Eyetracking and schizophrenia: a selective review. Schizophr Bull 1994; 20:47–62Crossref, Medline, Google Scholar
10. Cornblatt BA, Keilp JG: Impaired attention, genetics, and the pathophysiology of schizophrenia. Schizophr Bull 1994; 20:31–46Crossref, Medline, Google Scholar
11. Kremen WS, Seidman LJ, Pepple JR, Lyons MJ, Tsuang MT, Faraone SV: Neuropsychological risk indicators for schizophrenia: a review of family studies. Schizophr Bull 1994; 20:103–118Crossref, Medline, Google Scholar
12. Faraone SV, Seidman LJ, Kremen WS, Pepple JR, Lyons MJ, Tsuang MT: Neuropsychological functioning among the nonpsychotic relatives of schizophrenic patients: a diagnostic efficiency analysis. J Abnorm Psychol 1995; 104:286–304Crossref, Medline, Google Scholar
13. Park S, Holzman PS, Goldman-Rakic PS: Spatial working memory deficits in the relatives of schizophrenic patients. Arch Gen Psychiatry 1995; 52:821–828Crossref, Medline, Google Scholar
14. Nuechterlein K: Annotation: childhood precursors of adult schizophrenia. J Child Psychol Psychiatry 1986; 27:133–144Crossref, Medline, Google Scholar
15. Hans SL, Marcus J, Nuechterlein KH, Asarnow RF, Styr B, Auerbach JG: Neurobehavioral deficits at adolescence in children at risk for schizophrenia: the Jerusalem Infant Development Study. Arch Gen Psychiatry 1999; 56:741–748Crossref, Medline, Google Scholar
16. Walker EF, Savoie T, Davis D: Neuromotor precursors of schizophrenia. Schizophr Bull 1994; 20:441–451Crossref, Medline, Google Scholar
17. Cornblatt BA, Lenzenweger MF, Dworkin RH, Erlenmeyer-Kimling L: Childhood attentional dysfunctions predict social deficits in unaffected adults at risk for schizophrenia. Br J Psychiatry 1992; 161:59–64Crossref, Medline, Google Scholar
18. Jones PB, Rantakallio P, Hartikainen A-L, Isohanni M, Sipila P: Schizophrenia as a long-term outcome of pregnancy, delivery, and perinatal complications: a 28-year follow-up of the 1966 North Finland general population birth cohort. Am J Psychiatry 1998; 155:355–364Link, Google Scholar
19. McNeil TF: Perinatal risk factors and schizophrenia: selective review and methodological concerns. Epidemiol Rev 1995; 17:107–112Crossref, Medline, Google Scholar
20. Erlenmeyer-Kimling L, Squires-Wheeler E, Adamo UH, Bassett AS, Cornblatt BA, Kestenbaum CJ, Rock D, Roberts SA, Gottesman II: The New York High-Risk Project: psychoses and cluster A personality disorders in offspring of schizophrenic parents at 23 years of follow-up. Arch Gen Psychiatry 1995; 52:857–865Crossref, Medline, Google Scholar
21. Erlenmeyer-Kimling L, Adamo UH, Rock D, Roberts SA, Bassett AS, Squires-Wheeler E, Cornblatt BA, Endicott J, Pape S, Gottesman II: The New York High-Risk Project: prevalence and comorbidity of axis I disorders in offspring of schizophrenic parents at 25-year follow-up. Arch Gen Psychiatry 1997; 54:1096–1102Google Scholar
22. Erlenmeyer-Kimling L, Cornblatt B: A summary of attentional findings in the New York High-Risk Project. J Psychiatr Res 1992; 26:405–426Crossref, Medline, Google Scholar
23. Spitzer RL, Endicott J: Schedule for Affective Disorders and Schizophrenia (SADS), 3rd ed. New York, New York State Psychiatric Institute, Biometrics Research, 1978Google Scholar
24. Spitzer RL, Endicott J, Robins E: Research Diagnostic Criteria (RDC) for a Selected Group of Functional Disorders, 3rd ed. New York, New York State Psychiatric Institute, Biometrics Research, 1978Google Scholar
25. Rosvold HE, Mirsky AF, Sarason I, Bransome ED Jr, Beck LH: A continuous performance test of brain damage. J Consult Psychol 1956; 20:343–350Crossref, Medline, Google Scholar
26. Hawks DV, Robinson KN: Information processing in schizophrenia: the effect of varying rate of presentation and introducing interference. Br J Soc Clin Psychol 1971; 10:30–41Crossref, Medline, Google Scholar
27. Koppitz EM: The Visual Aural Digit Span Test. New York, Grune & Stratton, 1977Google Scholar
28. Sloan W: The Lincoln-Oseretsky Motor Development Scale. Gen Psychol Monogr 1955; 51:183–252Medline, Google Scholar
29. Kaiser HF: The varimax criteria for analytic rotation in factor analysis. Psychometrika 1958; 23:187–200Crossref, Google Scholar
30. Kraepelin E: Dementia Praecox and Paraphrenia. Translated by Barclay RM; edited by Robertson GM. Edinburgh, E & S Livingstone, 1919Google Scholar
31. Cornblatt B, Winters L, Erlenmeyer-Kimling L: Attentional markers of schizophrenia: evidence from the New York High Risk Study, in Schizophrenia: Scientific Progress. Edited by Schulz SC, Tamminga CA. New York, Oxford University Press, 1989, pp 83–92Google Scholar
32. Marcus J, Hans SL, Auerbach JG, Auerbach AG: Children at risk for schizophrenia: the Jerusalem Infant Development Study, II: neurobehavioral deficits at school age. Arch Gen Psychiatry 1993; 50:797–809Crossref, Medline, Google Scholar
33. Meehl PE: Specific genetic etiology, psychodynamics, and therapeutic nihilism. Int J Ment Health 1972; 1:10–27Google Scholar
34. Murray RM, Lewis SW: Is schizophrenia a neurodevelopmental disorder? (letter). Br Med J (Clin Res Ed) 1988; 296:63Crossref, Medline, Google Scholar
35. Weinberger DR: The pathogenesis of schizophrenia: a neurodevelopmental theory, in The Neurobiology of Schizophrenia. Edited by Nasrallah HA, Weinberger DR. Amsterdam, Elsevier, 1986, pp 397–406Google Scholar
36. Crespo-Facorro B, Paradiso S, Andreasen NC, O’Leary DS, Watkins GL, Boles Ponto LL, Hichwa RD: Recalling word lists reveals “cognitive dysmetria” in schizophrenia: a positron emission tomography study. Am J Psychiatry 1999; 156:386–392Abstract, Google Scholar
37. Andreasen NC, O’Leary DS, Cizadlo T, Arndt S, Rezai K, Boles Ponto LL, Watkins GL, Hichwa RD: Schizophrenia and cognitive dysmetria: a positron-emission tomography study of dysfunctional prefrontal-thalamic-cerebellar circuitry. Proc Natl Acad Sci USA 1996; 93:9985–9990Google Scholar



