Screening, Diagnosis, and Treatment of Dyslipidemia Among Persons With Persistent Mental Illness: A Literature Review
Abstract
Objective:
Cardiovascular disease is the most frequent cause of death of persons with severe and persistent mental illness, and there is evidence of a widening mortality gap with the general population. Modifiable risk factors for cardiovascular disease, including dyslipidemia, are frequently underrecognized and undertreated. This review provides practitioners with an update on screening, diagnosis, and referral or treatment of dyslipidemia in this population.
Methods:
A literature search in PubMed from 1990 to 2012 that used various combinations of the terms cholesterol, screening, diagnosis, treatment, and severe mental illnesses identified 74 clinically relevant articles for review, and reference lists guided further exploration of sources. Additional material was selected with a focus on emerging guidelines to create clinically relevant recommendations for practitioners.
Results:
Multiple barriers can prevent clinicians from obtaining samples from fasting patients, which can be detrimental to successful screening. Dyslipidemia can be successfully screened for with nonfasting total cholesterol, high-density lipoprotein (HDL) cholesterol, and triglycerides, with follow-up measurement of fasting low-density lipoprotein (LDL) cholesterol if total cholesterol is greater than 200 mg/dl or triglycerides are above 500 mg/dl. Compelling evidence supports pharmacologic treatment of dyslipidemia to reduce cardiovascular events among high-risk patients.
Conclusions:
When obtaining samples from fasting patients is not feasible, use of samples from nonfasting patients can radically improve management of dyslipidemia among persons with severe and persistent mental illness. Common medications used to treat dyslipidemia are inexpensive, safe, and effective and could be more liberally employed to address comorbidities in this population. (Psychiatric Services 63:693–701, 2012; doi: 10.1176/appi.ps.201100475)
Cardiovascular disease remains the leading cause of death in the United States. One of the most widespread risk factors for cardiovascular disease is dyslipidemia, defined as elevated total cholesterol, elevated low-density lipoprotein (LDL), or low high-density lipoprotein (HDL); dyslipidemia affects up to 25% of adults in the general population (1). There are linear relationships between plasma levels of LDL, total cholesterol, and HDL (inverse relationship) and the development of cardiovascular disease and mortality (2,3). Repeated studies have shown that lowering total cholesterol levels of individuals at moderate to high risk of cardiovascular disease lowers mortality and morbidity rates (2,4).
Cardiovascular disease is a leading cause of excess morbidity among patients with severe and persistent mental illness (5–8). A widening gap in cardiovascular disease mortality from 1975 to 1995 has been found between this population and the general population (9,10). Although multiple factors probably contribute to excess cardiovascular disease among persons with severe and persistent mental illness, this review focused on dyslipidemia, which is highly prevalent, underrecognized, undertreated, and costly if not corrected (11,12).
The prevalence of dyslipidemia among persons with severe and persistent mental illness ranges from 25% to 70% (12,13). This rate is higher than that seen in age-matched persons without severe and persistent mental illness (13). The higher prevalence of dyslipidemia likely results from many factors, such as sedentary lifestyle, poor diet, socioeconomic status, obesity, and tobacco use. Also, the use of second-generation antipsychotics probably exacerbates this problem (10,14–17). In addition, for those with affective-spectrum illness, valproic acid and lithium use are associated with dyslipidemia (18,19).
Unfortunately, there is poor adherence to standard guidelines for screening and recognizing dyslipidemia (20–22). Data from the CATIE trial (Clinical Antipsychotic Trials of Intervention Effectiveness) showed that only 12% of adults and 5% of nonwhite females with recognized dyslipidemia were receiving treatment (13,23,24).
Barriers to screening and treatment of cardiovascular disease risk factors among persons with severe and persistent mental illness include access to medical care, reluctance of clients to seek medical treatment, and inadequate follow-up (24,25). Perhaps the most readily addressable barrier is lack of practitioner experience with screening and diagnosis of dyslipidemia. Many persons with severe and persistent mental illness receive medical services only in the mental health system, and their psychiatrist is frequently the only medical practitioner with whom they maintain regular contact (26,27).
This article seeks to provide mental health care providers with the knowledge to appropriately screen, diagnose, refer, and potentially begin treatment for dyslipidemia among persons with severe and persistent mental illness in the absence of adequate primary care consultation.
Methods
We searched MEDLINE and PubMed databases using the following keywords in the initial search: cholesterol AND screening AND schizophrenia. Further searches were conducted by substituting dyslipidemia for the search term cholesterol; diagnosis or treatment for the term screening, and bipolar disorder, bipolar affective disorder, treatment resistant depression and refractory depression, and combinations thereof for the term schizophrenia. No language restrictions were applied. Of 622 articles indexed between January 1, 1990, and January 28, 2012, 74 were selected as most clinically relevant, and their abstracts were reviewed. An additional 23 of the 74 were reviewed in depth. Reference lists from identified and related studies were used to identify additional studies and publications. When applicable, expert consensus guidelines and commentaries were also included to allow for the most simplified and practical approach to dyslipidemia for practicing clinicians.
Results
Lipid biology and role in atherosclerosis
Cholesterol is a waxy compound manufactured by the intestines and the liver, is an essential component of cell membranes of mammals, and is present in nearly all animal products consumed (eggs, meat, milk, and so forth). HMG CoA reductase inhibitors (“statins”) block the rate-limiting step of endogenous cholesterol production. Cholesterol is not water soluble and needs lipoproteins for transport to different tissues. Lipoproteins are traditionally identified by their relative density and (in ascending order) are noted to be chylomicrons, VLDL (very-low-density lipoprotein), IDL (intermediate-density lipoprotein), LDL, and HDL. Of these, LDL is the most prevalent and the most scrutinized for its role in atherosclerosis. However, other non-HDL lipoprotein particles also contribute to atherosclerosis risk, particularly among patients with high triglycerides. Indeed, LDL levels in such patients may be misleadingly low. Triglycerides are the primary components of dietary fats, transported in chylomicrons and VLDL, and are closely associated with abnormal glucose metabolism. As concentrations increase, triglycerides are degraded to lipoproteins of various densities containing cholesterol and have been weakly linked to cardiovascular disease.
Cholesterol-rich LDL and other particles (IDL and VLDL) that are not absorbed by body tissues for metabolism can enter the vascular wall (particularly if it is already damaged by smoking, hypertension, or diabetes). Cholesterol accumulates in arterial linings to eventually form atherosclerotic plaques. This process leads to the progressive narrowing of blood vessels, resulting in claudication or angina and eventually plaque rupture, clot formation, and myocardial infarction or stroke. Cholesterol can be removed from tissues through the actions of HDL (reverse cholesterol transport) and transported to the liver for processing and conversion to bile.
Measured total cholesterol is the sum of cholesterol present in all lipid particles, including chylomicrons. Total cholesterol, triglycerides, and HDL are measured directly in common laboratory tests. Because of the cost, LDL is usually calculated by using measures of the other three, or it can be directly measured. Total cholesterol and HDL vary little with respect to fasting status; however, triglycerides vary widely. VLDL is estimated to be one-fifth of total triglyceride concentration, as long as triglycerides are <400 mg/dl (28). The formula (equation 1) for approximating LDL concentration (known as the Friedewald formula) is as follows (TC is total cholesterol, cLDL is calculated low-density lipoprotein cholesterol, TG is triglyceride concentration, and HDL is high-density lipoprotein cholesterol):
cLDL ≈ TC − HDL − VLDL (1)
VLDL is almost equal to triglycerides divided by 5. Therefore, substituting VLDL for TG/5 allows approximation of LDL concentration (cLDL) with TC, HDL, and TG concentrations (equation 2):
cLDL ≈ TC − HDL − triglycerides/5 (2)
Because of this principle, LDL can be calculated from a fasting patient by subtracting HDL and triglycerides divided by 5 from the value for total cholesterol, which yields what is known as the cLDL (calculated LDL). At lower triglyceride concentrations, IDL and chylomicrons make up an exceedingly small fraction of total cholesterol and are not measured. Because of fluctuations in triglyceride levels, specimens from nonfasting patients cannot be used to reliably estimate cLDL (29).
Well-established studies have linked cLDL, total cholesterol, and HDL (inversely) to rates of cardiovascular disease mortality, independent of other risk factors (4). There is evidence that reducing cLDL, especially among adults with moderate to high risk of cardiovascular disease, results in significant reductions (up to 50%) in death or myocardial infarction after treatment (30).
Non-HDL cholesterol, defined as serum total cholesterol minus HDL, appears to represent the portion of total cholesterol most clearly related to overall cardiovascular disease risk. Measured or cLDL may not accurately represent cardiovascular disease risk for persons with even modestly elevated triglycerides and could lead to undertreatment. This is especially true for persons with metabolic syndrome or exposure to second-generation antipsychotics. Guidelines for the treatment of non-HDL cholesterol are achieved through the same diet, lifestyle, and pharmacotherapy options as for LDL therapy and may represent a better treatment target than cLDL values for persons with severe and persistent mental illness. Treatment targets for non-HDL cholesterol are typically set 30 mg/dl higher than LDL goals and may be a surrogate benchmark for nonfasting patients. Although current guidelines favor the use of cLDL targets in initiating and measuring therapy, the use of nonfasting non-HDL cholesterol as a therapy target may offset challenges in arranging for follow-up fasting specimens among persons with severe and persistent mental illness.
Screening among persons with persistent mental illness
For adults in the general population, screening for dyslipidemia every five years is recommended. Current guidelines from the U.S. Preventive Services Task Force recommend screening men who have an average risk of cardiovascular disease every five years beginning at age 35 and screening women who have an average risk beginning at age 45. Adults with elevated risk, as defined by either a cardiovascular disease risk-equivalent (a ten-year risk of about 20% for major myocardial infarction, stroke, or death; or the presence of diabetes, preexisting cardiovascular disease, abdominal aortic aneurysm, peripheral arterial disease, or carotid artery stenosis) or a major risk factor (hypertension, myocardial infarction for a male first-degree relative by age 55 or a female first-degree relative by age 65, smoking, and HDL<40 mg/dl), should be screened beginning at age 20 (class B recommendation: there is moderate to high certainty that the net benefit is moderate to substantial) (31). By some estimates, these criteria would include up to 60% of persons seen annually at community mental health centers (32,33), regardless of antipsychotic use. There is evidence of a markedly elevated risk of diabetes or cardiovascular disease independent of exposure to second-generation antipsychotics among persons with severe and persistent mental illness (34,35).
The consensus conference of the American Diabetes Association and the American Psychiatric Association recommended that patients who are taking second-generation antipsychotics undergo screening for dyslipidemia (36). [A table summarizing the recommendations is available online as a data supplement to this article.] Some experts now recommend cholesterol screening every six months for individuals taking second-generation antipsychotics after one year of treatment if baseline or follow-up serum cLDL or non-HDL concentration is greater than 130 mg/dl or 160 mg/dl, respectively (37,38).
Rarely is the triglyceride level greater than 500 mg/dl, at which point it is associated with an increased risk of pancreatitis. Combined with checking hemoglobin A1C as a diagnostic indicator of diabetes (39), screening for dyslipidemia with non-HDL cholesterol may now be offered to nonfasting clients without the inconvenience of their having to return later for a fasting measurement (40). Individuals who screen positive for dyslipidemia (total serum cholesterol >200 mg/dl for males and females, HDL <40 mg/dl for males and <50 mg/dl for females, or triglycerides >500 mg/dl for males and females) should undergo follow-up fasting measurement of triglycerides and cLDL to guide interventions and referrals. Direct or add-on LDL measurement is a possibility, but it has not been rigorously evaluated in large epidemiologic studies, and there is some variance in laboratory assay standards.
Diagnosis and therapeutic targets of dyslipidemia
Elevated total cholesterol and low HDL cholesterol.
The following recommendations are based largely on Adult Treatment Panel III (ATPIII) guidelines of the National Cholesterol Education Program (NCEP) (4), which were last revised in 2004. Total cholesterol values >240 mg/dl have consistently been recognized as high, and values >200 mg/dl generally indicate an intermediate risk that requires fasting LDL measurement or the use of non-HDL cholesterol as the treatment target. HDL measurements of <40 mg/dl for males or <50 mg/dl for females warrant intervention, whereas HDL values >60 mg/dl have been recognized to lower an individual's risk of cardiovascular disease.
Triglycerides and the metabolic syndrome.
Fasting triglycerides have been associated with cardiovascular disease and remain a component in the diagnosis of metabolic syndrome. [A table summarizing metabolic syndrome criteria is available online as a data supplement to this article.] However, fasting triglyceride levels may be of less value than total cholesterol, LDL, and low HDL concentrations in predicting development of cardiovascular disease (40,41). For this reason, the primary goal of therapy is reduction of cLDL and elevation of HDL, reserving dedicated treatment of triglycerides to concentrations >500 mg/dl. For fasting triglyceride levels between 150 mg/dl and 500 mg/dl, interventions are first targeted at lowering LDL and subsequently non-HDL cholesterol (total cholesterol minus HDL cholesterol). After appropriate non-HDL cholesterol targets are obtained, there is little evidence to support targeted drug treatment of triglyceride levels <500 mg/dl.
Elevated LDL.
NCEP ATPIII guidelines use an individual's cardiovascular risk and current cLDL levels to define treatment modalities and cLDL targets (4). Cardiovascular risk can be assessed as the number of major risk factors (as defined previously), with three levels defined as 0 to 1 (low), ≤2 (medium), or the presence of cardiovascular disease (high). Risk can also be calculated as ten-year risk of heart attack or death from cardiovascular disease on the basis of the Framingham Heart risk equation. To calculate an individual's Framingham Risk Score, online tools are readily available (hp2010.nhlbihin.net/atpiii/calculator.asp?usertype=prof), and tools are also embedded in electronic health records. The Framingham equation gives a risk score that can be divided into three different categories—0%–10% (low), 10%–20% (medium), and >20% (high)—and is useful only in guiding therapy targets for individuals without cardiovascular disease risk-equivalent but with two or more clinical risk factors. Certain diagnoses (mainly preexisting cardiovascular disease or diabetes mellitus) automatically place individuals in the high-risk (>20%) category, because persons with these diagnoses are considered to have the same or higher risk as those with existing known cardiovascular disease (a “risk-equivalent”).
Table 1 lists cLDL and non-HDL targets for the three clinical levels of risk. Each higher risk level lowers thresholds and goals by 30 mg/dl. For persons in the highest risk group (Framingham risk >20%, cardiovascular disease, or coronary risk-equivalent), thresholds for starting medications are cLDL >100 mg/dl or non-HDL >130 mg/dl after three months of failed intensive diet and exercise therapy.
Since the publication of the NCEP ATPIII guidelines, several trials have demonstrated the benefits of lowering LDL to <70 mg/dl in some instances. An update to the NCEP guidelines in 2004 suggested that patients with diabetes or cardiovascular disease may benefit from further lowering of cLDL (42). This would be particularly important in cases of a recent cardiovascular event (past year) or if diabetes or cardiovascular disease is combined with other, poorly controlled risk factors (that is, smoking or metabolic syndrome), as is often the case among persons with severe and persistent mental illness.
Because cardiovascular disease risk increases in a linear relationship with LDL concentration, individuals at high clinical risk who have relatively low LDL concentrations may still benefit from lipid-lowering pharmacotherapy. Some countries (Canada and the United Kingdom) have proposed guidelines for treatment based on total ten-year risk scores and clinical data regardless of LDL levels (43), in part to simplify screening and minimize costs. This emerging trend argues strongly for aggressive attention to dyslipidemia in the population with severe and persistent mental illness and favors a low threshold for treatment based on existing guidelines.
Treatment of dyslipidemia
Broadly defined, treatment of dyslipidemia is aimed at lowering LDL and non-HDL and raising HDL through therapeutic lifestyle changes, pharmacotherapy, or both.
Diet and lifestyle.
Diet and lifestyle interventions designed to reduce cardiovascular risk are often effective in reducing total cholesterol and LDL by up to 10%, but they usually involve resource-intensive (costly, labor-intensive) nutritional changes or exercise regimens that can be challenging to implement (44–50). Several factors may make these interventions particularly burdensome for persons with severe and persistent mental illness, including poor socioeconomic status, access to adequate facilities for safe exercise, concomitant medications that affect metabolism and sap energy, and lack of knowledge about nutrition and diet (38,51,52). Nevertheless, targeted recommendations and guidance from health professionals have been shown to assist persons with severe and persistent mental illness to lose weight and improve lipoprotein profiles (53–55), and they should be offered routinely.
Table 2 presents basic recommendations in the areas of diet and exercise that have been shown to improve cholesterol profiles. Routine dietary interventions involve the elimination of saturated and trans fats from the diet (56). Common sources of saturated fats include butter, cream, cheese, lard, coconut oil, palm oil, and chocolate. Common sources of trans fats include partially hydrogenated vegetable oils (most famously Crisco) that are commonly found in the fast-food industry in the production of fried foods and baked goods. Interventions that combine various components of exercise, dietary additives (such as omega-3 fatty acids and red-yeast extract), and fat reduction have been shown to be highly effective for some people, achieving up to a 35% reduction in LDL, which is similar to that achieved by therapy with some statins (56,57). More recently, newer approaches have been tested that include additional nutritional components that lower circulating LDL. These “portfolio” diets consist of a combination of soluble fiber, soy protein, tree nuts, and plant sterols (in margarine), have good adherence after just two dietary counseling sessions, and demonstrate robust lowering of cLDL compared with a diet of increased fiber and reduced saturated fat (reductions of 13% compared with 3% at six months) (58).
Exercise, defined as moderate aerobic exercise of around 120 minutes per week, has been shown to elevate HDL by up to 9% (approximately 3 mg/dl) and lower LDL, total cholesterol and triglycerides by up to 11%. Although modest increases in moderate-intensity exercise routines may not change LDL significantly, reports indicate that any structured physical activity may be beneficial beyond its cholesterol-lowering effects (52,59).
Medications.
Table 3 lists medications used to manage dyslipidemia and the relative effect on cholesterol lowering in each category, as well as side effects during treatment. Before initiation of pharmacotherapy for treatment of dyslipidemia, consideration should be given to switching to an alternative antipsychotic, such as aripiprazole or ziprasidone, if indicated (22,60,61). Results of switching studies should be scrutinized for the role of funding in outcomes and the relative paucity of data supporting use of first-generation antipsychotics.
Studies examining the role of adjunctive pharmacologic weight-loss therapies, including metformin, ramelteon, topiramate, and others, have been reviewed elsewhere and are generally focused on attenuation of weight gain (62–64). Some limited evidence exists for topiramate, metformin, and ramelteon in improving cholesterol profiles of persons treated with antipsychotics.
Generally, statins are the first-line agents for treatment of most dyslipidemias. This approach is supported by several trials demonstrating significant reduction in nonfatal myocardial infarction and cardiovascular events among white and nonwhite patients receiving statin treatment (65–69). Studies evaluating statin use systematically among persons with severe mental illness are limited but generally positive and have found rates of adherence to treatment and outcomes similar to those in the general population (70,71).
Liver transaminases should be monitored at the initiation of therapy at three months, but only annually thereafter in the presence of comorbid liver disease. With adequate supervision, statins have been used safely for persons with comorbid liver disease (hepatitis C, in the absence of alcohol consumption or end-stage liver disease) as long as total transaminase levels do not increase more than threefold above established patient baselines (72). Myalgias are a common side effect and may be underreported, occurring for up to 10% of those on statin therapy. Life-threatening rhabdomyolysis is a very rare occurrence (73). Individuals who experience myalgias should have their statin held and creatinine kinase (CK) levels checked. Those with CK elevations less than tenfold over normal and without evidence of rhabdomyolysis (myoglobinuria) may safely be rechallenged with pravastatin or fluvastatin, both considered to have less overall muscle toxicity.
Statin medications are pregnancy category X and should be prescribed to women of childbearing age only after appropriate informed consent. In addition, simvastatin coadministered with risperidone has been associated with rhabdomyolysis and should be used with caution by persons taking tricyclic antidepressants for risk of higher antidepressant levels and QTc prolongation (74). Although mood stabilizers are generally safe, carbamazepine may induce hepatic metabolism and lower the efficacy of many statins. Because many psychiatric medications are metabolized through the CYP450 enzyme system, pravastatin should be considered a first-line treatment for lower-risk patients who do not need substantial cLDL or non-HDL reductions. Pravastatin is relatively more hydrophilic and has a dual metabolism that does not depend solely on liver or kidney function (75).
Some statin medications are readily available as generics (atorvastatin, lovastatin, pravastatin, simvastatin), which makes cost less of a barrier to access. Dosages should be titrated as tolerated to achieve adequate reductions in cLDL or non-HDL. Current evidence suggests that high-dose simvastatin (80 mg daily) is associated with an unacceptable risk of myositis and is generally avoided in practice (76); however, lower doses seem safe. High-dose rosuvastatin has been found to cause impaired renal function and may contribute in small amounts to the development of diabetes. Because of this, creatinine and hemoglobin A1C should be monitored at least yearly among patients taking this drug.
Because the majority of hepatic cholesterol synthesis occurs in the nighttime during fasting, statins with short half-lives (simvastatin, lovastatin, and pravastatin) should be administered in the evening. Certain statins with long half-lives (atorvastatin and rosuvastatin) may be dosed variably throughout the day without reduction in efficacy (77). If patients do not achieve cLDL goals with appropriately titrated statin treatment, a switch to a more potent statin, such as atorvastatin or rosuvastatin, may be recommended.
Treatment of hypertriglyceridemia (>500 mg/dl) and mixed dyslipidemia starts with a statin alone. Statin therapy lowers triglyceride levels among patients with schizophrenia (70). If triglyceride levels remain substantially abnormal (>500 mg/dl) with statin treatment, additional drugs may be used, such as fish oil, niacin, or fibrates in order of preference. When added to a statin, fenofibrate is the fibrate of choice, primarily because of increased risk of myositis with gemfibrozil. A specialist should manage augmentation strategies for lipid reduction, and referral is indicated if monotherapy is insufficient to improve the cholesterol profile.
Figure 1 presents a summary flowchart of screening and treatment guidelines. Persons with dyslipidemia who are on cholesterol-lowering therapy should have their cholesterol checked at least yearly, once stabilized, to demonstrate continued treatment success.
Discussion and conclusions
Efforts should be made to aggressively reduce all cardiovascular disease risk factors among persons with severe and persistent mental illness, including dyslipidemia. Smoking cessation is also of paramount importance. Unfortunately, smoking cessation is often associated with weight gain, which could adversely affect lipoprotein profiles and may necessitate closer monitoring.
Efforts to reduce cardiovascular disease risk must continually be viewed in light of a holistic strategy for achieving individualized health and well-being. Numerous interventions have demonstrated improved health outcomes and consistent, sustainable reduction in cardiovascular disease risk, specifically among persons with severe and persistent mental illness (4,78–81). Achieving desirable cholesterol goals should be a high priority in cardiovascular disease risk reduction because of a clearly defined evidence base, simple screening tests, and the availability of cheap, effective, and safe interventions.
1 : Serum total cholesterol concentrations and awareness, treatment, and control of hypercholesterolemia among US adults: findings from the National Health and Nutrition Examination Survey, 1999 to 2000. Circulation 107:2185–2189, 2003 Crossref, Medline, Google Scholar
2 : The cholesterol facts: a summary of the evidence relating dietary fats, serum cholesterol, and coronary heart disease. Circulation 81:1721–1733, 1990 Crossref, Medline, Google Scholar
3 : Serum cholesterol, lipoproteins, and the risk of coronary heart disease. Annals of Internal Medicine 74:1–12, 1971 Crossref, Medline, Google Scholar
4 Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 285:2486–2497, 2001 Crossref, Medline, Google Scholar
5 : The association between mood and anxiety disorders with vascular diseases and risk factors in a nationally representative sample. Journal of Psychosomatic Research 70:145–154, 2011 Crossref, Medline, Google Scholar
6 : Congruencies in increased mortality rates, years of potential life lost, and causes of death among public mental health clients in eight states. Preventing Chronic Disease 3(2):A42; Epub March 15, 2006 Medline, Google Scholar
7 : Mortality and medical comorbidity among patients with serious mental illness. Psychiatric Service 57:1482–1487, 2006 Link, Google Scholar
8 : Integrating Behavioral Health and Primary Care Services? Opportunities and Challenges for State Mental Health Authorities. Technical Report. Alexandria, Va, National Association of State Mental Health Program Directors, Medical Directors Council, 2005 Google Scholar
9 : Time trends in schizophrenia mortality in Stockholm County, Sweden. BMJ 321:483–484, 2000 Crossref, Medline, Google Scholar
10 : A systematic review of mortality in schizophrenia: is the differential mortality gap worsening over time? Archives of General Psychiatry 64:1123–1131, 2007 Crossref, Medline, Google Scholar
11 : Healthcare spending among community-dwelling adults with schizophrenia. American Journal of Managed Care 11(suppl 8):S242–S247, 2005 Medline, Google Scholar
12 : Prevalence of the metabolic syndrome in patients with schizophrenia treated with antipsychotic medication. Schizophrenia Research 83:87–93, 2006 Crossref, Medline, Google Scholar
13 : Low rates of treatment for hypertension, dyslipidemia and diabetes in schizophrenia: data from the CATIE schizophrenia trial sample at baseline. Schizophrenia Research 86:15–22, 2006 Crossref, Medline, Google Scholar
14 : A 4-fold risk of metabolic syndrome in patients with schizophrenia: the Northern Finland 1966 Birth Cohort study. Journal of Clinical Psychiatry 66:559–563, 2005 Crossref, Medline, Google Scholar
15 : The unhealthy lifestyle of people with schizophrenia. Psychological Medicine 29:697–701, 1999 Crossref, Medline, Google Scholar
16 : Cigarette smoking and overweight/obesity among individuals with serious mental illnesses: a preventive perspective. Harvard Review of Psychiatry 14:212–222, 2006 Crossref, Medline, Google Scholar
17 : The effects of antipsychotic therapy on serum lipids: a comprehensive review. Schizophrenia Research 70:1–17, 2004 Crossref, Medline, Google Scholar
18 : A 12-month randomized, open-label study of the metabolic effects of olanzapine and risperidone in psychotic patients: influence of valproic acid augmentation. Journal of Clinical Psychiatry 72:1602–1610, 2011 Crossref, Medline, Google Scholar
19 : Treatment with lithium, alone or in combination with olanzapine, relieves oxidative stress but increases atherogenic lipids in bipolar disorder. Tohoku Journal of Experimental Medicine 213:79–87, 2007 Crossref, Medline, Google Scholar
20 : Metabolic screening in children receiving antipsychotic drug treatment. Archives of Pediatrics and Adolescent Medicine 164:344–351, 2010 Crossref, Medline, Google Scholar
21 : Metabolic considerations in the use of antipsychotic medications: a review of recent evidence. Journal of Clinical Psychiatry 68(suppl 1):20–27, 2007 Crossref, Medline, Google Scholar
22 : A multicenter, randomized, double-blind study of the effects of aripiprazole in overweight subjects with schizophrenia or schizoaffective disorder switched from olanzapine. Journal of Clinical Psychiatry 69:1046–1056, 2008 Crossref, Medline, Google Scholar
23 : The Atypical Antipsychotic Therapy and Metabolic Issues National Survey: practice patterns and knowledge of psychiatrists. Journal of Clinical Psychopharmacology 24(5 suppl 1):S1–S6, 2004 Crossref, Medline, Google Scholar
24 : Quality of preventive medical care for patients with mental disorders. Medical Care 40:129–136, 2002 Crossref, Medline, Google Scholar
25 : Health care for patients with serious mental illness: family medicine's role. Journal of the American Board of Family Medicine 22:187–195, 2009 Crossref, Medline, Google Scholar
26 : From silos to bridges: meeting the general health care needs of adults with severe mental illnesses. Health Affairs 25:659–669, 2006 Crossref, Medline, Google Scholar
27 : Specialty care medical homes for people with severe, persistent mental disorders. Health Affairs 29:867–873, 2010 Crossref, Medline, Google Scholar
28 : Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clinical Chemistry 18:499–502, 1972 Medline, Google Scholar
29 : Reliability of low-density lipoprotein cholesterol, non-high-density lipoprotein cholesterol, and apolipoprotein B measurement. Journal of Clinical Lipidology 5:264–272, 2011 Crossref, Medline, Google Scholar
30 : Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 366:1267–1278, 2005 Crossref, Medline, Google Scholar
31 Screening for Lipid Disorders in Adults. Rockville, Md, US Preventive Services Task Force, 2008. Available at www.uspreventiveservicestaskforce.org/uspstf/uspschol.htm Google Scholar
32 : Association between tobacco smoking and bipolar affective disorder: clinical, epidemiological, cross-sectional, retrospective study in outpatients. Comprehensive Psychiatry, PMID:
33 : Findings of a US national cardiometabolic screening program among 10,084 psychiatric outpatients. Psychiatric Services 61:892–898, 2010 Link, Google Scholar
34 : Elevated prevalence of obesity, metabolic syndrome, and cardiovascular risk factors in bipolar disorder. Annals of Clinical Psychiatry; 20:131–137, 2008 Crossref, Medline, Google Scholar
35 : The distribution of body mass index among individuals with and without schizophrenia. Journal of Clinical Psychiatry 60:215–220, 1999 Crossref, Medline, Google Scholar
36 Consensus Development Conference on Antipsychotic Drugs and Obesity and Diabetes. Diabetes Care 27:596–601, 2004 Crossref, Medline, Google Scholar
37 : Implementation of monitoring and management guidelines for second-generation antipsychotics. Journal of Clinical Psychiatry 68(suppl 4):14–18, 2007 Crossref, Medline, Google Scholar
38 : Physical health monitoring of patients with schizophrenia. American Journal of Psychiatry 161:1334–1349, 2004 Link, Google Scholar
39 Diagnosis and classification of diabetes mellitus. Diabetes Care 33(suppl 1):S62–S69, 2010 Crossref, Medline, Google Scholar
40 : Major lipids, apolipoproteins, and risk of vascular disease. JAMA 302:1993–2000, 2009 Crossref, Medline, Google Scholar
41 : Fasting compared with nonfasting lipids and apolipoproteins for predicting incident cardiovascular events. Circulation 118:993–1001, 2008 Crossref, Medline, Google Scholar
42 : Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 110:227–239, 2004 Crossref, Medline, Google Scholar
43 : 2009 Canadian Cardiovascular Society/Canadian guidelines for the diagnosis and treatment of dyslipidemia and prevention of cardiovascular disease in the adult: 2009 recommendations. Canadian Journal of Cardiology 25:567–579, 2009 Crossref, Medline, Google Scholar
44 : Reduced or modified dietary fat for preventing cardiovascular disease. Cochrane Database of Systematic Reviews 3:CD002137, 2001 Medline, Google Scholar
45 : Lifestyle approaches and dietary strategies to lower LDL-cholesterol and triglycerides and raise LDL-cholesterol. Endocrinology and Metabolism Clinics of North America; 38:45–78, 2009 Crossref, Medline, Google Scholar
46 : The evidence for dietary prevention and treatment of cardiovascular disease. Journal of the American Dietetic Association 108:287–331, 2008 Crossref, Medline, Google Scholar
47 : In the statin era, how important are intense lifestyle changes? Journal of the American College of Cardiology 41:273–274, 2003 Crossref, Medline, Google Scholar
48 : Chinese red yeast rice (Monascus purpureus) for primary hyperlipidemia: a meta-analysis of randomized controlled trials. Chinese Medicine 1:4, , 2006; doi:
49 : Cardiovascular benefits of omega-3 fatty acids. Cardiovascular Research 73:310–315, 2007 Crossref, Medline, Google Scholar
50
51 : Cardiovascular disease and diabetes in people with severe mental illness position statement from the European Psychiatric Association (EPA), supported by the European Association for the Study of Diabetes (EASD) and the European Society of Cardiology. European Psychiatry 24:412–424, 2009 Crossref, Medline, Google Scholar
52 : Physical activity patterns in adults with severe mental illness. Journal of Nervous and Mental Disease 193:641–646, 2005 Crossref, Medline, Google Scholar
53 : A pilot evaluation of the In SHAPE individualized health promotion intervention for adults with mental illness. Community Mental Health Journal 46:540–552, 2010 Crossref, Medline, Google Scholar
54 : Lifestyle interventions for adults with serious mental illness: a systematic literature review. Psychiatric Services 61:774–782, 2010 Link, Google Scholar
55 : Non-pharmacological management of antipsychotic-induced weight gain: systematic review and meta-analysis of randomised controlled trials. British Journal of Psychiatry 193:101–107, 2008 Crossref, Medline, Google Scholar
56 : Diet and exercise in the management of hyperlipidemia. American Family Physician 81:1097–1102, 2010 Medline, Google Scholar
57 : Diet and cholesterol reduction. Annals of Internal Medicine 142:793–795, 2005 Crossref, Medline, Google Scholar
58 : Effect of a dietary portfolio of cholesterol-lowering foods given at 2 levels of intensity of dietary advice on serum lipids in hyperlipidemia. JAMA 306:831–839, 2011 Crossref, Medline, Google Scholar
59 : Prevention of cardiovascular disease in developing countries. Lancet 370:720–722, 2007 Crossref, Medline, Google Scholar
60 : Switching antipsychotics as a treatment strategy for antipsychotic-induced weight gain and dyslipidemia. Journal of Clinical Psychiatry 68(suppl 4):34–39, 2007 Medline, Google Scholar
61 : A randomized trial examining the effectiveness of switching from olanzapine, quetiapine, or risperidone to aripiprazole to reduce metabolic risk: comparison of antipsychotics for metabolic problems. American Journal of Psychiatry 168:947–956, 2011 Link, Google Scholar
62 : Placebo-controlled pilot study of ramelteon for adiposity and lipids in patients with schizophrenia. Journal of Clinical Psychopharmacology 31:653–658, 2011 Medline, Google Scholar
63 : Topiramate for prevention of olanzapine associated weight gain and metabolic dysfunction in schizophrenia: a double-blind, placebo-controlled trial. Schizophrenia Research 118:218–223, 2010 Crossref, Medline, Google Scholar
64 : Systematic review and meta-analysis of pharmacological interventions for weight gain from antipsychotics and mood stabilizers. Current Psychiatry Reviews 8:25–36, 2012 Crossref, Medline, Google Scholar
65 : Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366:1849–1861, 2005 Crossref, Medline, Google Scholar
66 : Race, ethnicity, and the efficacy of rosuvastatin in primary prevention: the Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER) trial. American Heart Journal 162:106–114.e2; Epub June 12, 2011 Crossref, Medline, Google Scholar
67 : Relation of different measures of low-density lipoprotein cholesterol to risk of coronary artery disease and death in a meta-regression analysis of large-scale trials of statin therapy. American Journal of Cardiology 105:1289–1296, 2010 Crossref, Medline, Google Scholar
68 : After the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study: implications for fenofibrate. American Journal of Cardiology 102(12A):34L–40L, 2008 Crossref, Medline, Google Scholar
69 : Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm (ASCOT-LLA): a multicentre randomised controlled trial. Lancet 361:1149–1158, 2003 Crossref, Medline, Google Scholar
70 : Treatment with rosuvastatin for severe dyslipidemia in patients with schizophrenia and schizoaffective disorder. Journal of Clinical Psychiatry 67:1889–1896, 2006 Crossref, Medline, Google Scholar
71 : Adherence to antihyperlipidemic medication and lipid control in diabetic Veterans Affairs patients with psychotic disorders. Psychosomatics 52:310–318, 2011 Crossref, Medline, Google Scholar
72 : Use of statins in patients with chronic hepatitis C. Southern Medical Journal 103:1018–1022, 2010 Crossref, Medline, Google Scholar
73 : Long-term safety and efficacy profile of simvastatin. American journal of Cardiology 68:1127–1131, 1991 Crossref, Medline, Google Scholar
74 : Rhabdomyolysis and compartment syndrome with co-administration of risperidone and simvastatin. Journal of Psychopharmacology 18:432–434, 2004 Crossref, Medline, Google Scholar
75 : Clinical pharmacokinetics of pravastatin mechanisms of pharmacokinetic events. Clinical Pharmacokinetics 39:397–412, 2000 Crossref, Medline, Google Scholar
76
77 : Pharmacodynamic effects and pharmacokinetics of atorvastatin after administration to normocholesterolemic subjects in the morning and evening. Journal of Clinical Pharmacology 36:604–609, 1996 Crossref, Medline, Google Scholar
78 : Successful tobacco dependence treatment in schizophrenia. American Journal of Psychiatry 164:222–227, 2007 Link, Google Scholar
79 : Effectiveness of medications used to attenuate antipsychotic-related weight gain and metabolic abnormalities: a systematic review and meta-analysis. Neuropsychopharmacology 35:1520–1530, 2010 Crossref, Medline, Google Scholar
80 : Integrated medical care for patients with serious psychiatric illness: a randomized trial. Archives of General Psychiatry 58:861–868, 2001 Crossref, Medline, Google Scholar
81 : Effects of a 10-week weight control program on obese patients with schizophrenia or schizoaffective disorder: a 12-month follow up. Psychiatry and Clinical Neurosciences 63:17–22, 2009 Crossref, Medline, Google Scholar
Figures and Tables
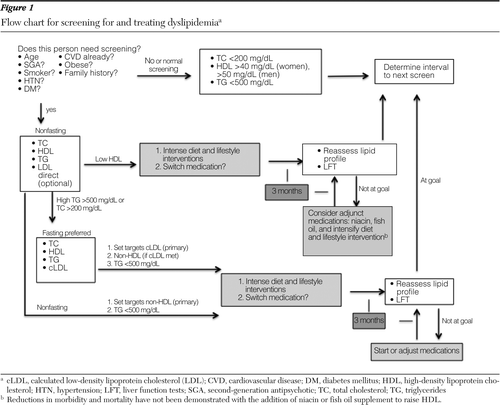
Figure 1 Flow chart for screening for and treating dyslipidemia
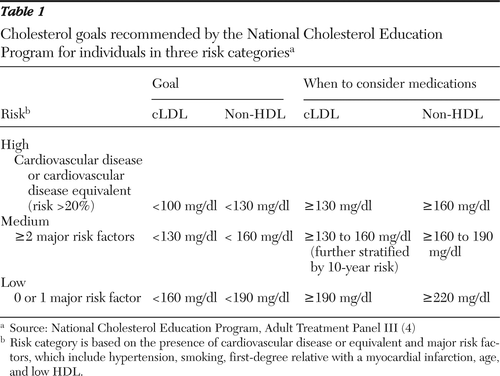
Table 1 Cholesterol goals recommended by the National Cholesterol Education Program for individuals in three risk categories
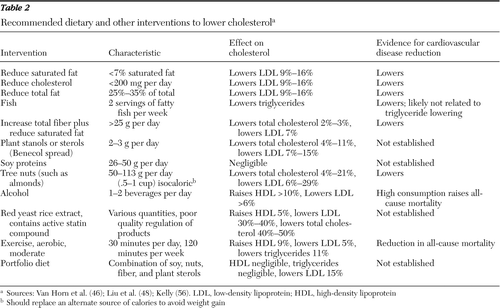
Table 2 Recommended dietary and other interventions to lower cholesterol
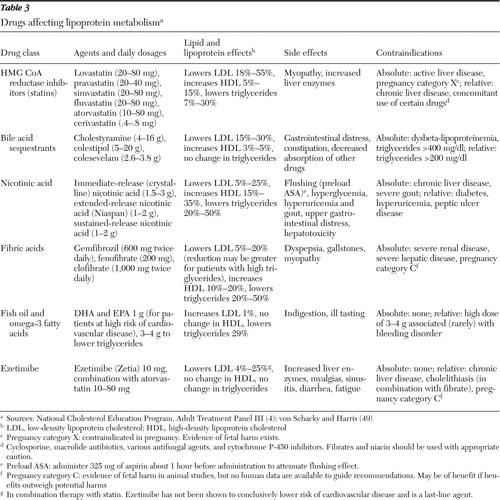
Table 3 Drugs affecting lipoprotein metabolism



