Predictive Value of Acute Neuroplastic Response to rTMS in Treatment Outcome in Depression: A Concurrent TMS-fMRI Trial
Abstract
Objective:
The study objective was to investigate the predictive value of functional connectivity changes induced by acute repetitive transcranial magnetic stimulation (rTMS) for clinical response in treatment-resistant depression.
Methods:
Cross-sectional changes in functional connectivity induced by a single concurrent rTMS-fMRI session were assessed in 38 outpatients with treatment-resistant depression (26 of them female; mean age, 41.87 years) who subsequently underwent a 4-week course of rTMS. rTMS was delivered at 1 Hz over the right dorsolateral prefrontal cortex. Acute rTMS-induced functional connectivity changes were computed and subjected to connectome-based predictive modeling to test their association with changes in score on the Montgomery-Åsberg Depression Rating Scale (MADRS) after rTMS treatment.
Results:
TMS-fMRI induced widespread, acute, and transient alterations in functional connectivity. The rTMS-induced connectivity changes predicted about 30% of the variance of improvement in the MADRS score. The most robust predictive associations involved connections between prefrontal regions and motor, parietal, and insular cortices and between bilateral regions of the thalamus.
Conclusions:
Acute rTMS-induced connectivity changes in patients with treatment-resistant depression may index macro-level neuroplasticity, relevant to interindividual variability in rTMS treatment response. Large-scale network phenomena occurring during rTMS might be used to inform prospective clinical trials.
Major depressive disorder is the leading cause of disability in the world (1). It is estimated that approximately 40% of patients with major depression do not respond to antidepressants and suffer from treatment-resistant depression (2). Repetitive transcranial magnetic stimulation (rTMS) is a first-line treatment option for treatment-resistant depression (3), and it is becoming widely implemented and used around the world. rTMS most commonly targets either the right or the left dorsolateral prefrontal cortex (DLPFC), both of which achieve similar efficacy and tolerability (4, 5).
Stimulation of the DLPFC with rTMS induces local activation of neurons as well as transsynaptic activation of distal functionally or structurally connected brain regions (6–8). Therefore, its therapeutic effect on the depressive syndrome may span several symptomatic domains, such as mood, suicidal ideation, neurovegetative symptoms, and anxiety (9).
Recent methodological developments enabling concurrent functional MRI (fMRI) in conjunction with rTMS have opened a window for investigating mechanistic questions about rTMS (10). During concurrent rTMS-fMRI, the TMS pulses are delivered inside the MRI scanner during acquisition of the blood-oxygen-level-dependent fMRI signal. An advantage of this technique is that it permits quantification of acute rTMS-induced changes in the fMRI signal at the target site and across the whole brain (11, 12). A comprehensive evaluation of rTMS-induced functional changes is an essential step in understanding its therapeutic action in treatment-resistant depression, as depression is known to involve functional dysregulation in multiple neural networks (13).
Emerging yet compelling evidence in healthy individuals suggests that a single application of TMS targeting prefrontal or hippocampal regions induces acute changes in the connectivity and activity of the target and distal brain regions (14). Moreover, these alterations can have immediate behavioral consequences; for example, a single session of rTMS targeting the hippocampus has been associated with enhancing memory encoding and memory formation (14, 15). Two studies have provided evidence of acute rTMS-induced changes in patients with major depression; rTMS application over the left DLPFC induced acute alterations in the activity of bilateral prefrontal regions and of subcortical structures including the thalamus, insula, and amygdala (7, 8).
Neuroplasticity is currently used as a general umbrella term to describe the ability of the brain to respond to external influences by altering its organization at multiple levels, from molecular to macro-level changes in regional brain activity and connectivity. Accordingly, the acute changes induced by TMS are likely to reflect macro-level neuroplasticity. Whether these acute macro-level neuroplastic changes are relevant to the rTMS antidepressant response in treatment-resistant depression has not yet been assessed, as studies have focused exclusively on pretreatment neuroimaging features of the rTMS treatment response (6, 16).
In this context, the aim of the present study was to characterize acute rTMS-induced neuroplasticity in treatment-resistant depression, as inferred from functional connectivity changes induced by a concurrent TMS-fMRI session, and to assess its association with clinical response to subsequent course of rTMS treatment. Since major depression is linked to abnormal prefrontal-subcortical connectivity involved with emotion regulation (13), we hypothesized that acute rTMS-induced changes in the connectivity of these regions might have predictive value for treatment response to a course of rTMS.
Methods
Participants
Participants were adult outpatients with treatment-resistant depression recruited from November 1, 2018, to March 30, 2020, as part of a 4-week open-label clinical trial of rTMS in treatment-resistant depression; enrolled participants were offered the option of additionally participating in a single session of concurrent TMS-fMRI at study entry. The study was approved by the Clinical Research Ethics Board at University of British Columbia and the Vancouver Coastal Health Research Institute. All participants provided informed consent.
Eligible patients met DSM-5 criteria for major depressive disorder, were in an acute depressive episode (defined as a minimum score of 22 on the Inventory of Depressive Symptomatology [17]) at study enrollment, had a history of treatment resistance to antidepressants, and had been on the same psychotropic medication regimen for the preceding 4 weeks. Participants were screened to exclude medical and psychiatric comorbidity, suicidal ideation, and substance use disorders in the preceding 3 months and to exclude current treatment with anticonvulsants or high-dose benzodiazepines (see the online supplement).
Diagnostic assessments were conducted using the Mini-International Neuropsychiatric Interview, version 5.0 (18), and treatment resistance was defined according to the Antidepressant Treatment History Form (19). The composite crystallized cognition score derived from the NIH Toolbox (www.nihtoolbox.org) was used as an estimate of premorbid IQ. Handedness was assessed by self-report.
A total of 73 patients were screened, of whom 58 were eligible for and enrolled in the clinical trial. Of these patients, 52 were eligible and willing to participate in the neuroimaging protocol; however, 14 were later excluded because of missing data, noncompletion of the clinical trial, or poor imaging quality (see the online supplement). The final sample comprised 38 patients who completed both the neuroimaging and the treatment protocols and had high-quality neuroimaging data. The sample’s demographic and clinical characteristics are summarized in Table 1.
| Characteristic | ||
|---|---|---|
| Mean | SD | |
| Age (years) | 41.84 | 16.12 |
| Educationa (years) | 15.21 | 2.12 |
| Crystallized cognition scoreb | 109.39 | 15.45 |
| N | % | |
| Female | 26 | 68 |
| Right-handed | 32 | 84 |
| Unemployed | 19 | 50 |
| Mean | SD | |
| Length of current episode (months) | 46.08 | 68.00 |
| Antidepressant Treatment History Form | ||
| Score | 9.18 | 4.83 |
| N | % | |
| One failed trial | 21 | 55 |
| Two failed trials | 13 | 34 |
| Three or more failed trials | 4 | 11 |
| Psychotropic medication | 33 | 87 |
| None | 5 | 13 |
| Antidepressants | 32 | 84 |
| Benzodiazepines | 9 | 24 |
| Antipsychotics | 16 | 42 |
| Mood stabilizers | 2 | 5 |
| Sedatives | 5 | 13 |
| Stimulants | 9 | 24 |
| Combination of psychotropics | 33 | 87 |
| Mean | SD | |
| Antidepressant dosage (escitalopram equivalents, mg) | 24.05 | 21.48 |
| Benzodiazepine dosage (lorazepam equivalents, mg) | 0.53 | 1.06 |
| Montgomery-Åsberg Depression Rating Scale | ||
| Score at study entry | 29.50 | 6.84 |
| Score at study end | 18.63 | 9.62 |
| N | % | |
| Responderc | 16 | 42 |
| Remitterd | 8 | 21 |
TABLE 1. Demographic and clinical characteristics of the sample (N=38)
Neuroimaging Acquisition and Concurrent rTMS-fMRI Procedures
Whole-brain T1-weighted and resting-state fMRI (rsfMRI) data were acquired in a single session during which the TMS coil was placed on patients’ heads in a Philips Achieva 3-T scanner (Best, the Netherlands) at the University of British Columbia MRI Research Centre. An array of two single-channel SENSE Flex-L coils were used in place of a volume head coil to allow optimal positioning of the TMS coil. Following the T1-weighted scan, three separate 5-minute echo planar imaging (EPI) sequences were sequentially acquired in a sandwich design: 1) rsfMRI0, 2) concurrent rTMS-fMRI, and 3) rsfMRI1. There was no difference between rsfMRI0 and rsfMRI1 in terms of equipment settings, and the only difference between an rsfMRI scan and a concurrent rTMS-fMRI scan was that during the latter, a 1-Hz TMS pulse was delivered, whereas during rsfMRI scans, the TMS stimulator was not enabled. Details of the acquisition parameters are provided in the online supplement.
The concurrent rTMS-fMRI scan followed established procedures (20). Prior to conducting the clinical trial, the implementation of the TMS-fMRI system at our site involved a systematic process and comprehensive quality assessment (see Figures S1 and S2 in the online supplement) (21, 22). The individual resting motor threshold was measured in accordance with standard clinical practice (23) by visual inspection of contraction of the right pollicis brevis muscles with the aid of the TMS Motor Threshold Assessment Tool (https://www.clinicalresearcher.org/software.htm). Low-frequency (1 Hz) rTMS pulses were delivered at 100% of resting motor threshold over the right DLPFC, which was determined by the Beam-F3 method (24), with an MR-compatible figure-of-eight MRi-B91 coil and a MagPro-X100 stimulator (MagVenture, Farum, Denmark). These procedures are detailed in the online supplement.
Construction of the Whole-Brain Functional Network
Functional imaging data preprocessing was carried out using SPM12 (http://www.fil.ion.ucl.ac.uk/spm) (see the online supplement). No significant differences in data quality were noted across the three rsfMRI acquisitions with respect to mean framewise displacement and temporal signal-to-noise ratio (see Figure S3 in the online supplement). Subsequently, and for each of the three fMRI acquisitions (rsfMRI0, concurrent rTMS-fMRI, and rsfMRI1), the Human Brainnetome Atlas was used to parcellate the brain into 210 cortical and 36 subcortical regions (total, 246 regions) (25). For each participant, the rsfMRI time course of each brain region (i.e., node) was averaged across all its constituent voxels. Then a 246×246 correlation matrix was calculated modeling the Fisher z-transformed Pearson correlations between the time courses of each pair of nodes. Thus, each element (i.e., edge) in the correlation matrix represents the functional connectivity strength between two nodes (see the online supplement for details).
Repetitive Transcranial Magnetic Stimulation Treatment
Following the neuroimaging data acquisition, all participants received rTMS treatment once daily on weekdays for 4 consecutive weeks. Each of these sessions followed a protocol delivering 1-Hz rTMS over the right DLPFC at 120% of resting motor threshold for a total of 1,800 pulses per each 30-minute treatment session. The right DLPFC was localized with an MRI-guided neuronavigation system (Visor 2.0, ANT Neuro, Enschede, the Netherlands), using previously established coordinates (Montreal Neurological Institute coordinates, x=36; y=44; z=26) (26). All but five participants received concomitant medication (Table 1), which remained the same throughout the clinical trial. (A schematic diagram of the clinical trial design is provided in Figure S4 in the online supplement.)
Depressive symptom severity was measured with the Montgomery-Åsberg Depression Rating Scale (MADRS) (27). Treatment efficacy was based on the degree of global symptomatic change at the completion of the 4 weeks of treatment compared to the pretreatment baseline (i.e., MADRS score at study start minus MADRS score at study end); hereafter we refer to this measure as the MADRS change score.
Statistical Analysis
Assessment of rTMS-induced connectivity changes.
This analysis had the dual aim of quantifying the acute rTMS connectivity changes and assessing their persistence after the conclusion of the rTMS administration. Accordingly, we conducted pairwise t tests between the rsfMRI0 scan and the concurrent rTMS-fMRI scan and between the rsfMRI0 scan and the rsfMRI1 scan. The significance threshold for detecting changes between scans was set at p<0.05 following false discovery rate (FDR) correction.
Connectome-based predictive modeling of clinical outcome.
We used a connectome-based predictive modeling (CPM) technique to predict the MADRS change score based on whole-brain functional connectivity of the rsfMRI0 and rTMS-fMRI scans. This is a fully data-driven approach that accommodates uncertainty about the spatial distribution of connectivity changes relevant to clinical outcome in treatment-resistant depression. The protocol and scripts of CPM are freely available in Shen et al. (28), and a detailed description is provided in the online supplement. Briefly, CPM involves three stages: 1) feature selection (i.e., selection of the edges to be entered into the model), 2) construction of the predictive model, and 3) evaluation of its accuracy. Using 10-fold cross-validation, we randomly divided participants into 10 subsets; nine folds were used as training sets, and the remaining fold was used as a testing set. In implementing feature selection, all edges that changed between the rsfMRI0 and rTMS-fMRI scans in the training set (at p<5×10–4 uncorrected; see the online supplement and Figure S6 in the online supplement) were first tested for their association with potential confounders, namely, sex, framewise displacement, age, antidepressant dosage (in escitalopram equivalents), benzodiazepine dosage (in lorazepam equivalents), medication burden (total number of psychotropic medications of different classes prescribed concomitantly), and rTMS dose. Age, sex, and framewise displacement were the only confounders with nominally significant associations, predicting up to just 2.3% of the changed edges. Edges that showed a statistically nominal association (p<5×10–2, uncorrected) with the MADRS change score were then identified using partial correlation analyses adjusted for sex, age, framewise displacement, antidepressant dosage, benzodiazepine dosage, medication burden, and rTMS dose. (See the online supplement and Figure S6 in the online supplement.) By summing the selected edge strengths (Fisher-normalized r values) from a participant’s connectivity matrix (28, 29), we generated a measure of each participant’s network strength. A multiple linear regression model of the association between the network strength and the MADRS change scores was built in the training set, with the aforementioned confounders as covariates (28). After establishing the optimal parameters, we repeated the above prediction procedure 10 times to generate 10 prediction scores for each participant, which were further averaged to obtain a single robust estimate for each participant. These scores were then correlated with the actual MADRS change scores using the Pearson correlation coefficient. Statistical significance was established by randomly shuffling the actual MADRS change scores across participants 1,000 times; linear models were estimated for each permutation using the 10-fold cross-validation procedure with 10 repetitions. This resulted in a distribution of correlation coefficients of the null hypothesis. The number of times the permuted r value was equal to or higher than the r value from the original data was divided by the number of permutations to provide a pperm value estimation. Edges selected to construct the network strength across 50% or more of the cross-validation repetitions were considered robust. The 50% threshold was considered a balanced option, as it accommodates the relatively small sample size while informing about the range of the edges that could be affected by rTMS.
Sensitivity Analysis
First, we repeated the analysis using the percentage change of the MADRS score as the outcome measure. Second, to illustrate the reproducibility of the results independent of parcellation scheme, we repeated all analyses with the HCP-MMP cortical atlas (360 cortical regions) (30) and the Melbourne subcortical atlas (32 subcortical regions) (31).
Results
rTMS-Induced Functional Connectivity Changes
Comparison of the functional connectivity between the rsfMRI0 and the rTMS-fMRI scans identified 43 edges that were significantly changed (i.e., decreased at the rTMS-fMRI scan relative to the rsfMRI0 scan) at pFDR<0.05 (Figure 1; see also Table S1 in the online supplement). No differences between the rsfMRI0 and rsfMRI1 scans survived correction for multiple testing (see Figure S5 in the online supplement).
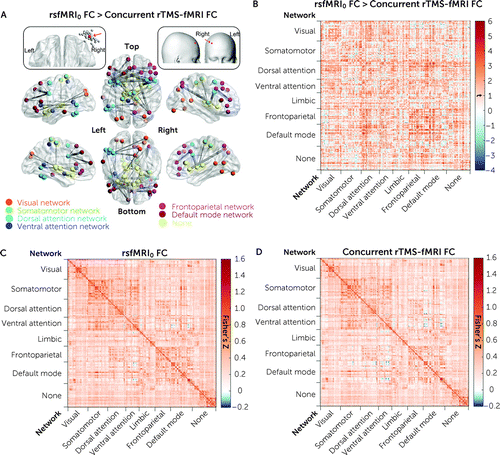
FIGURE 1. Changes in functional connectivity between rsfMRI0 and concurrent rTMS-fMRIa
aIn panel A, significant functional connectivity (FC) changes between the rsfMRI0 and concurrent rTMS-fMRI scans included 43 edges between 50 nodes; distribution of individualized targets (gray dots) over the scalp across the spatial extent of the Montreal Neurological Institute space is presented, and the red arrows indicate the centroid, which is shown with a red dot. Panel B shows the difference in functional connectivity between the rsfMRI0 scan and the concurrent rTMS-fMRI scan. Panel C shows the functional connectivity matrices of the rsfMRI0 scan, and panel D shows the functional connectivity matrices of the concurrent rTMS-fMRI scan. Significance was determined by false discovery rate (FDR) correction at p<0.05. Details of the brain nodes are presented in Table S1 in the online supplement. Brain nodes were labeled according to the Yeo 7-network parcellation (49).
Connectome-Based Predictive Modeling of Clinical Outcome
The mean of the MADRS change score was −10.87 (SD=11.20), and Cohen’s d was 0.97 (95% CI=0.58–1.35) (Table 1). CPM yielded a significant correlation between actual and predicted MADRS change scores (r=0.526, pperm<0.001) (Figure 2A,B). All edges that were included in all cross-validation iterations are listed in Table S3 in the online supplement. Notably, 19 edges appeared in at least 50% of the cross-validation iterations (Figure 2A). These edges were spatially distributed, and they did not preferentially involve specific networks.
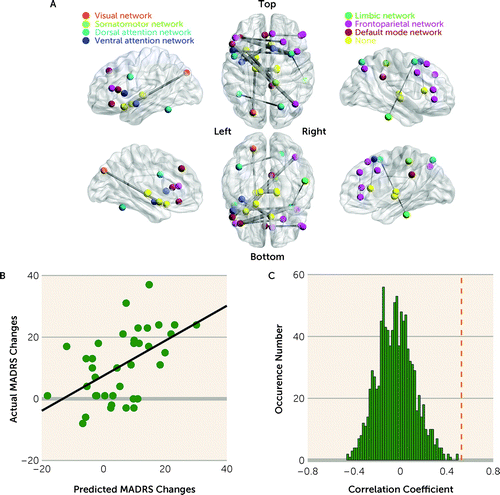
FIGURE 2. Connectome-based predictive modeling for MADRS changesa
aIn panel A, edges that were robust features for predicting the Montgomery-Åsberg Depression Rating Scale (MADRS) change scores were defined as those present in at least 50% of the cross-validation iterations; the thickness of the edges indicates their occurrence frequency (further details are provided in Table S3 in the online supplement). Panel B shows the correspondence between actual and predicted MADRS change scores generated using connectome-based predictive modeling (CPM), where Pearson’s correlation coefficient between actual and predicted MADRS change scores was 0.526. Panel C shows the distribution of the correlation coefficients between actual and predicted MADRS change scores over 1,000 permutations of the 10-fold cross-validation process used to determine statistical significance (pperm value). CPM successfully predicted MADRS changes (pperm<0.001). The vertical dotted line indicates the location of the actual correlation coefficient (r=0.526).
Results of Sensitivity Analysis
The results of the analyses using the percentage change of the MADRS score as the outcome measure remained the same as when using the MADRS change score (see Figure S7 in the online supplement). The results with the HCP-MMP cortical atlas and the Melbourne subcortical atlas (see Figures S9–S11 in the online supplement) largely replicated the results of the primary analyses, indicating the robustness of the model used here.
Discussion
Acute Neuroplastic Response to rTMS Predicts Treatment Outcome
Neuroimaging studies in major depression have documented abnormalities primarily implicating impaired connectivity between cortical and subcortical regions involved in reward responsiveness and voluntary control of emotion (32). The most robust findings involved prefrontal regions and their connections to motor, parietal, and insular cortices and between the thalamus bilaterally, which act as core nodes in emotional response and regulation circuits.
To our knowledge, this is the first study to test the predictive value of acute rTMS-induced functional connectivity changes in treatment-resistant depression. Importantly, the rTMS protocol employed during the concurrent rTMS-fMRI session matched the clinical protocol subsequently used for the treatment trial. We showed that stimulation of the right DLPFC induced widespread acute alterations in functional connectivity that predicted clinical outcome after a 4-week course of rTMS treatment. However, the functional rTMS-induced connectivity changes were present during the concurrent rTMS-fMRI but not in the rsfMRI1 scan. Therefore, these findings can be construed as a measure of the capacity of the brain to change its functional configuration in response to rTMS, and hence serve as an index of short-term macro-scale neuroplasticity. Differences in short-term neuroplasticity may provide a mechanistic explanation for interindividual variability in rTMS treatment response.
The Relevance of Macro-Level Neuroplasticity for Depression
Neuroplasticity is the umbrella term commonly used to encompass a multitude of biological changes observed at micro-level (e.g., gene expression, molecular levels), meso-level (e.g., synaptic changes), and macro-level (e.g., resting-state functional connectivity) phenotypes. Here we showed that acute rTMS-induced changes in connectivity may be predictive of treatment response because they may reflect patients’ neuroplastic potential. This interpretation is aligned with the conventional use of TMS as a neuroplasticity probe (33) and is directly relevant to major depressive disorder, which has been conceptualized as a disorder of compromised neuroplasticity. The neuroplasticity hypothesis of depression proposes that dysfunction in neural plasticity is an underlying pathogenic mechanism in depression. Major depressive disorder has been associated with abnormalities in structural plasticity (e.g., neurite morphology, adult neurogenesis), in functional synaptic plasticity, and in molecular and cellular mechanisms that support neural adaptation (34–36).
Since the acute rTMS-induced connectivity alterations were short-lived, our findings suggest that repeated stimulations or longer runs of stimulation (e.g., 1,800 pulses) may be required to induce long-lasting effects on connectivity (37). Our study design did not allow us to test whether repeated rTMS administration reinforces these acute changes or whether the pattern of rTMS-induced connectivity evolves further over multiple rTMS administrations. The most plausible explanation for the association between acute rTMS-induced connectivity changes and rTMS treatment response is that these early changes function as an index of the capacity of the brain to respond to the intervention.
Low-Frequency Right DLPFC rTMS Is Associated With Widespread Distal Functional Connectivity Changes
Our choice of whole-brain connectivity analysis was motivated by the existing knowledge gap regarding the effects of low-frequency rTMS protocols on the right DLPFC. We found widespread connectivity changes even in regions with no known direct functional or anatomical connections with the target site (Figure 1). Furthermore, there was a preponderance of interhemispheric functional connectivity changes in our data, a feature that merits further investigation, as a similar pattern has been reported in offline rTMS imaging studies (6, 38, 39). These findings add to the evidence that rTMS has the potential to influence brain functional organization beyond that predicted by the connectivity of the target site (40–42).
Of note, the predictive accuracy of the model for MADRS score improvement was based on functional connectivity reductions, since all the significant edges showed lower connectivity after rTMS compared to baseline. This observation resonates with propositions that low-frequency TMS pulses have an “inhibitory” effect on cortical excitability, as opposed to high-frequency TMS pulses, which may be excitatory (43, 44). Further rTMS-fMRI studies with different frequency pulses are required to test the association between the TMS-induced changes in functional connectivity and rTMS treatment response in major depression.
Limitations and Future Directions
Our results should be interpreted in light of several limitations. First, recent work using precision fMRI has shown that individualized functional parcellations require longer resting-state data acquisition protocols (45). While our design attempted to maximize the amount of fMRI data collected, long scanning sessions are challenging in clinical populations, and the addition of the TMS apparatus in the experimental setup further constrains the ability to conduct long fMRI runs. Future work may explore the use of multi-echo EPI sequences as a way to further optimize the amount of information over time in fMRI runs (46). Second, our results may not generalize to other rTMS protocols used in clinical practice. Our choice of rTMS protocol was motivated by four factors: 1) 1-Hz stimulation to the right DLPFC is an efficacious protocol widely used for treatment-resistant depression in clinical practice; 2) low-frequency rTMS is better tolerated than high-frequency protocols; 3) the occurrence of seizures is rare with 1-Hz stimulation, although seizure rates are generally low with other protocols as well; and 4) 1-Hz rTMS is associated with less fMRI data distortion (15).
The present findings require replication in larger cohorts with a sham-controlled design accommodating other prefrontal regions that are used as targets for rTMS application (47, 48). Acute rTMS-induced functional changes were not associated with antidepressant or benzodiazepine dosage or total medication burden. Although ideally, inclusion of medication-free patients would eliminate any potential confounding effect of psychotropic medication, this is not an ethical option for treatment-resistant depression.
Conclusions
This study provides evidence that rTMS-induced connectivity changes in the resting-state functional architecture of patients with treatment-resistant depression may index macro-level neuroplasticity. Furthermore, the acute neuroplasticity changes may be relevant to interindividual variability in rTMS treatment response. These findings provide a solid foundation for future studies to assess their generalizability in larger samples, across different rTMS protocols, and their relevance to connectivity changes following repeated rTMS administration.
1 : Depression and Other Common Mental Disorders: Global Health Estimates. Geneva, World Health Organization, 2017Google Scholar
2 : Diagnosis and definition of treatment-resistant depression. Biol Psychiatry 2003; 53:649–659Crossref, Medline, Google Scholar
3 : Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder, section 4: Neurostimulation treatments. Can J Psychiatry 2016; 61:561–575Crossref, Medline, Google Scholar
4 : Repetitive transcranial magnetic stimulation for the acute treatment of major depressive episodes: a systematic review with network meta-analysis. JAMA Psychiatry 2017; 74:143–152Crossref, Medline, Google Scholar
5 : Left versus right repetitive transcranial magnetic stimulation in treating major depression: a meta-analysis of randomised controlled trials. Psychiatry Res 2013; 210:1260–1264Crossref, Medline, Google Scholar
6 : Functional connectivity of the anterior cingulate cortex predicts treatment outcome for rTMS in treatment-resistant depression at 3-month follow-up. Brain Stimul 2020; 13:206–214Crossref, Medline, Google Scholar
7 : Acute left prefrontal transcranial magnetic stimulation in depressed patients is associated with immediately increased activity in prefrontal cortical as well as subcortical regions. Biol Psychiatry 2004; 55:882–890Crossref, Medline, Google Scholar
8 : Global connectivity and local excitability changes underlie antidepressant effects of repetitive transcranial magnetic stimulation. Neuropsychopharmacology 2020; 45:1018–1025Crossref, Medline, Google Scholar
9 : Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomised non-inferiority trial. Lancet 2018; 391:1683–1692Crossref, Medline, Google Scholar
10 : Mapping causal interregional influences with concurrent TMS-fMRI. Exp Brain Res 2008; 191:383–402Crossref, Medline, Google Scholar
11 : Causal interactions between fronto-parietal central executive and default-mode networks in humans. Proc Natl Acad Sci USA 2013; 110:19944–19949Crossref, Medline, Google Scholar
12 : Interleaved transcranial magnetic stimulation and functional magnetic resonance imaging: a translational tool. Clin Pharmacol Ther 2019; 106:714–716Crossref, Medline, Google Scholar
13 : Large-scale network dysfunction in major depressive disorder: a meta-analysis of resting-state functional connectivity. JAMA Psychiatry 2015; 72:603–611Crossref, Medline, Google Scholar
14 : Targeted enhancement of cortical-hippocampal brain networks and associative memory. Science 2014; 345:1054–1057Crossref, Medline, Google Scholar
15 : Evidence for immediate enhancement of hippocampal memory encoding by network-targeted theta-burst stimulation during concurrent fMRI. J Neurosci 2020; 40:7155–7168Crossref, Medline, Google Scholar
16 : Efficacy of transcranial magnetic stimulation targets for depression is related to intrinsic functional connectivity with the subgenual cingulate. Biol Psychiatry 2012; 72:595–603Crossref, Medline, Google Scholar
17 : The Inventory of Depressive Symptomatology (IDS): psychometric properties. Psychol Med 1996; 26:477–486Crossref, Medline, Google Scholar
18 : The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998; 59:22–33Medline, Google Scholar
19 A computer algorithm for calculating the adequacy of antidepressant treatment in unipolar and bipolar depression. J Clin Psychiatry 2003; 64:825–833Crossref, Medline, Google Scholar
20 : Vertex stimulation as a control site for transcranial magnetic stimulation: a concurrent TMS/fMRI study. Brain Stimul 2016; 9:58–64Crossref, Medline, Google Scholar
21 : Implementation of a TMS-fMRI system: a primer. bioRxiv 2021. doi: 10.1101/2021.05.19.444738
22 : A primer on developing interleaved transcranial magnetic stimulation with functional magnetic resonance imaging capability: the UBC experience. Brain Stimul 2017; 10:520–521 Crossref, Google Scholar
23 : Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J Clin Psychiatry 2017; 79:16cs10905 Google Scholar
24 : An efficient and accurate new method for locating the F3 position for prefrontal TMS applications. Brain Stimul 2009; 2:50–54Crossref, Medline, Google Scholar
25 : The Human Brainnetome Atlas: a new brain atlas based on connectional architecture. Cereb Cortex 2016; 26:3508–3526Crossref, Medline, Google Scholar
26 : Identification of reproducible individualized targets for treatment of depression with TMS based on intrinsic connectivity. NeuroImage 2013; 66:151–160 Crossref, Medline, Google Scholar
27 : A new depression scale designed to be sensitive to change. Br J Psychiatry 1979; 134:382–389 Crossref, Medline, Google Scholar
28 : Using connectome-based predictive modeling to predict individual behavior from brain connectivity. Nat Protoc 2017; 12:506–518Crossref, Medline, Google Scholar
29 : A neuromarker of sustained attention from whole-brain functional connectivity. Nat Neurosci 2016; 19:165–171 Crossref, Medline, Google Scholar
30 : A multi-modal parcellation of human cerebral cortex. Nature 2016; 536:171–178Crossref, Medline, Google Scholar
31 : Topographic organization of the human subcortex unveiled with functional connectivity gradients. Nat Neurosci 2020; 23:1421–1432 Crossref, Medline, Google Scholar
32 : Identifying predictors, moderators, and mediators of antidepressant response in major depressive disorder: neuroimaging approaches. Am J Psychiatry 2015; 172:124–138Link, Google Scholar
33 : Disrupting the brain to guide plasticity and improve behavior. Prog Brain Res 2006; 157:315–404Crossref, Medline, Google Scholar
34 : Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med 2016; 22:238–249Crossref, Medline, Google Scholar
35 : Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology 2008; 33:88–109Crossref, Medline, Google Scholar
36 : Neuroplasticity and cellular resilience in mood disorders. Mol Psychiatry 2000; 5:578–593Crossref, Medline, Google Scholar
37 : Trajectories of response to dorsolateral prefrontal rTMS in major depression: a THREE-D study. Am J Psychiatry 2019; 176:367–375Link, Google Scholar
38 : Abnormal functional connectivity within resting-state networks is related to rTMS-based therapy effects of treatment resistant depression: a pilot study. J Affect Disord 2017; 218:75–81Crossref, Medline, Google Scholar
39 : Functional disconnectivity of the hippocampal network and neural correlates of memory impairment in treatment-resistant depression. J Affect Disord 2019; 253:248–256 Crossref, Medline, Google Scholar
40 : Effects of repetitive transcranial magnetic stimulation on resting-state connectivity: a systematic review. Neuroimage 2020; 211:116596Crossref, Medline, Google Scholar
41 : Towards understanding rTMS mechanism of action: stimulation of the DLPFC causes network-specific increase in functional connectivity. Neuroimage 2017; 162:289–296Crossref, Medline, Google Scholar
42 : Resting-state networks link invasive and noninvasive brain stimulation across diverse psychiatric and neurological diseases. Proc Natl Acad Sci USA 2014; 111:E4367–E4375Crossref, Medline, Google Scholar
43 : Modulating functional connectivity between medial frontopolar cortex and amygdala by inhibitory and excitatory transcranial magnetic stimulation. Hum Brain Mapp 2019; 40:4301–4315Crossref, Medline, Google Scholar
44 : Bidirectional effects on interhemispheric resting‐state functional connectivity induced by excitatory and inhibitory repetitive transcranial magnetic stimulation. Hum Brain Mapp 2014; 35:1896–1905Crossref, Medline, Google Scholar
45 : Precision functional mapping of individual human brains. Neuron 2017; 95:791–807.e7 Crossref, Medline, Google Scholar
46 : Rapid precision functional mapping of individuals using multi-echo fMRI. Cell Rep 2020; 33:108540 Crossref, Medline, Google Scholar
47 : rTMS of the dorsomedial prefrontal cortex for major depression: safety, tolerability, effectiveness, and outcome predictors for 10 Hz versus intermittent theta-burst stimulation. Brain Stimul 2015; 8:208–215Crossref, Medline, Google Scholar
48 : 1Hz rTMS of the right orbitofrontal cortex for major depression: safety, tolerability, and clinical outcomes. Eur Neuropsychopharmacol 2018; 28:109–117Crossref, Medline, Google Scholar
49 : The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol 2011; 106:1125–1165 Crossref, Medline, Google Scholar



