Polygenic Risk and Social Support in Predicting Depression Under Stress
Abstract
Objective:
Despite substantial progress in identifying genomic variation associated with major depression, the mechanisms by which genomic and environmental factors jointly influence depression risk remain unclear. Genomically conferred sensitivity to the social environment may be one mechanism linking genomic variation and depressive symptoms. The authors assessed whether social support affects the likelihood of depression development differently across the spectrum of genomic risk in two samples that experienced substantial life stress: 1,011 first-year training physicians (interns) in the Intern Health Study (IHS) and 435 recently widowed Health and Retirement Study (HRS) participants.
Methods:
Participants’ depressive symptoms and social support were assessed with questionnaires that were administered before and after the life stressor. Polygenic risk scores (PRSs) for major depressive disorder were calculated for both samples.
Results:
Depressive symptom scores increased by 126% after the start of internship in the IHS sample and by 34% after widowing in the HRS sample. There was an interaction between depression PRS and change in social support in the prediction of depressive symptoms in both the IHS sample (incidence rate ratio [IRR]=0.96, 95% CI=0.93, 0.98) and the HRS sample (IRR=0.78, 95% CI=0.66, 0.92), with higher depression PRS associated with greater sensitivity to changes in social support. Johnson-Neyman intervals indicated a crossover effect, with losses and gains in social support moderating the effect of PRS on depressive symptoms. (Johnson-Neyman interval in the IHS sample, −0.02, 0.71; in the HRS sample, −0.49, 1.92).
Conclusions:
The study findings suggest that individuals with high genomic risk for developing increased depressive symptoms under adverse social conditions also benefit more from nurturing social environments.
Depression is the leading cause of disability worldwide (1). Genomic variation and proximal environment each account for substantial proportions of variability in depression risk. Understanding the interplay between genomic predisposition and specific environmental triggers in the development of depression has the potential to help elucidate the etiology of depression (2, 3). With the collection of large samples and joint analysis through genome-wide association studies (GWASs), there has been marked recent progress in identifying the genomic variation associated with major depression (4). Polygenic risk scores (PRSs) build on GWASs by summing the number of risk variant alleles in each individual weighted by the impact of each allele on risk of disease, aggregating the impact of common polymorphisms and providing a tool to interrogate the cumulative effects of genomic risk in gene-by-environment studies (5).
Two major models on how genomic predisposition and environmental risk interact in depression have been proposed: the diathesis-stress model (6) and the differential susceptibility model (7). In the diathesis-stress framework, genetic risk predisposes the individual to a poor outcome when exposed to an environmental adversity. Family-based genetic studies have demonstrated support for this hypothesis, such that genomically at-risk individuals develop depression at higher rates than low-risk peers following stressful life events, although molecular genetics studies have yielded less consistent results (8). In contrast, the differential susceptibility framework posits that factors that confer risk in negative environments also confer benefits in positive environments “for better or for worse” (7). With the increased availability of GWAS data in large cohorts, investigators are beginning to more comprehensively evaluate gene-by-environment interactions for concordance with either theory, particularly with regard to responses to stressful life events (8).
Robust evidence indicates that loss of social support places an individual at heightened risk for depression (8–10). Recent work has implicated social factors as one of the domains with the strongest causal links to depression (9). However, little is known about who is most sensitive to social support. To assess for the presence of, and to understand the nature of, an interaction between genomic risk and changes in social support, we leveraged two prospective longitudinal cohorts across a time period when they experienced a marked increase in depressive symptoms: 1) individuals who experienced the onset of physician training stress, assessed through the Intern Health Study (11), and 2) individuals who experienced the loss of a spouse, assessed through the Health and Retirement Study (12). In both cohorts, we assessed whether changes in social support affected the likelihood of depression differently across the polygenic risk spectrum and whether the nature of this interaction was more consistent with the diathesis-stress (non-crossover) or differential susceptibility (crossover) models of gene-by-social support interactions.
Methods
Sample
Intern Health Study.
The Intern Health Study (IHS) is a prospective cohort study of first-year medical residents (interns) in the United States. Details on sample selection and recruitment are provided elsewhere (11). Briefly, incoming interns completed a secure online survey 2 months before the start of internship and then every 3 months during internship year. The study was approved by the institutional review board at the University of Michigan, and participants were compensated for their time with $25 gift cards after completing each assessment. Within the 2016 cohort utilized for this study, 1,011 individuals were of European ancestry (the population for whom validated depressive symptom PRSs were available), had a validated PRS, and completed pre-internship and during-internship psychosocial measures (mean age, 27.6 years [SD=2.7]; 47.6% female).
Health and Retirement Study.
The Health and Retirement Study (HRS) is a longitudinal panel study of older adults in the United States (12) utilizing biannual interviews and surveys to assess physical and mental health, family and work life, and aging. Consistent with the literature supporting the classification of widowing as a stressful life event, we selected loss of a spouse as a life stressor (13). Within the sample, 435 individuals had lost a spouse between 2008 and 2016, were of European ancestry, had a validated PRS, and had sufficient pre- and post-loss psychosocial data to be included in the analysis (mean age, 75.6 years [SD=9.6]; 70.7% female).
Measures
Participants in both studies provided information on general demographic factors such as age, self-reported ethnicity, and psychological factors.
Depressive symptoms.
At baseline and quarterly through the year, interns completed the Patient Health Questionnaire–9 (PHQ-9) (14), a validated nine-item self-report measure that asks participants to rate the frequency of symptoms corresponding to the nine symptoms of a major depressive episode according to DSM-5 criteria over the past 2 weeks (0=not at all, 4=nearly every day).
Biannually, HRS participants completed the eight-item version of the Center for Epidemiologic Studies–Depression Scale (CES-D), which has been validated in older adults (15, 16).
Social support.
At baseline and at the 9-month assessment, interns completed the Multidimensional Scale of Perceived Social Support (17), a validated 12-item self-report measure that asks participants to rate how much they agree or disagree (1=strongly disagree, 5=strongly agree) with statements regarding their perceived social support from family, friends, and significant others.
Biannually, as part of the Leave-Behind Questionnaire (18), HRS participants completed a social support measure consisting of 12 items asking how much they feel their spouse/partner, other family, children, and friends “really understand the way [they] feel,” how often they feel they “can rely on [support person] if there is a serious problem,” and how much they feel they “can really open up to them if [they] need to talk about [their] worries” (reverse coded to 1=not at all, 4=a lot). Since the stressful event in this population was loss of a spouse, we defined social support as the mean scores from the three non–spouse/partner categories. We defined pre-widowing as the nearest interview year before reporting losing their spouse, and post-widowing as the interview year in which they reported having been widowed.
Covariates
In the main zero-inflated Poisson regression models, we included neuroticism, the first five PRS ancestry principal components, and sex (biological sex as noted by genotype) as covariates, as these factors have previously been found to be related to the development of depressive symptoms during internship (11) and after loss of a spouse (19). Supplemental models of incident depressive symptoms and of the main model without adjusting for neuroticism are presented in Tables S1–S4 in the online supplement.
PRS Calculation
Interns and HRS participants provided saliva samples using the Oragene salivary DNA kits (20). The Illumina Infinium CoreExome-24+ Chip was used for genotyping for the IHS sample and the Illumina HumanOmni2.5 BeadChips were used for genotyping for the HRS sample. In both samples, using summary statistics from a meta-analysis of 23andMe and Psychiatric Genetics Consortium Phase 2 data on major depression, each participant’s PRS for major depressive disorder (depression PRS) was calculated as the sum of the summary statistic effect sizes for each effect allele multiplied by the number of effect alleles. PRSs are then standardized to have a mean of 0 and standard deviation of 1. We extracted the first five principal components and included these as covariates in all models that use the PRS, to control for population and ancestry-related effects.
Briefly, the PRS of the IHS sample was calculated with the “C+T” method by PRSice, version 2, using genome-wide single-nucleotide polymorphisms (SNPs) (with minor allele frequency ≥0.1 and outside the major histocompatibility complex region, no clumping) that overlap with summary statistics data from the major depressive disorder GWAS. This approach follows the established protocol from Fang et al. (21), in which the PRS generated with this method was shown to have a significant association with depression during the internship year. The PRSs for the HRS sample were obtained from the HRS data repository, a publicly available site (https://hrsdata.isr.umich.edu/). These PRSs were also generated by the “C+T” method using genome-wide SNPs (without clumping) that overlap with summary statistics data from a major depressive disorder GWAS.
Statistical Analysis
To determine whether social support change moderated the effect of genomic risk on depressive symptoms, we assessed for the presence of an interaction between depression PRS and changes in social support using a zero-inflated Poisson regression with standardized predictors first in the IHS cohort, then replicated in the HRS cohort. For both samples, we regressed social support scores at follow-up on baseline scores to derive a residual change in the social support measure used as a predictor in our main models. Zero-inflated Poisson regressions produce two estimated models: a logistic model for the presence of a zero or non-zero outcome, and a count model (Poisson model) across all values of the outcome. In nonclinical populations, the number (or count) of depressive symptoms endorsed is expected to follow a Poisson distribution, with additional zeros present for individuals who are not experiencing any symptoms of depression (22). In these analyses, the count portion of the zero-inflated Poisson model provides estimates of the predictors’ effects on the individual’s depressive symptom total score, while the logistic portion of the model provides information on the predictors’ effects on the likelihood of an individual reporting any versus no symptoms. As our study hypothesis is most directly assessed through the quantitative change in depressive symptom level, the focus of our interpretation is the count portion of the model, but we include the logistic portion of the model for completeness.
The regression equation for the presence of an interaction between depression PRS and changes in social support was:
YDEP = βPRS + βSS + βSEX + βAGE + βNEU + βPRS*βSS + βPRS*βSEX + βPRS*βAGE + βPRS*βNEU + βPC1+ βPC2 + βPC3 + βPC4 + βPC5,
where PRS=polygenic risk score, SS=social support, NEU=neuroticism, and PC=principal component. The term in boldface is the interaction of interest.
In two-way interactions, the Johnson-Neyman technique (23) estimates the values of a moderator where the slope of the predictor differs significantly from 0. Therefore, we used Johnson-Neyman intervals from the “interactions” package in R to identify the values of social support for which depression PRS was significantly associated with depressive symptoms. Specifically, we were interested in whether any observed interaction effects were consistent with non-crossover or crossover interactions, driven by the theories of diathesis-stress and differential susceptibility, respectively.
All analyses were conducted using RStudio, version 1.1.456 (R Core Team, 2019). Missing data were managed using an imputation step for covariates only and cases with <25% missing covariate data. We adjusted the models in both samples for baseline covariates associated with depressive symptoms (age, sex, neuroticism).
Results
The demographic and baseline characteristics of the two samples are summarized in Table 1.
| Characteristic | Intern Health Study (N=1,011) | Health and Retirement Study (N=435) | ||||
|---|---|---|---|---|---|---|
| N | % | N | % | |||
| Female | 481 | 47.58 | 308 | 70.80 | ||
| Scale | Mean | SD | Scale | Mean | SD | |
| Age (years) | 27.6 | 2.68 | 75.62 | 9.70 | ||
| Neuroticism score | NEO-FFI | 21.16 | 8.73 | MDI | 2.63 | 0.47 |
| Baseline depressive symptoms | PHQ-9 | 2.51 | 2.75 | CES-D-8 | 1.75 | 2.11 |
| Baseline social support | MSPSS | 4.37 | 0.74 | LBQ items | 3.12 | 0.56 |
TABLE 1. Demographic and baseline characteristics of the Intern Health Study and Health and Retirement Study samplesa
Changes in Social Support and Depressive Symptoms
In the IHS sample, mean social support decreased from 4.37 (SD=0.74) at baseline to 4.20 (SD=0.75) during internship (paired t=9.40, p<0.001), and mean PHQ-9 depressive symptom scores increased from 2.51 (SD=2.75) at baseline to 5.68 (SD=4.57) during internship (t=−24.99, p<0.001). In the HRS sample, mean perceived social support increased from 3.12 (SD=0.55) to 3.20 (SD=0.53) (t=−2.75, p=0.006) after loss of a spouse, and mean CES-D depressive symptom scores increased from 1.75 (SD=2.11) to 2.44 (SD=2.37) after loss of a spouse (t=−8.36, p<0.001).
To assess whether genomic risk for depression was moderated by changes in social support, we tested the interaction between depression PRS and changes in social support on depressive symptoms after the stressor (start of internship, loss of spouse). In the IHS, after accounting for baseline factors previously associated with internship depression (age, biological sex, and neuroticism) as well as interactions between the depression PRS and other predictors, the association between depression PRS and internship depressive symptoms (incidence rate ratio [IRR]=1.14, 95% CI=1.04, 1.24, p<0.001) was moderated by change in social support (adjusted IRR of interaction term, 0.96, 95% CI=0.93, 0.98, p<0.001) (Table 2).
| Predictor | IRR | 95% CI | p |
|---|---|---|---|
| Count portion of model | |||
| Intercept | 6.48 | 5.95, 7.05 | <0.001 |
| Depression PRS | 1.14 | 1.04, 1.24 | 0.003 |
| Support change | 0.88 | 0.86, 0.90 | <0.001 |
| Biological sex | 0.94 | 0.89, 0.99 | 0.015 |
| Age | 1.00 | 0.97, 1.03 | 0.941 |
| Neuroticism | 1.30 | 1.27, 1.34 | <0.001 |
| Support change×depression PRS | 0.96 | 0.93, 0.98 | <0.001 |
| Logistic portion of model | |||
| Intercept | 0.17 | 0.08, 0.36 | <0.001 |
| Depression PRS | 1.43 | 0.69, 2.95 | 0.335 |
| Support change | 1.49 | 1.15, 1.92 | 0.002 |
| Biological sex | 0.56 | 0.34, 0.92 | 0.022 |
| Age | 1.2 | 0.99, 1.46 | 0.069 |
| Neuroticism | 0.42 | 0.32, 0.55 | <0.001 |
| Support change×depression PRS | 1.13 | 0.88, 1.46 | 0.333 |
TABLE 2. Results of analyses testing the interaction of depression PRS and social support on postexposure depressive symptoms in the Intern Health Study sample: zero-inflated Poisson regression for postexposure depressive symptomsa
Similarly in the HRS sample, after accounting for baseline factors previously associated with depression (age, sex, neuroticism) as well as interactions between the depression PRS and other predictors, the association between depression PRS and prevalent depressive symptoms (IRR=0.98, 95% CI=0.82, 1.17, p=0.84) was moderated by change in perceived social support (adjusted IRR of interaction term, 0.78, 95% CI=0.66, 0.92, p=0.004) (Table 3).
| Predictor | IRR | 95% CI | p |
|---|---|---|---|
| Count portion of model | |||
| Intercept | 3.04 | 2.62, 3.52 | <0.001 |
| Depression PRS | 0.98 | 0.82, 1.17 | 0.842 |
| Support change | 1.04 | 0.89, 1.22 | 0.594 |
| Biological sex | 0.93 | 0.78, 1.10 | 0.404 |
| Age | 0.91 | 0.85, 0.98 | 0.008 |
| Neuroticism | 0.9 | 0.84, 0.97 | 0.004 |
| Support change×depression PRS | 0.78 | 0.66, 0.92 | 0.004 |
| Logistic portion of model | |||
| Intercept | 0.28 | 0.15, 0.53 | <0.001 |
| Depression PRS | 0.6 | 0.28, 1.28 | 0.188 |
| Support change | 0.92 | 0.51, 1.65 | 0.772 |
| Biological sex | 1.12 | 0.57, 2.17 | 0.747 |
| Age | 1.08 | 0.82, 1.42 | 0.576 |
| Neuroticism | 1.45 | 1.11, 1.89 | 0.006 |
| Support change×depression PRS | 0.57 | 0.29, 1.12 | 0.102 |
TABLE 3. Results of analyses testing the interaction of depression PRS and social support on postexposure depressive symptoms in the Health and Retirement Study sample: zero-inflated Poisson regression for postexposure depressive symptomsa
To assess whether genomic interaction was present across the range of negative and positive environments (a crossover effect), we calculated the Johnson-Neyman interval for the effects of change in social support on the relationship between depression PRS and depressive symptoms. The Johnson-Neyman interval indicated that depression PRS was associated with depressive symptoms at social support change scores below −0.02 and above 0.71 in the IHS sample and below −0.49 and above 1.92 in the HRS sample, but not with the intermediate changes in support between those values. Specifically, higher depression PRS was associated with higher depressive symptom scores in the context of decreased social support but with lower depressive symptom scores in the context of increased social support in both samples.
Finally, to test the effect of depression PRS and change in social support on incident depressive symptoms, we included pre-exposure depressive symptom scores in an additional model. In this incident symptom model, we noted a similar pattern of results in both samples (Figure 1), although the depression PRS-by-support interaction of interest weakened with increased variability around the effect in the HRS sample (adjusted IRR for interaction term, 0.85, 95% CI=0.72, 1.00, p=0.046) (see Tables S1 and S2 in the online supplement).
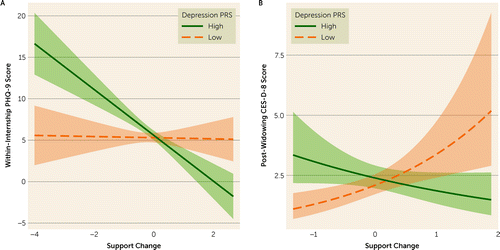
FIGURE 1. Depressive symptom polygenic risk score (PRS) and changes in social support interact to predict depressive symptoms during internship and after loss of a spousea
aDepressive symptoms were measured with the Patient Health Questionnaire–9 (PHQ-9) during internship in the Intern Health Study (panel A) and with the eight-item Center for Epidemiological Studies Depression Scale (CES-D-8) after loss of a spouse in the Health and Retirement Study (panel B). Analyses controlled for biological sex, age, and neuroticism. In both samples, change in social support interacted with depression PRS in a crossover manner.
Missingness
Compared with individuals in the IHS who did not complete internship measures, individuals who completed internship measures reported lower baseline depressive symptoms but did not differ significantly in age, sex, PRS, or baseline social support. In the HRS, individuals included in the analysis reported higher post-loss depressive symptoms compared with those who were excluded due to lack of post-loss psychosocial questionnaires but did not differ significantly in age, sex, PRS, baseline social support, or pre-loss depressive symptoms.
Discussion
In this analysis of two cohorts undergoing developmentally normative stressful life events—young adults transitioning to a stressful workplace environment and older adults transitioning to widowhood—we found consistent evidence that an environmental factor, social support, moderates genomic risk for depression. Individuals at highest genomic risk for depression were most sensitive to the loss of social support but were also the most responsive to gaining social support.
The finding of an interaction between depression PRS and social support provides insight into one pathway through which genomic risk leads to depression. Specifically, individuals with higher depression PRS scores appear to be more susceptible to developing depression in the context of losing social support, consistent with differential susceptibility (7). Elucidating genomic susceptibility to specific precipitants of depression can help in the development of personalized guidance for individuals at risk. Future studies should refine the genomic profile that predisposes to susceptibility to social stress. Further, exploring the genomic sensitivity to other precipitants of depression, such as sleep deprivation, interpersonal conflict, job loss, and other stressors, may facilitate identification of individualized differential risk pathways.
In addition to the presence of the interaction between genomic risk and changes in social support, we expand on the current literature by testing whether the interaction is seen only when individuals at high genomic risk lose support (diathesis-stress) or when individuals at high genomic risk gain support as well (differential susceptibility). We found that the interaction was present in both the direction of loss in social support and the direction of gain in social support. Specifically, those who were most sensitive to loss of social support also benefited the most from gaining social support. This finding provides support for the differential sensitivity hypothesis of genetic risk and identifies a potential explanation for evolutionary selection mechanisms (24) that allow for genomic elements that confer risk for psychiatric disorders to persist in the population.
There are limitations to this work. First, because the PRSs available for these samples were validated for individuals of European ancestry, our analyses focused only on those individuals. Establishing validated PRSs for non-European samples is critical to ensure equitable benefits from any advances in genomic medicine. Second, our findings regarding the nature of moderation were stronger in the IHS sample, while other contextual factors, such as the spouse’s health status, age at time of death, and caregiving roles, may have weakened the moderation in the HRS sample, suggesting some potential differences in key mechanisms across the life course. Third, our missing data were not missing at random, and interns in the IHS cohort who did not provide within-internship data had reported higher scores on the PHQ-9 prior to internship, so we are missing individuals who started internship with more depression and potentially those who go on to become most depressed, although this is mitigated by full information modeling. Finally, social support and depressive symptoms were measured at static points, which precludes us from testing a directional relationship between social support and depressive symptoms, and this relationship is important in targeting interventions. In this study, we focused on the effect of social support on depression. However, it is possible for depression to cause changes in social support, and, in particular, self-reported social support. Future work may focus on more fine-grained temporal measurement of how social support and depressive symptoms change interdependently over time.
This work also has several strengths. By measuring social support and depressive symptoms both before and after the onset of incident depressive symptoms, the effect of retrospective reporting bias is minimized, allowing for a more accurate assessment of how genomic variation influences the relationship between social support and depression (9). Additionally, the consistent finding of both the presence of the interaction and the nature of the interaction in two samples that differ markedly in age and social circumstances raises confidence in the results. Further, this work examined the differential impact of a crucial environmental factor, changes in social support, across a cumulative measure of genetic risk. These methods allowed us to interrogate a significant gene-by-environment interaction in a more comprehensive manner than was previously possible.
In summary, we show that genomic risk for major depression differentially affects how changes in social support relate to depressive symptoms, in a manner consistent with differential susceptibility. Future work should test these findings in other populations and stressors. Specifically, investigating the mechanisms by which changes in social support across the life course or in the context of chronic stressors relate to incident depression would provide valuable insight into the ways in which genetic risk predisposes individuals to, or protects them from, depression across development. Comprehensive work that investigates factors that individuals and society can change to mitigate their genomic risk of developing depression across the lifespan is integral in depression prevention and promotion of mental well-being.
1. : Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry 2005; 62:593–602Crossref, Medline, Google Scholar
2. : Candidate gene-environment interaction research: reflections and recommendations. Perspect Psychol Sci 2015; 10:37–59Crossref, Medline, Google Scholar
3. : The implications of gene-environment interactions in depression: will cause inform cure? Mol Psychiatry 2008; 13:1070–1078Crossref, Medline, Google Scholar
4. : Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat Neurosci 2019; 22:343–352Crossref, Medline, Google Scholar
5. : Power and predictive accuracy of polygenic risk scores. PLoS Genet 2013; 9:e1003348Crossref, Medline, Google Scholar
6. : Schizotaxia, schizotypy, schizophrenia. Am Psychol 1962; 17:827–838 Crossref, Google Scholar
7. : For better and for worse: differential susceptibility to environmental influences. Curr Dir Psychol Sci 2007; 16:300–304 Crossref, Google Scholar
8. : Genetic heterogeneity in depressive symptoms following the death of a spouse: polygenic score analysis of the US Health and Retirement Study. Am J Psychiatry 2017; 174:963–970Link, Google Scholar
9. : An exposure-wide and Mendelian randomization approach to identifying modifiable factors for the prevention of depression. Am J Psychiatry 2020; 177:944–954Link, Google Scholar
10. : The association between social relationships and depression: a systematic review. J Affect Disord 2015; 175:53–65Crossref, Medline, Google Scholar
11. : A prospective cohort study investigating factors associated with depression during medical internship. Arch Gen Psychiatry 2010; 67:557–565Crossref, Medline, Google Scholar
12. : Cohort profile: the Health and Retirement Study (HRS). Int J Epidemiol 2014; 43:576–585Crossref, Medline, Google Scholar
13. : The social readjustment rating scale. J Psychosom Res 1967; 11:213–218Crossref, Medline, Google Scholar
14. : The PHQ‐9: validity of a brief depression severity measure. J Gen Intern Med 2001; 16:606–613Crossref, Medline, Google Scholar
15. : A revised CES-D measure of depressive symptoms and a DSM-based measure of major depressive episodes in the elderly. Int Psychogeriatr 1999; 11:139–148Crossref, Medline, Google Scholar
16. : Validation of the eight-item Center for Epidemiologic Studies Depression Scale (CES-D) among older adults. Curr Psychol 2015; 34:681–692 Crossref, Google Scholar
17. : The Multidimensional Scale of Perceived Social Support. J Pers Assess 1988; 52:30–41 Crossref, Google Scholar
18. : Supportive interactions, negative interactions, and depressed mood. Am J Community Psychol 1990; 18:423–438Crossref, Medline, Google Scholar
19. : Depression through the first year after the death of a spouse. Am J Psychiatry 1991; 148:1346–1352Link, Google Scholar
20. : New saliva DNA collection method compared to buccal cell collection techniques for epidemiological studies. Am J Hum Biol 2007; 19:319–326Crossref, Medline, Google Scholar
21. : Genomic prediction of depression risk and resilience under stress. Nat Hum Behav 2020; 4:111–118Crossref, Medline, Google Scholar
22. : Structural zeroes and zero-inflated models. Shanghai Arch Psychiatry 2014; 26:236–242Medline, Google Scholar
23. : The Johnson-Neyman technique, its theory and application. Psychometrika 1950; 15:349–367Crossref, Medline, Google Scholar
24 : The evolutionary basis of risky adolescent behavior: implications for science, policy, and practice. Dev Psychol 2012; 48:598–623Crossref, Medline, Google Scholar



