Adapting Smoking Cessation Treatment According to Initial Response to Precessation Nicotine Patch
Abstract
Objective
The authors evaluated an adaptive smoking cessation treatment strategy in which nicotine patch treatment was initiated before a quit date, and then, depending on initial therapeutic response, either the nicotine patch was continued or alternative pharmacotherapies were provided.
Method
The study was a double-blind, parallel-arm adaptive treatment trial. A total of 606 cigarette smokers started open-label nicotine patch treatment 2 weeks before the quit date. Those whose ad lib smoking did not decrease by >50% after 1 week were randomly assigned to one of three double-blind treatments: nicotine patch alone (control condition); “rescue” treatment with bupropion augmentation of the patch; or rescue treatment with varenicline alone. Participants whose precessation smoking decreased >50% but who lapsed after the quit date were also randomly assigned to the two rescue treatments or to nicotine patch alone. Logistic regression analyses compared each rescue treatment against the control condition in terms of abstinence at the end of treatment (weeks 8–11) and at 6 months.
Results
Smokers who did not respond adequately to precessation nicotine patch benefited from bupropion augmentation; abstinence rates at end of treatment were 16% with nicotine patch alone and 28% with bupropion augmentation (odds ratio=2.04, 95% CI=1.03–4.01). Switching to varenicline produced less robust effects, but point abstinence at 6 months was 6.6% with the patch alone and 16.5% with a switch to varenicline (odds ratio=2.80, 95% CI=1.11–7.06). Postquit adaptive changes in treatment had no significant effects on any abstinence outcome.
Conclusions
It is possible to rescue a significant portion of smokers who would have failed to achieve abstinence if left on nicotine patch alone by identifying these smokers before their quit date and implementing adaptive changes in treatment.
Little information is available to guide health care providers when recommending which smoking cessation medication to use for a given smoker. While some algorithms have been suggested for smoking cessation treatment (1, 2), there is a paucity of experimental data validating these approaches.
Nicotine replacement therapy is widely used and, because of its safety and tolerability, has been available for many years as an over-the-counter treatment (3). Two prescription drugs, bupropion and varenicline, have also achieved widespread use (4–7).
To assess early therapeutic response to nicotine replacement therapy before the quit date, we monitored ad lib smoking during the first week of precessation open-label nicotine patch treatment by measuring expired-air CO levels. In previous studies, we observed that a reduction in ad lib smoking while on precessation nicotine patch treatment strongly predicted abstinence after the quit date (12–14). Our primary hypothesis was that smokers who did not respond to nicotine patch alone would achieve higher abstinence rates if they were switched to either of two alternative therapies (see phase 1 below). A secondary, exploratory hypothesis was that smokers who responded well initially to precessation nicotine patch treatment but who lapsed in the first week after their target quit date could be “rescued” by switching to alternative treatments at that point and setting a second quit date 1 week later (see phase 2 below). In contrast, for smokers who initially responded with sufficient smoking reduction with nicotine patch treatment, it was reasonable for them to remain on nicotine patch alone and not be exposed to the greater risks and side effects of the available prescription medications.
Method
Study Design
The study was a double-blind, parallel-arm adaptive treatment trial comprising two distinct phases (Figure 1).
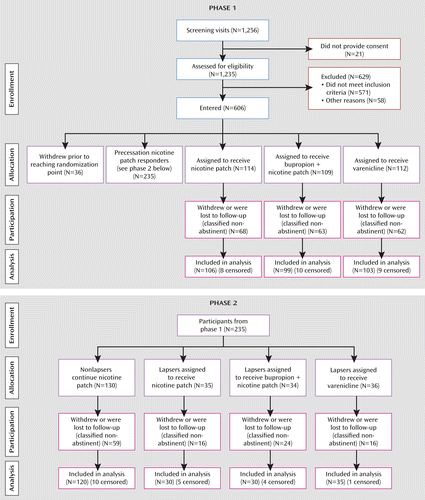
a In phase 1, cigarette smokers received precessation nicotine patch treatment, and those whose ad lib smoking did not decrease by >50% after 1 week were randomly assigned to remain on nicotine patch treatment, switch to varenicline, or augment nicotine patch with bupropion. In phase 2, participants whose precessation smoking decreased >50% but who lapsed after the quit date were also randomly assigned to the two rescue treatments or to nicotine patch alone.
Phase 1 assessed the effects of two potential precessation rescue treatments (varenicline or bupropion augmentation of nicotine patch) for smokers who did not respond adequately to open-label nicotine patch treatment before the target quit date. Smokers were classified as precessation “responders” or “nonresponders” to nicotine patch treatment, depending on whether their ad lib smoking (assessed by expired-air CO levels) decreased by more than 50% in the first week of precessation patch treatment. Nonresponders at that point (1 week before their target quit date) were randomly assigned to one of three double-blind treatment conditions: 1) a switch to varenicline (stopping nicotine patch treatment); 2) augmentation of nicotine patch with bupropion; and 3) continuation on nicotine patch alone. Varenicline was administered as a monotherapy because current labeling recommends against combining nicotine patch and varenicline because of an increased frequency of nausea and other side effects. Nicotine patch responders remained on open-label nicotine patches until Phase 2.
Phase 2 assessed the effects of postquit adaptive treatment changes for precessation nicotine patch responders who lapsed in the first week after the quit date. Lapsers were identified either by self-report of smoking (even a puff) within the first week after the quit date or by an expired-air CO level >10 ppm at 1 week after the quit date. These lapsers were scheduled for a second target quit date 1 week later and were randomly assigned to the same three double-blind treatment conditions as in phase 1. Nonlapsers remained on open-label nicotine patches for the duration of treatment.
Recruitment, Eligibility, and Compensation
Adult smokers expressing a desire to quit smoking were recruited through newspaper, radio, and television advertisements. To be eligible, participants had to be 18–65 years of age, report smoking an average of ≥10 cigarettes per day for 3 cumulative years, have an expired-air CO level ≥10 ppm, and have no exclusionary feature on history, physical examination, or laboratory evaluation (see the data supplement that accompanies the online edition of this article). After receiving a complete description of the study, participants provided written informed consent. Participants were compensated up to $320 for study participation.
Study Procedures
After screening and enrollment in the study, participants were seen weekly at our research center for 2 weeks before the quit date and attended four to six sessions after the quit date. Brief (<15 minutes) support was provided at each session, and clinical trial materials were dispensed. Smoking diaries and measures of expired-air CO levels, withdrawal symptoms, and other adverse effects were also collected. Six months after the target quit date, participants were contacted and those who reported point (7-day) abstinence were invited to return to the center for an assessment of expired-air CO level.
The study was conducted in a double-blind and double-dummy fashion; all participants who were assigned to a treatment condition received active nicotine skin patches (NicoDerm CQ, GlaxoSmithKline, Philadelphia) or placebo (Rejuvenation Laboratories, Salt Lake City), capsules containing varenicline (Pfizer, New York) or placebo, and bupropion sustained-release tablets (GlaxoSmithKline) or placebo (Glatt Pharmaceutical Services, Ramsey, N.J.). The recommended dosing titration schedule was used for both varenicline (0.5 mg once daily on days 1–3, 0.5 mg twice daily on days 4–7, and 1 mg twice daily through week 12) and bupropion (150 mg daily for 3 days, followed by 150 mg twice daily through week 12).
Initial nicotine patch dosing was based on initial expired-air CO reading; participants with CO levels >30 ppm at baseline received 42 mg/day (two 21 mg/day patches) daily, and the remaining participants received a single 21 mg/day patch daily. In the 21 mg/day condition, an active patch was applied each morning; in the 42 mg/day condition, an active 21 mg patch was applied each morning and a second patch at noon. This personalized dosing regimen was based on previous research suggesting that heavy smokers may not receive adequate nicotine replacement with a 21 mg patch (15, 16). Dose reductions were allowed in the event of side effects.
Statistical Analysis
For each of the two phases of the study, treatment groups were initially compared on demographic and smoking history variables, in order to identify potential confounding factors (none were identified).
To evaluate the main hypothesis of phase 1—that precessation adaptive treatment changes would rescue precessation nicotine patch nonresponders—abstinence outcomes were compared between each potential rescue treatment and the corresponding randomized nicotine patch group.
To evaluate the main hypothesis of phase 2—that postcessation adaptive treatment changes would rescue participants who lapsed in the first week after their quit date—abstinence outcomes were compared between each potential rescue treatment and the corresponding randomized nicotine patch group.
Logistic regression analyses were used to evaluate the primary outcome of continuous abstinence at end of treatment (abstinence during weeks 8–11 after the target quit date). Secondary outcomes included continuous 11 weeks of abstinence from the quit date, point (7-day) abstinence at 6 months, and continuous abstinence from the quit date at 6-months. Abstinence was identified by self-report of no smoking confirmed by end-expired CO levels ≤10 ppm. Participants who withdrew from the study or were lost to follow-up were classified as nonabstinent.
In addition to the above analyses, we sought confirmatory evidence in support of the two main assumptions underlying the adaptive treatment approach. In phase 1, to verify the assumption that precessation nicotine patch nonresponders would do poorly if left on nicotine patch alone, we compared the abstinence rate of precessation nicotine patch responders to that of nicotine patch nonresponders who were randomly assigned to remain on nicotine patch alone. Given that nicotine patch responders who lapsed after the quit date may have been assigned to treatments other than nicotine patch in phase 2, we eliminated the confounding effects of these postquit treatment changes by taking the outcome to be continuous abstinence from the initial quit date through end of treatment (11 weeks after the quit date). By using the continuous-abstinence endpoint, lapsers were considered nonabstinent before any potential confounding effects of postquit treatment changes could occur.
The second main assumption, in phase 2, was that participants who lapsed in the first week after the quit date who were assigned to nicotine patch alone would have a lower abstinence rate than participants on nicotine patch alone who had no lapses. To evaluate this assumption, abstinence outcome (4-week continuous abstinence at the end of treatment) was compared between lapsers and nonlapsers receiving nicotine patch alone in phase 2.
Forty-seven participants were excluded from data analyses (27 in phase 1 and 20 in phase 2) because of having initiated contraindicated medications (opiates, antidepressants, or other CNS-active medications) during the study or failing to meet other entry criteria. Another individual died before reaching the primary outcome point and was also censored from analysis. One participant was excluded from the analysis of percent change in CO level because of a value >35 standard deviations from the mean of the remaining sample.
Results
A total of 606 participants were enrolled in the study (see Figure 1). The demographic characteristics and smoking histories of participants were similar across treatment conditions for both phases (Table 1).
| Study Phase and Characteristic | Pre- and Postcessation Study Conditions | |||||||
|---|---|---|---|---|---|---|---|---|
| Phase 1 | Randomized, Double-Blind Precessation Conditions | |||||||
| Nicotine Patch (N=106) | Bupropion Plus Nicotine Patch (N=99) | Varenicline (N=103) | ||||||
| N | % | N | % | N | % | |||
| Male | 51 | 48.1 | 53 | 53.5 | 62 | 60.2 | ||
| Race/ethnicity | ||||||||
| European American | 67 | 63.2 | 61 | 61.6 | 66 | 64.1 | ||
| African American | 34 | 32.1 | 27 | 27.3 | 34 | 33.0 | ||
| Other | 5 | 4.7 | 11 | 11.1 | 3 | 2.9 | ||
| 21 mg nicotine patch | 70 | 66.0 | 64 | 64.6 | 69 | 66.9 | ||
| Menthol cigarette smoker | 55 | 51.9 | 49 | 49.5 | 52 | 50.5 | ||
| Mean | SD | Mean | SD | Mean | SD | |||
| Cigarettes/day | 21.3 | 8.9 | 21.9 | 8.8 | 21.8 | 10.6 | ||
| Age (years) | 44.3 | 10.8 | 46.0 | 10.8 | 44.7 | 10.7 | ||
| Years smoked | 26.0 | 11.2 | 26.8 | 10.6 | 25.9 | 11.0 | ||
| Fagerström Test for Nicotine Dependence score | 5.8 | 2.0 | 5.8 | 2.0 | 5.8 | 1.7 | ||
| Expired-air CO (ppm) | 28.6 | 14.1 | 28.5 | 11.6 | 28.9 | 11.5 | ||
| Cotinine (ng/mL) | 346 | 214 | 366 | 189 | 359 | 218 | ||
| Phase 2 | Randomized, Double-Blind Postcessation Conditions | |||||||
| Nicotine Patch (N=30) | Bupropion Plus Nicotine Patch (N=30) | Varenicline (N=35) | Open-Label Nicotine Patch (N=120) | |||||
| N | % | N | % | N | % | N | % | |
| Male | 11 | 36.7 | 15 | 50.0 | 18 | 51.4 | 60 | 50.0 |
| Race/ethnicity | ||||||||
| European American | 18 | 60.0 | 20 | 66.7 | 21 | 60.0 | 89 | 74.2 |
| African American | 11 | 36.7 | 8 | 26.7 | 14 | 40.0 | 25 | 20.8 |
| Other | 1 | 3.3 | 2 | 6.7 | 0 | 0.0 | 6 | 5.0 |
| 21 mg nicotine patch | 14 | 46.7 | 13 | 43.3 | 15 | 42.9 | 57 | 47.5 |
| Menthol cigarette smoker | 16 | 53.3 | 12 | 40.0 | 16 | 45.7 | 50 | 41.7 |
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Cigarettes/day | 19.1 | 9.2 | 22.9 | 10.5 | 20.5 | 8.2 | 20.8 | 7.4 |
| Age (years) | 40.8 | 12.8 | 43.1 | 11.6 | 44.1 | 11.2 | 43.5 | 10.1 |
| Years smoked | 22.7 | 12.0 | 24.7 | 11.4 | 24.6 | 10.5 | 24.6 | 10.3 |
| Fagerström Test for Nicotine Dependence score | 5.7 | 1.9 | 6.3 | 2.0 | 5.9 | 2.3 | 5.6 | 1.6 |
| Expired-air CO (ppm) | 26.8 | 13.4 | 28.9 | 8.6 | 27.9 | 12.7 | 30.2 | 12.4 |
| Cotinine (ng/mL) | 334 | 189 | 369 | 237 | 339 | 235 | 384 | 214 |
Phase 1: Evaluation of Precessation Rescue Effects
| Randomized Double-Blind Conditions | |||||||
|---|---|---|---|---|---|---|---|
| Phase 1 (Precessation Nonresponders) | Phase 2 (Postcessation Lapsers) | ||||||
| Abstinence Outcome | Nicotine Patch (N=106) | Bupropion Plus Nicotine Patch (N=99) | Varenicline (N=108) | Nicotine Patch (N=30) | Bupropion Plus Nicotine Patch (N=30) | Varenicline (N=35) | Open-label Nicotine Patch (N=120) |
| Weeks 8–11 | 16.0 | 28.3a | 23.3 | 26.7 | 26.7 | 37.1 | 59.2 |
| Quit date to week 11 | 6.6 | 19.2a | 11.7 | 23.3 | 20.0 | 28.6 | 51.7 |
| Quit date to 6 months | 5.8 | 13.1a | 5.8 | 10.0 | 10.0 | 14.3 | 20.0 |
| Point abstinence at 6 months | 6.6 | 17.2a | 16.5a | 13.3 | 10.0 | 20.0 | 21.7 |
The precessation adaptive change in treatment to varenicline did not yield a significant effect on the primary outcome of continuous abstinence during weeks 8–11 or on the measures of continuous abstinence from the quit date, but there was a significant effect on point abstinence at 6 months (p=0.03; odds ratio=2.80, 95% CI=1.11–7.06). To examine whether the emergence of an effect of varenicline at 6 months may have been due to smokers’ recovery from early lapses, a follow-up analysis compared 6-month point abstinence across conditions among smokers who lapsed at least once during the last 4 weeks of treatment (weeks 8–11). Among these participants, the percentage of smokers who ultimately achieved point abstinence at 6 months was 8.9% (7/79) in the varenicline condition, but only 1.1% (1/89) in the nicotine patch condition and 0% (0/71) in the bupropion plus nicotine patch condition (p=0.01 for varenicline compared with the other conditions; odds ratio=15.46, 95% CI=1.87–128.00). This 7%–8% difference between varenicline and the other treatment conditions accounts for most of the 10% higher 6-month point abstinence obtained with varenicline (Table 2).
Phase 2: Evaluation of Postcessation Rescue Effects
Postquit adaptive changes in treatment had no significant effects on any of the abstinence outcomes (Table 2).
Confirmation of Assumptions Underlying the Adaptive Treatment Design
The assumption underlying the design of phase 1 was that participants showing less of a decrease in ad lib smoking after 1 week of nicotine patch would have lower abstinence rates than those showing larger spontaneous reductions in smoking. This assumption was confirmed based on several analyses. First, among participants receiving nicotine patch, continuous smoking abstinence 11 weeks after the quit date was strongly predicted by the reduction of ad lib smoking 1 week before the quit date. Abstinence was significantly related to all measures of smoking reduction analyzed at this time point, including absolute CO level (p<0.0001; odds ratio=1.09, 95% CI=1.05–1.12), percent decrease in CO level (p<0.0001; odds ratio=1.04, 95% CI=1.03–1.06), and dichotomous classification of whether or not there was greater than 50% decrease in CO level (p<0.0001; odds ratio=6.76, 95% CI=2.98–15.32). When all three variables were entered into a multiple logistic regression analysis, percent reduction in CO level was the only predictor that remained significant (p=0.001). This result suggested that quantitative information about the extent of smoking reduction, above and beyond the dichotomization of >50% or not, has additional predictive value. Quartiles of percent CO level reduction, however, captured the relevant information, reflected by the findings that the correlation between the quartile score and abstinence was as high as for percent reduction (r=0.38 and r=0.36, respectively) and that when quartile was entered into a multiple logistic regression model with percent CO reduction, the percent reduction variable was rendered nonsignificant (p=0.16). Moreover, the quartile percent reduction in CO was a highly significant univariate predictor in its own right (p<0.0001; odds ratio=2.51, 95% CI=1.88–3.36). Figure 2 shows the monotonic increase in abstinence rate as a function of quartile percent reduction in CO level.
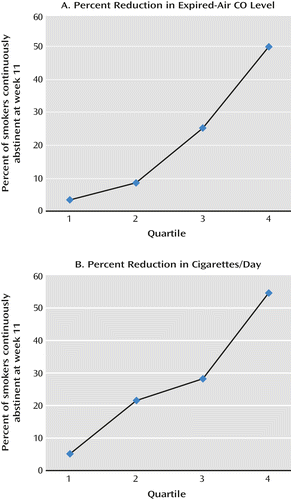
a For smokers receiving nicotine patch treatment, the graphs show the relationship between 4-week continuous smoking abstinence and two measures of initial reduction of ad lib smoking 1 week before the quit date.
A similar analysis on prequit reduction in cigarettes per day yielded a similar conclusion.
The second assumption underlying the adaptive treatment design (phase 2), that participants who lapsed in the first week after their initial quit date would do worse at the end of treatment than those who did not lapse, was evaluated by comparing outcomes between the precessation nicotine patch responders who had a lapse and those who did not lapse and remained on nicotine patch alone. The lapsers showed a much lower rate of continuous 4-week abstinence at the end of treatment: 26.7% compared with 59.2% (p=0.002; odds ratio=0.25, 95% CI=0.10–0.61).
Adverse Events and Side Effects
Treatments were generally well tolerated; however, 25% of participants received dose reductions at some point during the study, with no difference between treatment conditions (range 20%–30%). A number of adverse events occurred that had no definitive causal relationship to treatment: one death believed to be due to stroke, two nonfatal heart attacks, one case of cancer, one participant with tachycardia with transient loss of consciousness, five participants reporting chest pain (including one left bundle branch block, one unifocal premature ventricular contractions, and one report of chest pain with vomiting and hallucinations), one participant with nausea/vomiting, three participants with hives or rash, one participant diagnosed with depression, and one participant reporting weakness, lightheadedness, and difficulty breathing.
Discussion
The results of this study show that smokers who fail to respond adequately to precessation nicotine patch treatment can benefit from being switched to alternative therapies. For these smokers, augmenting nicotine patch treatment with bupropion produced robust increases of approximately 10% in both primary and secondary abstinence outcomes, yielding an abstinence rate that was 2–3 times higher than if they remained on nicotine patch alone. Switching precessation nicotine patch nonresponders to varenicline produced less robust effects, but there was an enhancement in point abstinence at 6 months of about the same magnitude. Thus, it is possible to rescue a significant portion of smokers who would have failed to achieve abstinence if left on nicotine patch alone by identifying these smokers before their quit date and implementing adaptive changes in treatment.
The low success rate of precessation nicotine patch nonresponders who remained on nicotine patch alone replicates findings from previous research in our center. What is novel and important in phase 1 is that adaptive changes in treatment were implemented before the quit date, thereby averting failure instead of waiting for a relapse to occur before attempting to intervene. In so doing, we could minimize the deleterious effects of relapse on motivation and retention in treatment.
Phase 2, which explored the potential of post-quit date adaptive changes in treatment, yielded less promising results. Smokers who responded well to precessation nicotine patch but lapsed in the first week after the quit date did poorly relative to nonlapsers, extending the results of previous trials indicating that early lapses predict failure (17, 18). Unfortunately, however, there was no indication that either of the two potential rescue treatments increased abstinence rates relative to remaining on nicotine patch. In view of the small group sizes studied in this phase of the study, however, the results should be interpreted with caution. Nonetheless, this finding is similar to other negative results from studies attempting to provide alternative treatments after relapse to smoking (19).
In addition to showing that expired-air CO level is a clinically useful biomarker of response to precessation nicotine patch, the results suggest that other simple measures of ad lib smoking may prove useful as well to classify smokers as nicotine patch responders or nonresponders. For example, the decline in self-reported cigarettes per day was also predictive of outcome, and this measure might be more easily gathered in a variety of health care settings. For any measure of ad lib smoking, the question arises as to whether the absolute level or the relative change in the index is a better predictor of outcome. In this study, classification of responder status based on a relative change in expired-air CO level during the first week of precessation patch treatment was found to be a better predictor than absolute level of smoking at the end of week 1. Thus, although some clinicians have recommended using the level of smoking to guide adaptive treatment changes (1), the present data support percent reduction as a more predictive index. Additionally, we found that the relevant information contained in the percent CO reduction measure was captured by a quartile categorization.
Although the fraction of smokers who can be rescued by adaptive treatment with varenicline was similar to that for bupropion plus nicotine patch (based on 6-month point abstinence), it is possible that these smokers were different in terms of baseline characteristics. If so, and if the two distinct subsets could be identified at baseline, then a potentially greater increase in abstinence rates could be achieved by recommending the specific change in adaptive treatment most likely to succeed for a given smoker. Future development of adaptive smoking cessation treatment algorithms should incorporate additional predictive markers, including genotypic as well as phenotypic variables (14, 20). Ultimately, a combination of several predictor variables may be used to identify which smokers need adaptive treatment changes and which specific treatment will yield the highest probability of success with the smallest risk of adverse effects.
This study had several strengths, including the unique feature of using an early prequit marker of response to nicotine patch to decide whether to modify treatment using prescription medications. In addition, the algorithm we used is supported by randomized controlled trial data, which has not been the case for previously proposed algorithms. The study also had limitations. One limitation, as noted earlier, was the relatively small group sizes in the postcessation randomization phase. Thus, we cannot confidently rule out a potential rescue effect for lapsers after the quit date. Second, the study was limited in using a tailored dosage of precessation nicotine patch therapy that is not in accordance with current product labeling in the United States (although precessation nicotine patch is approved in several other countries). Additionally, we did not ascertain the efficacy of other adaptive treatment strategies, such as increasing the length of precessation nicotine patch treatment or increasing nicotine dosage for initial nonresponders. Finally, we did not evaluate whether bupropion plus nicotine patch might prove more efficacious as an initial treatment for nicotine patch responders as well as nonresponders; however, our rationale was to avoid the additional risks and side effects of bupropion for nicotine patch responders, who had a reasonable chance of quitting using nicotine replacement alone.
In conclusion, our results support the use of an adaptive treatment regimen in which smokers initially receive precessation nicotine patch treatment, and if there is an insufficient smoking reduction within the first week, treatment is revised accordingly, either switching to varenicline or adding bupropion to nicotine patch. Further studies should confirm this finding, which has promise for practical implementation in health care settings to increase smoking cessation rates while minimizing treatment-related risks and side effects.
1 : A combination nicotine replacement therapy (NRT) algorithm for hard-to-treat smokers. J Smok Cessat. 2006; 1:3–6Crossref, Google Scholar
2 : An algorithm for choosing among smoking cessation treatments. J Subst Abuse Treat 2008; 34:426–432Crossref, Medline, Google Scholar
3 : Treating tobacco use and dependence: an introduction to the US Public Health Service Clinical Practice Guideline. Respir Care 2000; 45:1196–1199Medline, Google Scholar
4 : Evaluation of the safety of bupropion (Zyban) for smoking cessation from experience gained in general practice use in England in 2000. Eur J Clin Pharmacol 2003; 59:767–773Crossref, Medline, Google Scholar
5 ;
6 ;
7 : Bupropion for smoking cessation: a randomized trial. Arch Intern Med 2004; 164:1797–1803Crossref, Medline, Google Scholar
8 : Suicidal behavior and depression in smoking cessation treatments. PLoS ONE 2011; 6:e27016Crossref, Medline, Google Scholar
9 : Efficacy and safety of the novel selective nicotinic acetylcholine receptor partial agonist, varenicline, for smoking cessation. Arch Intern Med 2006; 166:1571–1577Crossref, Medline, Google Scholar
10 : Varenicline and the evaluation of neuropsychiatric adverse events in smokers. Biol Psychiatry 2011; 69:1017–1018Crossref, Medline, Google Scholar
11 : Risk of serious adverse cardiovascular events associated with varenicline: a systematic review and meta-analysis. CMAJ 2011; 183:1359–1366Crossref, Medline, Google Scholar
12 : Precessation treatment with nicotine skin patch facilitates smoking cessation. Nicotine Tob Res 2006; 8:89–101Crossref, Medline, Google Scholar
13 : Precessation treatment with nicotine patch significantly increases abstinence rates relative to conventional treatment. Nicotine Tob Res 2009; 11:1067–1075Crossref, Medline, Google Scholar
14 : Personalized smoking cessation: interactions between nicotine dose, dependence, and quit-success genotype score. Mol Med 2010; 16:247–253Crossref, Medline, Google Scholar
15 : High-dose nicotine patch therapy: percentage of replacement and smoking cessation. JAMA 1995; 274:1353–1358Crossref, Medline, Google Scholar
16 : Varying nicotine patch dose and type of smoking cessation counseling. JAMA 1995; 274:1347–1352Crossref, Medline, Google Scholar
17 : Predicting smoking cessation: who will quit with and without the nicotine patch. JAMA 1994; 271:589–594Crossref, Medline, Google Scholar
18 : Smoking behavior on the first day of a quit attempt predicts long-term abstinence. Arch Intern Med 1997; 157:335–340Crossref, Medline, Google Scholar
19 : Nicotine patch therapy based on smoking rate followed by bupropion for prevention of relapse to smoking. J Clin Oncol 2003; 21:914–920Crossref, Medline, Google Scholar
20 : Molecular genetics of successful smoking cessation: convergent genome-wide association study results. Arch Gen Psychiatry 2008; 65:683–693Crossref, Medline, Google Scholar



