Comprehensive Versus Usual Community Care for First-Episode Psychosis: 2-Year Outcomes From the NIMH RAISE Early Treatment Program
Abstract
Objective:
The primary aim of this study was to compare the impact of NAVIGATE, a comprehensive, multidisciplinary, team-based treatment approach for first-episode psychosis designed for implementation in the U.S. health care system, with community care on quality of life.
Method:
Thirty-four clinics in 21 states were randomly assigned to NAVIGATE or community care. Diagnosis, duration of untreated psychosis, and clinical outcomes were assessed via live, two-way video by remote, centralized raters masked to study design and treatment. Participants (mean age, 23) with schizophrenia and related disorders and ≤6 months of antipsychotic treatment (N=404) were enrolled and followed for ≥2 years. The primary outcome was the total score of the Heinrichs-Carpenter Quality of Life Scale, a measure that includes sense of purpose, motivation, emotional and social interactions, role functioning, and engagement in regular activities.
Results:
The 223 recipients of NAVIGATE remained in treatment longer, experienced greater improvement in quality of life and psychopathology, and experienced greater involvement in work and school compared with 181 participants in community care. The median duration of untreated psychosis was 74 weeks. NAVIGATE participants with duration of untreated psychosis of <74 weeks had greater improvement in quality of life and psychopathology compared with those with longer duration of untreated psychosis and those in community care. Rates of hospitalization were relatively low compared with other first-episode psychosis clinical trials and did not differ between groups.
Conclusions:
Comprehensive care for first-episode psychosis can be implemented in U.S. community clinics and improves functional and clinical outcomes. Effects are more pronounced for those with shorter duration of untreated psychosis.
Schizophrenia is associated with enormous personal suffering, disability, family burden, premature death, and societal cost (1, 2). Randomized trials suggest that intervention close to psychosis onset improves symptoms and functioning more than traditional care (3, 4). Comprehensive first-episode psychosis programs that emphasize low-dose antipsychotic medications, cognitive-behavioral psychotherapy, family education and support, and vocational and educational recovery have been implemented worldwide (5–11), but few randomized controlled trials have compared multimodal, multidisciplinary team approaches to usual care in first-episode psychosis (12–16). Such programs can be easier to implement in settings with a national health care system, which is perhaps why a multisite study of first-episode psychosis treatment has never been conducted in the United States in nonacademic, community clinics under existing reimbursement mechanisms. Despite the fact that academic centers play a key role in developing and testing new treatment strategies, such strategies must be implemented in typical “real world” settings.
This report presents 2-year outcome data from first-episode psychosis subjects participating in a multisite, randomized controlled trial comparing comprehensive, team-based treatment with usual care in U.S. community treatment centers. We also explored how the duration of untreated psychosis influences treatment response.
Method
The Early Treatment Program (ETP) study is part of the National Institute of Mental Health’s (NIMH) Recovery After an Initial Schizophrenia Episode (RAISE) initiative. RAISE aims to develop, test, and implement person-centered, integrated treatment approaches for first-episode psychosis that promote symptomatic and functional recovery. The background, rationale, and design of the RAISE-ETP trial have been described elsewhere (17).
Subjects
A total of 404 individuals between ages 15 and 40 were enrolled (a consort diagram appears in Figure S1 in the data supplement that accompanies the online edition of this article.) DSM-IV diagnoses of schizophrenia, schizoaffective disorder, schizophreniform disorder, brief psychotic disorder, or psychotic disorder not otherwise specified were included. Diagnoses of affective psychosis, substance-induced psychotic disorder, psychosis due to general medical conditions, clinically significant head trauma, or other serious medical conditions were excluded. All participants had experienced only one episode of psychosis (i.e., individuals with a psychotic episode followed by full symptom remission and relapse to another psychotic episode were excluded) and had taken ≤6 months of lifetime antipsychotic medications. All spoke English.
Written informed consent was obtained from adult participants and from legal guardians of those younger than 18 years old, who provided written assent. The study was approved by the institutional review boards of the coordinating center and the participating sites. The NIMH Data and Safety Monitoring Board provided study oversight.
Clinical Sites and Randomization
Thirty-four community mental health treatment centers in 21 states were selected via national search. Site eligibility criteria included experience treating people with schizophrenia; interest in offering early intervention services for first-episode psychosis; sufficient staff to implement the experimental intervention; ability to recruit an adequate number of subjects; and institutional assurance that research assessments would be completed. Academic centers or sites with existing first-episode psychosis programs were excluded.
RAISE-ETP employed a cluster randomization design; that is, randomization by clinic rather than by individual patient (18). Clinics were randomly assigned to the experimental intervention (N=17) or to standard care (N=17). None withdrew after randomization.
Interventions
The experimental treatment, NAVIGATE (19), includes four core interventions: personalized medication management (assisted by COMPASS, a secure, web-based decision support system developed for RAISE-ETP); family psychoeducation; resilience-focused individual therapy; and supported employment and education (SEE). Treatment was supported through existing funding mechanisms except for SEE, which is not supported in many locations. SEE services (5 hours/week) were supported with research funds.
Treatment components were offered and implemented within a shared decision-making, patient-preference framework (20). Weekly team meetings facilitated communication and coordination. NAVIGATE sites received initial training in team-based first-episode psychosis interventions, and ongoing expert consultation facilitated fidelity (19). We continually assessed clinicians’ competence and monitored team functioning. These assessments will be reported later.
The control condition, “community care,” is psychosis treatment determined by clinician choice and service availability. Community care sites received no additional training or supervision except for guidance regarding subject recruitment, retention, and collection of research data.
Research Infrastructure
Part-time study directors and research assistants recruited subjects and performed on-site research assessments. All research personnel participated in training on goals and procedures. Subject attrition was minimized through regular contact by the coordinating team with research staff to reinforce retention efforts and through a progressive reimbursement schedule for trial participants completing outcome assessments.
Trial Duration
Enrollment occurred between July 2010 and July 2012. Each subject was provided at least 2 years of treatment. There was no threshold for discontinuing patients, even after lengthy interruptions. Study assessments were suspended during periods of incarceration or hospitalization but were resumed after release or discharge. Subjects could continue research assessments even if they discontinued NAVIGATE or community care treatment. The last subject who entered completed 2 years of treatment in July 2014.
Assessment Strategy and Measures
Trained interviewers using live, two-way video conferencing performed diagnostic interviews and assessments of symptoms and quality of life. Remote assessment via two-way video conferencing is comparable to face-to-face assessments in patient acceptability and reliability (21). Centralized assessors, who were masked to individual treatment assignments and overall study design, administered the Structured Clinical Interview for DSM-IV (SCID) for diagnosis and duration of untreated psychosis; the Positive and Negative Syndrome Scale (PANSS) (22); the Clinical Global Impressions Scale (CGI) severity of illness score; the Calgary Depression Scale for Schizophrenia (23); and the Heinrichs-Carpenter Quality of Life Scale (24), our primary outcome measure. The Quality of Life Scale has 21 items rated from a semistructured interview. It covers areas including sense of purpose, motivation, emotional and social interaction, role functioning, and engagement in regular activities. The SCID was completed at baseline and at 1 year; other measures were completed every 6 months.
Site research assistants interviewed participants monthly to complete the Service Use and Resource Form (25, 26) to capture participation in work or school; use of inpatient, residential, emergency, and outpatient mental health and medical services in the previous month; and self-reported days of alcohol or drug use. The Service Use and Resource Form includes questions concerning four specific NAVIGATE interventions, allowing treatment groups to be compared on receipt of key services. Time remaining in treatment was defined as the time from randomization to the time of the last mental health service received based on the Service Use and Resource Form assessments.
Data Analysis
The analysis of the primary outcome (the total Quality of Life score) compared treatments across 2 years (at baseline, at 6 months, at 12 months, at 18 months, and at 24 months). The analysis model was a three-level mixed-effects linear regression model with a linearized term for time, an interaction of treatment group by linearized time, and a random intercept and a random slope for linearized time at both patient and site levels. To enhance analysis interpretability, time was linearized through square root or logarithmic transformation because outcome plots over time for both treatment groups showed greater improvement in the earlier months and leveling off in the later months. The group organized by linearized time interaction was tested to assess the difference between treatments in the rate of improvement on the Quality of Life Scale. The alpha level for analysis of the total Quality of Life Scale score was preset at 0.05.
Clustered, randomized trials typically have a limited number of clusters that could cause an imbalance between treatment groups on baseline measures, and this imbalance may confound the relationship between treatments and patient-level outcomes. A generalized linear mixed-effects regression model with a random effect (intercept) for site was used to identify baseline measures that were significantly different between the treatment groups. The identified baseline variables that were also significantly correlated with the Quality of Life Scale were included in the above model. The main effect of treatment would have been included had the baseline Quality of Life Scale been significantly different between the two treatment groups because the baseline Quality of Life Scale was modeled as part of the longitudinal response. A sensitivity analysis (available upon request) with no baseline covariate adjustment was also conducted based on the expectation of no significant baseline differences between treatment groups due to randomization.
In a further analysis, time was coded into dummy variables for categorical levels following baseline (at 6 months, at 12 months, at 18 months, and at 24 months). An additional dummy variable for baseline time (time=0) would have been included had the baseline Quality of Life Scale been significantly different between the two treatment groups. Random effects for site and patient were also included. The same adjustment for potentially confounding baseline variables, as described above, was used. Interaction between treatment group and each of the dummy variables of time was tested to identify specific times at which there were significant differences between treatment groups.
In models with either linearized or categorical time, Akaike’s information criterion was used to compare independent, first-order autoregressive, and unstructured covariance structures for repeated measures. Analyses of secondary outcomes, using a comparable approach, were conducted on subscales of the Quality of Life Scale and on measures of symptoms (PANSS total score and five factors, see reference 27; CGI severity score; and the Calgary Depression Scale for Schizophrenia). Service use analyses were based on a mixed-effects Poisson regression model with both site- and patient-level random effects. The same approach, as described above, was applied to the inclusion of baseline covariates and time transformation. We did not adjust for multiple comparisons in the analysis of secondary outcomes, which were exploratory in nature (28).
To evaluate the effect of duration of untreated psychosis as a moderator of treatment effectiveness, an additional fixed effect of duration of untreated psychosis (representing values below or above the median) and a three-way interaction of duration of untreated psychosis by linearized time and by a treatment indicator (referring to one of the treatment groups) were evaluated. This three-way interaction compared the slope of linearized time for patients in the indicated treatment group who were above and who were below the median duration of untreated psychosis. The median split approach was selected to maximize statistical power and optimize interpretation of findings. The moderating effect of duration of untreated psychosis was tested using the three-way interaction only after the significant difference in the rate of improvement between the two treatment groups was declared.
The 2-year treatment effect size was determined by the change from baseline to 2 years using the estimates derived from the mixed model, divided by the pooled baseline standard deviation of the outcome measure (Cohen’s d) (29).
For each analysis, we checked the model assumptions and diagnostics, including the normality assumption for random effects and the distribution of residuals.
Sample size calculations for mixed-effects linear regression analyses assumed that the within-subject intraclass correlation coefficient (ICC) would range from 0.30 to 0.60 and that the within-site ICC would be 0.10. With at least N=145 per group, even after attrition, the proposed design provided power in excess of 0.90 to detect an overall group difference and the difference in rate of change over time for a standardized effect size at the 24-month visit as small as 0.40 standard deviation units (9 Quality of Life Scale points).
Results
Participant Characteristics
NAVIGATE and community care groups included 223 and 181 patients, respectively. Demographic and other baseline characteristics are presented in Table 1. The mean age was 23 in both groups. Of the two groups, 90% of community care patients and 89% of NAVIGATE patients met schizophrenia-spectrum criteria; the proportions for schizophrenia were 56% and 51%, respectively. Neither mean (Table 1) nor median (88 weeks for community care and 66 weeks for NAVIGATE, z=0.94, p=0.35) duration of untreated psychosis differed significantly between groups. Median duration was 74 weeks for the entire sample. Most patients (71% in both groups) lived with their families. Detailed descriptions of duration of untreated psychosis findings (30), baseline medication status and history (31), and baseline medical and metabolic measures (32) have been published elsewhere. NAVIGATE participants differed significantly from community care participants on four measures: NAVIGATE had significantly more males (77.6% compared with 66.2%, p=0.05); a smaller proportion with prior hospitalization (76.3% compared with 81.6%, p=0.05); worse PANSS total scores (p=0.02); and fewer attending school at baseline (15.7% compared with 26.0%, p=0.03).
| Characteristic | Community Care (N=181) | NAVIGATE (N=223) | Unadjusted p | Analysis | ||||
|---|---|---|---|---|---|---|---|---|
| Categorical Variables | N | % | N | % | Unadjusted pa | F | df | pb |
| Male | 120 | 66 | 173 | 78 | 0.04 | 3.86 | 1, 370 | 0.05 |
| Race | 0.01 | 1.28 | 2, 336 | 0.28 | ||||
| White | 80 | 44 | 138 | 62 | ||||
| African American | 89 | 49 | 63 | 28 | ||||
| Other | 12 | 7 | 22 | 10 | ||||
| Hispanic | 18 | 10 | 55 | 25 | 0.01 | 3.24 | 1, 370 | 0.07 |
| Marital status | 0.67 | 0.28 | 2, 336 | 0.76 | ||||
| Presently married | 10 | 6 | 14 | 6 | ||||
| Widowed/divorced/separated | 8 | 4 | 14 | 6 | ||||
| Never married | 163 | 90 | 195 | 87 | ||||
| Current residence | 0.97 | 0.08 | 3, 302 | 0.97 | ||||
| Independent living | 32 | 18 | 40 | 18 | ||||
| Supported or structured | 7 | 4 | 7 | 3 | ||||
| Family, parents, grandparents, or sibling | 129 | 71 | 158 | 71 | ||||
| Homeless, shelter, or other | 13 | 7 | 18 | 8 | ||||
| Patient’s education | 0.74 | 0.36 | 3, 301 | 0.78 | ||||
| Some college or higher | 54 | 30 | 71 | 32 | ||||
| Completed high school | 58 | 32 | 75 | 34 | ||||
| Some high school | 58 | 32 | 67 | 30 | ||||
| Some or completed grade school | 11 | 6 | 9 | 4 | ||||
| Mother’s education | 0.17 | 1.06 | 3, 302 | 0.37 | ||||
| Some college or higher | 65 | 36 | 102 | 46 | ||||
| Completed high school | 51 | 28 | 60 | 27 | ||||
| Some high school or grade school | 32 | 18 | 27 | 12 | ||||
| No school or unknown | 33 | 18 | 34 | 15 | ||||
| Current student | 47 | 26 | 35 | 16 | 0.01 | 4.77 | 1, 370 | 0.03 |
| Currently working | 30 | 17 | 28 | 13 | 0.25 | 1.30 | 1, 370 | 0.25 |
| Type of insurance | 0.03 | 1.28 | 2, 333 | 0.28 | ||||
| Private | 27 | 15 | 55 | 25 | ||||
| Public | 66 | 37 | 61 | 27 | ||||
| Uninsured | 88 | 49 | 104 | 47 | ||||
| SCIDc diagnosis | 0.60 | 0.58 | 5, 234 | 0.72 | ||||
| Schizophrenia | 101 | 56 | 113 | 51 | ||||
| Schizoaffective bipolar | 13 | 7 | 11 | 5 | ||||
| Schizoaffective depressive | 25 | 14 | 32 | 14 | ||||
| Schizophreniform provisional or definite | 24 | 13 | 43 | 19 | ||||
| Brief psychotic disorder | 1 | 1 | 1 | 1 | ||||
| Psychotic disorder not otherwise specified | 17 | 9 | 23 | 10 | ||||
| Lifetime alcohol use | 0.08 | 2.49 | 2, 336 | 0.08 | ||||
| Did not meet criteria | 123 | 68 | 134 | 60 | ||||
| Met abuse criteria | 16 | 9 | 36 | 16 | ||||
| Met dependence criteria | 42 | 23 | 53 | 24 | ||||
| Lifetime cannabis use | 0.22 | 146 | 2, 336 | 0.23 | ||||
| Did not meet criteria | 123 | 68 | 137 | 61 | ||||
| Met abuse criteria | 21 | 12 | 39 | 18 | ||||
| Met dependence criteria | 37 | 20 | 47 | 21 | ||||
| Prescribed one or more antipsychotics at consent | 155 | 86 | 182 | 82 | 0.28 | 0.36 | 1, 370 | 0.55 |
| Number of prior hospitalizations | 0.01 | 3.91 | 1, 368 | 0.05 | ||||
| 0 | 34 | 19 | 54 | 24 | ||||
| 1 | 75 | 41 | 106 | 48 | ||||
| 2 | 32 | 18 | 37 | 17 | ||||
| ≥3 | 40 | 22 | 24 | 11 | ||||
| Continuous Variables | Mean | SD | Mean | SD | Unadjusted pa | F | df | pb |
| Age | 23.08 | 4.90 | 23.18 | 5.21 | 0.83 | 0.14 | 1, 370 | 0.71 |
| Duration of untreated psychosis (weeks) | 211.43 | 277.49 | 178.91 | 248.73 | 0.35d | 0.97 | 1, 369 | 0.33 |
| Heinrichs-Carpenter Quality of Life Scale | ||||||||
| Total score | 54.77 | 18.99 | 50.89 | 18.44 | 0.04 | 2.70 | 1, 369 | 0.10 |
| Interpersonal relations | 20.07 | 8.53 | 19.51 | 8.84 | 0.52 | 0.51 | 1, 369 | 0.48 |
| Instrumental role | 6.82 | 6.86 | 4.54 | 6.07 | 0.01 | 7.51 | 1, 369 | 0.01 |
| Intrapsychic foundations | 21.39 | 7.29 | 20.36 | 6.69 | 0.14 | 1.60 | 1, 369 | 0.21 |
| Common objects and activities | 6.49 | 2.25 | 6.48 | 2.36 | 0.95 | 0.03 | 1, 369 | 0.86 |
| Positive and Negative Syndrome Scale | ||||||||
| Total score | 74.54 | 14.87 | 78.32 | 14.95 | 0.01 | 5.56 | 1, 369 | 0.02 |
| Factor scores (27) | ||||||||
| Positive | 12.13 | 3.79 | 12.32 | 3.88 | 0.62 | 0.32 | 1, 369 | 0.57 |
| Negative | 16.34 | 4.96 | 16.98 | 5.34 | 0.22 | 0.80 | 1, 369 | 0.37 |
| Disorganized/concrete | 7.34 | 2.63 | 8.18 | 2.83 | 0.01 | 7.03 | 1, 369 | 0.01 |
| Excited | 6.38 | 2.30 | 7.05 | 3.06 | 0.02 | 5.83 | 1, 369 | 0.02 |
| Depressed | 7.93 | 3.42 | 8.16 | 3.22 | 0.49 | 0.34 | 1, 369 | 0.56 |
| Calgary Depression Scale for Schizophrenia | 4.66 | 4.30 | 4.65 | 4.27 | 0.99 | 0.01 | 1, 369 | 0.99 |
| Clinical Global Impressions Severity Scale | 3.96 | 0.83 | 4.12 | 0.80 | 0.05 | 3.37 | 1, 369 | 0.07 |
| Duration of lifetime antipsychotic medication at consent (days) | 48.46 | 48.98 | 40.60 | 42.88 | 0.13d | 2.74 | 1, 369 | 0.10 |
TABLE 1. Participant Demographic and Clinical Characteristics at Baseline
Main Outcomes
On a series of treatment validity measures, NAVIGATE participants were much more likely to endorse receipt of key services included in the experimental intervention than patients in community care (Figure 1; p<0.0001 for each of the four services). Participants assigned to NAVIGATE remained in treatment longer than community care patients (a median of 23 months compared with a median of 17 months, p<0.004; see Figure S2 in the online data supplement) and were more likely to have received mental health outpatient services each month than community care subjects (a mean of 4.53 services, SD=5.07, compared with a mean of 3.67 services, SD=5.93) (t=2.49, p=0.013).
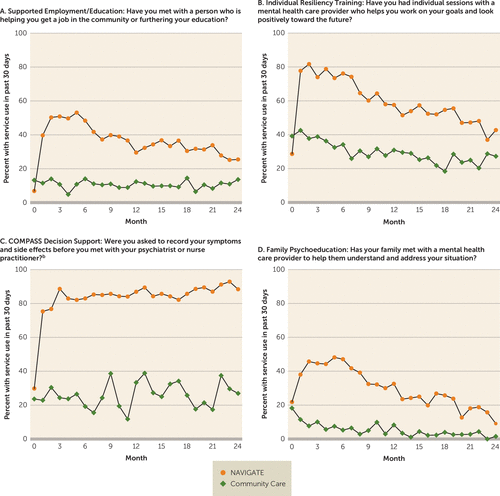
FIGURE 1. Patient Self-Reported Use of NAVIGATE Model Targeted Services During Study Period at NAVIGATE and Community Care Sitesa
a The test statistics and p values are of the treatment effect from a mixed logistic model. Comparing NAVIGATE with community care, for supported employment t=5.98 and p<0.0001; for individual resiliency training t=5.75 and p<0.0001; for COMPASS decision support t=5.15 and p<0.0001; and for family psychoeducation t=6.48 and p<0.0001.
b Limited to patients being prescribed medications.
On the primary outcome measure, the Quality of Life Scale total score, NAVIGATE participants experienced significantly greater improvement during the 2-year assessment period than those in community care (group by time interaction p<0.02; Figure 2; Table 2; and Table S1 in the data supplement), with an effect size of 0.31 and of a clinically meaningful magnitude (33). More improvement was also found on the subscales “interpersonal relations,” “intrapsychic foundations” (i.e., sense of purpose, motivation, curiosity, and emotional engagement), and engagement with “common objects and activities.” Service Use and Resource Form data showed significantly greater gains for NAVIGATE regarding the proportion of participants who were either working or going to school at any time during each month (group by time interaction p<0.05; see Figure S3 in the data supplement).
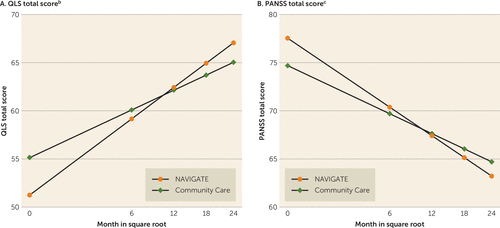
FIGURE 2. Model-Based Estimates of Heinrichs-Carpenter Quality of Life (QLS) Total Score and PANSS Total Scorea
a PANSS=Positive and Negative Syndrome Scale.
b Treatment by square root of time interaction, p=0.015.
c Treatment by square root of time interaction, p=0.016.
| Change From Baseline to 24 Months | Differential Change Between CC and NAV at 24 Months | Treatment by Time Interaction | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Measure | Mean | SE | Cohen’s d | Mean | SE | Cohen’s d | t | p | |
| Heinrichs-Carpenter Quality of Life Scale | |||||||||
| Total scoreb,c,d | CC | 9.891 | 1.918 | 0.53 | 5.902 | 2.408 | 0.31 | 2.45 | 0.0145 |
| NAV | 15.793 | 1.624 | 0.84 | ||||||
| Subscales | |||||||||
| Interpersonal relationsb,c,d | CC | 3.494 | 0.763 | 0.40 | 2.198 | 0.942 | 0.25 | 2.33 | 0.0199 |
| NAV | 5.691 | 0.635 | 0.65 | ||||||
| Instrumental roleb,c,d,e | CC | 3.418 | 0.801 | 0.52 | 1.854 | 1.059 | 0.28 | 1.75 | 0.0804 |
| NAV | 5.271 | 0.692 | 0.81 | ||||||
| Intrapsychic foundationsb,c,d | CC | 2.144 | 0.616 | 0.31 | 1.548 | 0.744 | 0.22 | 2.08 | 0.0377 |
| NAV | 3.692 | 0.510 | 0.53 | ||||||
| Common objects and activitiesb,c,d | CC | 0.971 | 0.184 | 0.42 | 0.483 | 0.221 | 0.21 | 2.18 | 0.0294 |
| NAV | 1.453 | 0.151 | 0.63 | ||||||
| Positive and Negative Syndrome Scale | |||||||||
| Total scoreb,c,e | CC | –9.989 | 1.383 | –0.67 | –4.324 | 1.792 | –0.29 | –2.41 | 0.0161 |
| NAV | –14.313 | 1.140 | –0.95 | ||||||
| Factor scores (27) | |||||||||
| Positiveb,c,e | CC | –2.519 | 0.385 | –0.66 | –0.224 | 0.502 | –0.06 | –0.45 | 0.6560 |
| NAV | –2.742 | 0.322 | –0.72 | ||||||
| Negativeb,c,e | CC | –2.280 | 0.530 | –0.44 | –0.693 | 0.680 | –0.13 | –1.02 | 0.3087 |
| NAV | –2.972 | 0.426 | –0.57 | ||||||
| Disorganized or concreteb,c,e | CC | –0.721 | 0.294 | –0.26 | –0.685 | 0.391 | –0.25 | –1.75 | 0.0804 |
| NAV | –1.406 | 0.258 | –0.51 | ||||||
| Excitedb,c,e | CC | –0.241 | 0.305 | –0.09 | –0.703 | 0.404 | –0.25 | –1.74 | 0.0823 |
| NAV | –0.944 | 0.265 | –0.34 | ||||||
| Depressedb,c,e | CC | –0.893 | 0.290 | –0.27 | –0.770 | 0.375 | –0.23 | –2.05 | 0.0407 |
| NAV | –1.662 | 0.239 | –0.50 | ||||||
| Clinical Global Impressions Severity Scaled | CC | –0.606 | 0.079 | –0.74 | –0.140 | 0.092 | –0.17 | –1.52 | 0.1292 |
| NAV | –7.460 | 0.066 | –9.12 | ||||||
| Calgary Depression Scale for Schizophreniab,d | CC | –1.196 | 0.327 | –0.28 | –0.785 | 0.365 | –0.18 | –2.15 | 0.0318 |
| NAV | –1.981 | 0.277 | –0.46 | ||||||
TABLE 2. The Estimated Model-Based Change From Baseline to 24 Months and the Differential Change by Treatment With Effect Sizea
NAVIGATE participants experienced greater improvement on PANSS total scores (p<0.02), on the PANSS depressive factor (p<0.05), and on the Calgary Depression Scale for Schizophrenia (p<0.04) between baseline and 24 months. There were no significant group differences on the CGI severity score.
The average rate of hospitalization was 3.2% per month for NAVIGATE participants and 3.7% per month for community care participants. Over the 2 years, 34% of the NAVIGATE group and 37% of the community care group (adjusted for length of exposure) had been hospitalized for psychiatric indications (n.s.).
Moderating Effect of Duration of Untreated Psychosis
Median duration of untreated psychosis was a significant moderator of the treatment effect on total Quality of Life Scale and PANSS scores over time (Figure 3; see Table S2 in the data supplement). There was a substantial difference in effect sizes comparing the change between treatments for participants with a duration of untreated psychosis of ≤74 weeks and those with a duration of untreated psychosis of >74 weeks: 0.54 compared with 0.07 for Quality of Life Scale, and 0.42 compared with 0.13 for PANSS scores, respectively.
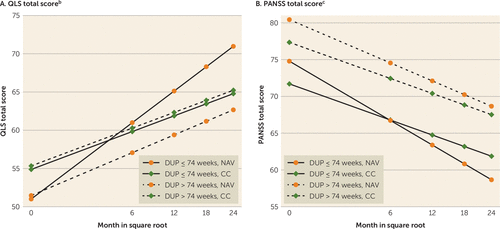
FIGURE 3. Heinrichs-Carpenter Quality of Life (QLS) Total Score and PANSS Total Score: Effects of Shorter or Longer Duration of Untreated Psychosis (DUP) Based on a Model With Square Root Transformation of Monthsa
a In the model, DUP and DUP by square root of time by treatment terms were included as covariates in addition to the covariates listed in Table 2. The DUP by square root of time term was found not to be significant for either outcome. PANSS=Positive and Negative Syndrome Scale; CC=Community Care; NAV=NAVIGATE.
b DUP by treatment by square root of time interaction, p=0.003.
c DUP by treatment by square root of time interaction, p=0.043.
Discussion
RAISE-ETP accomplished the primary goals of the NIMH RAISE initiative. We developed a comprehensive recovery-oriented, evidence-based intervention for first-episode psychosis (19); trained more than 100 community providers in early intervention principles and to deliver manual-based, coordinated specialty care; and successfully implemented the NAVIGATE model in 17 real-world community clinics serving a racially and ethnically heterogeneous patient mix. NAVIGATE programs operated continuously between 2010 and 2014, demonstrating sustained model implementation. RAISE-ETP is the first multisite, randomized controlled trial of coordinated specialty care conducted in the United States, and the first anywhere to simultaneously include all of the following elements: randomized concurrent controls; masked assessment of primary and secondary outcomes; and manual-driven intervention with ongoing training and fidelity metrics. Most importantly, NAVIGATE improved outcomes for patients over 24 months, with effects seen in length of time in treatment, quality of life, participation in work and school, and symptoms. These are outcomes of importance to service users, family members, and clinicians.
Our results are likely to generalize to many U.S. community care settings that wish to implement specialty care teams for young persons with first-episode psychosis. Insurance covered some NAVIGATE services (e.g., individual and family therapy, medication management), but supplements are needed to make first-episode services viable (34). Congress recently allocated additional funds to the Substance Abuse and Mental Health Services Administration (SAMHSA) to subsidize first-episode psychosis services not covered by insurance, such as assertive outreach, care coordination, and supported employment and education (35, 36). Since 2014, 32 states have moved toward earlier intervention by combining SAMHSA funds with services reimbursed by public or private insurance, and in some cases with increased state funding for first-episode psychosis programs.
Three multielement treatment studies have been conducted outside the United States, although only one (14, 15) included exclusively first-episode psychosis patients. The Lambeth Early Onset (LEO) study (12, 13, 37) randomly allocated 144 patients in London with a first or second psychotic episode to “specialist services” or to “care as usual” for 18 months. Patients had a median age of 25, 24% were Caucasian, and 58% were living with family. Data on duration of untreated psychosis were not provided. Individuals receiving specialist services had fewer readmissions (but were not less likely to have ever been readmitted or to have shorter admissions) and had better social and vocational functioning, quality of life, and medication adherence. At follow-up, only 58% of participants had a diagnosis of schizophrenia, schizotypal disorder, or delusional disorder.
In the Danish OPUS study (14, 15) 547 first-episode psychosis patients with <12 weeks of exposure to antipsychotic medications were randomly assigned to “integrated” or to “standard” treatment. Patients in OPUS and our study differed in age and living situation; patients in OPUS were 3 years older on average, and 78% lived alone or with a partner. The only data reported on duration of untreated psychosis was a median of <50 weeks. At 2-year follow-up, the integrated treatment group was more likely to have remained in treatment and had significantly lower levels of psychotic and negative symptoms, but there was no difference in mean number of days spent in hospital. The proportion of patients hospitalized was 59% in year 1 and 26% in year 2 among patients receiving integrated care. With standard care, the respective rates were 71% and 39%. Differences were significant during year 1 but not during year 2. Of note, overall hospitalization rates in both groups were considerably higher than in our study. Patients in integrated care experienced significantly less substance misuse, better adherence to care, and more satisfaction with care. Neither LEO nor OPUS included formal SEE, a robust evidence-based practice, which emphasizes further that these studies are not identical to our study.
Grawe et al. (38) studied 50 patients with less than 2 years of illness duration; most of the patients were diagnosed with schizophrenia, but not necessarily first episode. At 2 years, hospitalization rates were 33% in the enhanced intervention group and 50% among control subjects (difference not significant). Although individual outcomes did not differ, the percentage of participants having a good outcome based upon a “Clinical Composite Index” was significantly higher in the intervention group (53% compared with 25%).
In the United States, Srihari and colleagues (16) randomly assigned 120 first-episode psychosis patients with <12 weeks of antipsychotic medication exposure to the Specialized Treatment Early in Psychosis (STEP) program at an academic community mental health center or to usual care in the community. The mean duration of untreated psychosis was 40 weeks. Assessments were not masked. Compared with usual care recipients, after 1 year of participation, STEP recipients experienced significantly greater reductions in symptoms, required less inpatient care (hospitalization rates were 23% compared with 44%), and were more likely to be working or going to school. Neither quality of life nor social functioning differed between treatments.
Given NAVIGATE’s effect on treatment retention, quality of life, and symptom improvement, we expected a larger difference between treatment conditions in postenrollment hospitalization. However, the 34% rate for NAVIGATE is comparable to hospitalization rates for integrated treatment programs in the four prior multicomponent first-episode psychosis intervention studies (23%−59%). Postenrollment hospitalization rates for standard care in these studies (44%−71%) were uniformly higher than the rate in community care (37%). All sites randomized to community care had expressed eagerness to participate in RAISE-ETP and had the staff, administrative support, and desire to implement a coordinated specialty care program. Hence, community care sites may have had the motivation and resources available to serve clients with first-episode psychosis, resulting in lower hospitalization rates compared with unselected community sites.
The observation that patients with shorter duration of untreated psychosis derived substantially more benefit from NAVIGATE is important. Prolonged duration of untreated psychosis is an issue of national importance; reducing duration of untreated psychosis from current levels of >1 year to the recommended standard of <3 months (11) should be a major focus of applied research efforts.
A key question is the sustained benefit of comprehensive specialty care programs. The long-term OPUS trial outcomes suggest that the benefits of participation in a 2-year intensive early intervention program do not persist in a 5-year follow-up (39). It is also possible, as suggested by Linszen et al. (6), that the positive effects of intensive early treatment are sustained only when patients continue to receive specialized services. The length of time subjects were eligible to receive NAVIGATE services after the completion of the 2-year period that is the focus of this report varied. An ongoing follow-up study will extend outcome assessment for a total of 5 years to provide information on longer-term effects and optimal treatment duration.
Conclusions
The RAISE-ETP study demonstrates that diverse U.S. community clinics can implement a team-based model of first-episode psychosis care, producing greater improvement in clinical and functional outcomes as compared with standard care. These effects were more pronounced for those with shorter duration of untreated psychosis, suggesting that the receipt of appropriate first-episode psychosis treatment at the proper time in the illness course can have a substantial impact on outcomes.
1 : Global Burden of Disease and Risk Factors. Washington, DC, World Bank, 2006Crossref, Google Scholar
2 : A systematic review and meta-analysis of recovery in schizophrenia. Schizophr Bull 2013; 39:1296–1306Crossref, Medline, Google Scholar
3 : Early intervention services, cognitive-behavioural therapy and family intervention in early psychosis: systematic review. Br J Psychiatry 2010; 197:350–356Crossref, Medline, Google Scholar
4 : Preventing the second episode: a systematic review and meta-analysis of psychosocial and pharmacological trials in first-episode psychosis. Schizophr Bull 2011; 37:619–630Crossref, Medline, Google Scholar
5 : The promise of early intervention. Early Interv Psychiatry 2007; 1:294–307Crossref, Medline, Google Scholar
6 : Early intervention and a five year follow up in young adults with a short duration of untreated psychosis: ethical implications. Schizophr Res 2001; 51:55–61Crossref, Medline, Google Scholar
7 : One-year outcome in first episode psychosis patients in the Swedish Parachute project. Acta Psychiatr Scand 2002; 106:276–285Crossref, Medline, Google Scholar
8 McGorry PD, Jackson HJ (eds): Recognition and Management of Early Psychosis: A Preventive Approach. New York, Cambridge University Press, 1999Google Scholar
9 : Remission and recovery during the first outpatient year of the early course of schizophrenia. Schizophr Res 2011; 132:18–23Crossref, Medline, Google Scholar
10 : Evaluation of a multi-element treatment center for early psychosis in the United States. Soc Psychiatry Psychiatr Epidemiol 2012; 47:1607–1615Crossref, Medline, Google Scholar
11 : Early intervention and recovery for young people with early psychosis: consensus statement. Br J Psychiatry Suppl 2005; 48:s116–s119Crossref, Medline, Google Scholar
12 : The Lambeth Early Onset (LEO) Team: randomized controlled trial of the effectiveness of specialised care for early psychosis. Brit Med J 2004; 329:1067Crossref, Medline, Google Scholar
13 : Specialised care for early psychosis: symptoms, social functioning and patient satisfaction: randomised controlled trial. Br J Psychiatry 2006; 188:37–45; correction, 188:295Crossref, Medline, Google Scholar
14 : Integrated treatment ameliorates negative symptoms in first episode psychosis: results from the Danish OPUS trial. Schizophr Res 2005; 79:95–105Crossref, Medline, Google Scholar
15 : A randomised multicentre trial of integrated versus standard treatment for patients with a first episode of psychotic illness. Brit Med J. 2005; 331:602Crossref, Medline, Google Scholar
16 First-episode service for psychotic disorders in the U.S. public sector: a pragmatic, randomized control trial. Psychiatr Serv. 2015; 66:705–712Link, Google Scholar
17 : The RAISE early treatment program for first-episode psychosis: background, rationale, and study design. J Clin Psychiatry 2015; 76:240–246Crossref, Medline, Google Scholar
18 : Design and Analysis of Cluster Randomization Trials in Health Research, 1st ed. Chichester, UK, Wiley, 2010Google Scholar
19 : The NAVIGATE program for first-episode psychosis: rational, overview, and description of psychosocial components. Psychiatr Serv 2015; 66:680–690Link, Google Scholar
20 : Shared decision-making. Healthc Q 2009; 12:e186–e190Crossref, Medline, Google Scholar
21 : Applicability of telemedicine for assessing patients with schizophrenia: acceptance and reliability. J Clin Psychiatry 1997; 58:22–25Crossref, Medline, Google Scholar
22 : The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 1987; 13:261–276Crossref, Medline, Google Scholar
23 : Assessing depression in schizophrenia: the Calgary Depression Scale. Br J Psychiatry Suppl 1993; 163:39–44Crossref, Google Scholar
24 : The Quality of Life Scale: an instrument for rating the schizophrenic deficit syndrome. Schizophr Bull 1984; 10:388–398Crossref, Medline, Google Scholar
25 : Cost-effectiveness of second-generation antipsychotics and perphenazine in a randomized trial of treatment for chronic schizophrenia. Am J Psychiatry 2006; 163:2080–2089Link, Google Scholar
26 : Cost-effectiveness of supported housing for homeless persons with mental illness. Arch Gen Psychiatry 2003; 60:940–951Crossref, Medline, Google Scholar
27 : Searching for a consensus five-factor model of the Positive and Negative Syndrome Scale for schizophrenia. Schizophr Res 2012; 137:246–250Crossref, Medline, Google Scholar
28 : No adjustments are needed for multiple comparisons. Epidemiology 1990; 1:43–46Crossref, Medline, Google Scholar
29 : Statistical Power Analysis for the Behavioral Sciences, 2nd ed. New Jersey, Lawrence Erlbaum Associates, 1988Google Scholar
30 : Duration of untreated psychosis in community treatment settings in the United States. Psychiatr Serv 2015; 66:753–756 Link, Google Scholar
31 : Prescription practices in the treatment of first-episode schizophrenia spectrum disorders: data from the national RAISE-ETP study. Am J Psychiatry 2015; 172:237–248Link, Google Scholar
32 : Cardiometabolic risk in patients with first-episode schizophrenia spectrum disorders: baseline results from the RAISE-ETP study. JAMA Psychiatry 2014; 71:1350–1363Crossref, Medline, Google Scholar
33 : Defining the minimal clinically important difference (MCID) of the Heinrichs-carpenter quality of life scale (QLS). Int J Methods Psychiatr Res (Epub ahead of print, Aug 4, 2015)Google Scholar
34 : Economic grand rounds: financing first-episode psychosis services in the United States. Psychiatr Serv 2013; 64:506–508Link, Google Scholar
35 113th U.S. Congress (2013–2014): H.R. 3547: Consolidated Appropriations Act, 2014. https://www.congress.gov/bill/113th-congress/house-bill/3547Google Scholar
36 113th U.S. Congress (2013–2014): H.R. 83: Consolidated and Further Continuing Appropriations Act, 2015. https://www.congress.gov/bill/113th-congress/house-bill/83Google Scholar
37 : Effect of early intervention on 5-year outcome in non-affective psychosis. Br J Psychiatry 2010; 196:372–376Crossref, Medline, Google Scholar
38 : Two years of continued early treatment for recent-onset schizophrenia: a randomised controlled study. Acta Psychiatr Scand 2006; 114:328–336Crossref, Medline, Google Scholar
39 : Five-year follow-up of a randomized multicenter trial of intensive early intervention vs standard treatment for patients with a first episode of psychotic illness: the OPUS trial. Arch Gen Psychiatry 2008; 65:762–771Crossref, Medline, Google Scholar



