Reducing Suicidal Ideation Through Insomnia Treatment (REST-IT): A Randomized Clinical Trial
Abstract
Objective:
The authors sought to determine whether targeted treatment of insomnia with controlled-release zolpidem (zolpidem-CR) in suicidal adults with insomnia would provide a reduction in suicidal ideation superior to placebo.
Methods:
Reducing Suicidal Ideation Through Insomnia Treatment was an 8-week three-site double-blind placebo-controlled parallel-group randomized controlled trial of zolpidem-CR hypnotic therapy compared with placebo, in conjunction with an open-label selective serotonin reuptake inhibitor. Participants were medication-free 18- to 65-year-olds with major depressive disorder, insomnia, and suicidal ideation. Suicidal ideation was the main outcome, measured first by the Scale for Suicide Ideation and second by the Columbia–Suicide Severity Rating Scale (C-SSRS).
Results:
A total of 103 participants were randomly assigned to receive zolpidem-CR (N=51) or placebo (N=52) (64 women and 39 men; mean age=40.5 years). Zolpidem-CR had a robust anti-insomnia effect, especially in patients with the most severe insomnia symptoms. No significant treatment effect was observed on the Scale for Suicide Ideation (least squares mean estimate=−0.56, SE=0.83, 95% CI=−2.19, 1.08), but the reduction in scores was significantly positively related to improvement in insomnia after accounting for the effect of other depression symptoms. The C-SSRS indicated that zolpidem-CR had a significant treatment effect (least squares mean estimate=−0.26, SE=0.12, 95% CI=−0.50, −0.02). The advantage for zolpidem-CR in reducing suicidal ideation on the C-SSRS was greater in patients with more severe insomnia. No deaths or suicide attempts occurred.
Conclusions:
Although the results do not support the routine prescription of hypnotic medication for mitigating suicidal ideation in all depressed outpatients with insomnia, they suggest that coprescription of a hypnotic during initiation of an antidepressant may be beneficial in suicidal outpatients, especially in patients with severe insomnia.
In the United States, there have been recent reductions in mortality related to medical illness, but mortality rates from suicide steadily increased from 12.5 to 15.0 per 100,000 annually between 2001 and 2015 (1). Insomnia has been identified as a risk factor for suicidal ideation, suicidal behavior, and suicide death (hereafter collectively termed “suicide”) (2, 3), but thus far there have been no randomized clinical trials examining whether targeted pharmacologic treatment of insomnia would reduce the risk of suicide. It is reasonable to hypothesize that hypnotic medication might reduce the risk of suicide because changes in insomnia symptoms precede suicidal ideation (3, 4). Patients with insomnia (5) and survivors of suicide attempts show poorer performance on standardized measures of interpersonal problem solving compared with psychiatric patients who have not made a suicide attempt (6), as well as poorer performance in attention, working memory, and executive function (7–9). Thus, the impaired problem solving associated with insomnia could play a role in suicide. Hypnotic medication might reduce suicidal ideation, but the possibility of benefit from hypnotics must be balanced against the risks of suicide associated with hypnotics (10).
To examine the effect of treatment of insomnia on suicide risk, we conducted the randomized clinical trial Reducing Suicidal Ideation Through Insomnia Treatment (REST-IT) to study the effects of placebo relative to a bedtime dose of controlled-release zolpidem (zolpidem-CR), a hypnotic medication, on suicidal depressed outpatients with insomnia who were receiving open-label selective serotonin reuptake inhibitors (SSRIs). Our hypothesis was that the addition of zolpidem-CR would result in greater reduction of suicidal ideation.
Methods
The REST-IT study was a multisite parallel-arm randomized clinical trial, with the Medical College of Georgia as the coordinating and recruiting site, Duke University and the University of Wisconsin as recruiting sites, and Wake Forest School of Medicine as the data management site. A data safety monitoring board provided oversight and reviewed the results of two planned interim analyses with stopping rules in effect. The results of the analyses were shared with the monitoring board but not with the investigative team. Neither interim analysis pointed toward futility or toward an early significant effect. As a result, the monitoring board instructed the investigative team to continue recruitment. During the 8 weeks of randomized treatment, all patients received an open-label SSRI (95 patients received fluoxetine, three patients received sertraline, and five patients received citalopram) for depression, as well as either zolpidem-CR or placebo at bedtime. The initiation of the open-label SSRI and the double-blinded randomized assignment to receive either zolpidem-CR or placebo occurred simultaneously. Patients were given enough medication only to last until the next scheduled visit, plus 3 days. Follow-up visits were scheduled at 1, 2, 4, 6, and 8 weeks after randomization. At the conclusion of the intervention period, arrangements for care-as-usual outpatient visits were made for each participant, and the participants received prescriptions for a sufficient number of SSRIs to last until the first care-as-usual visit. The blinded study drug (zolpidem-CR or placebo) was stopped abruptly without tapering. Patient safety and additional clinical assessments were conducted by telephone for the first 2 weeks after the randomization phase of the trial and while in transition to care as usual. Other details regarding practices to ensure participant safety can be found elsewhere (11). All parties remained blind to zolpidem-CR or placebo treatment assignment.
Participants
Participant recruitment came from advertising and the routine flow of self-referrals to psychiatry clinics. Participants were outpatients 18–65 years of age with a diagnosis of major depressive disorder, confirmed by the Structured Clinical Interview for DSM-IV (SCID) (12), and who met research diagnostic criteria for insomnia (13). Participants were free of all psychotropic medications for ≥1 week prior to baseline assessment (except for fluoxetine, where the requirement was that the patient be off fluoxetine for >4 weeks) and reported a habitual sleep latency ≥30 minutes or a wake after sleep onset ≥30 minutes, as well as sleep efficiency of ≤85%. Baseline inclusion criteria included a score ≥20 on the 24-item Hamilton Depression Rating Scale (HAM-D) and a score >7 on the Insomnia Severity Index (14, 15). Mini-Mental State Examination scores were ≥24 (16). Inclusion criteria also required a score ≥3 on the Scale for Suicide Ideation (17), but participants were free of suicidal plans or intent, as indicated by Columbia–Suicide Severity Rating Scale (C-SSRS) scores ≤3 on the ideation dimension (18). Patients were excluded if they had a SCID-confirmed lifetime diagnosis of bipolar disorder, schizophrenia, or alcohol or substance abuse, or a prior diagnosis of any sleep disorder other than insomnia. Patients also were excluded if they had a body mass index >50, a positive result on a urine drug screen, or a positive urine pregnancy test; if they could not speak, read, or write English; or if they posed an imminent danger to others. In addition, all participants underwent in-lab polysomnography or a portable home sleep test to assess for sleep apnea. Participants with an apnea-hypopnea index >10 were excluded (19). All participants provided written informed consent after receiving a complete description of the study, and the protocol was approved by the institutional review board of each site. Patients were paid $50 for each visit after randomized assignment.
Randomized Assignment and Masking
Participants were stratified by site, sex, and prior history of suicide attempts and were randomly assigned within strata to receive zolpidem-CR or placebo with equal probability, using computer-generated variably sized permuted block randomization to ensure approximately equal accrual to each treatment throughout the study. Block sizes of varying length were used to ensure that future assignments could not be inferred from past assignments. At the time of randomization, all patients received 20 mg of fluoxetine, 50 mg of sertraline, or 20 mg of citalopram daily (all open label), but the dosage could be doubled at the end of 4 weeks if, at that time, participants had a HAM-D score >15. Fluoxetine was used in every case unless the participant strongly preferred sertraline or citalopram.
Recruitment began in November 2012. Study medications were overencapsulated to look identical, masked in a central pharmacy, and sent to the investigative teams. Participants were randomly assigned and blind to 12.5 mg of zolpidem-CR nightly or placebo until February 2013, when the Food and Drug Administration released guidance that zolpidem-CR should not be initially prescribed for women at a dosage greater than 6.25 mg at bedtime (20). We then adjusted dosing so that participants were randomly assigned either to 6.25 mg of zolpidem-CR or matching placebo, along with an open-label SSRI. At the end of week 1, the zolpidem-CR or placebo dosage could be increased to 12.5 mg if there was inadequate anti-insomnia effect in the absence of side effects. Participants brought pill bottles to each visit, and pill counts were made to estimate adherence.
Clinical Measurements
Suicidal ideation was the primary outcome variable. The Scale for Suicide Ideation and the C-SSRS were administered at each visit. The Scale for Suicide Ideation is a self-rated scale consisting of 19 items that evaluate suicidal desire and planning (17, 21, 22). Each item is rated on a 3-point scale, from 0 to 2, for a maximum score of 38; a lower score indicates less severe suicidal ideation. A score ≥3 on the Scale for Suicide Ideation is a significant predictor for suicide death over a period of up to 20 years (hazard ratio=6.6) (23). The C-SSRS (18) is an observer-rated scale (24). The suicidal ideation “intensity” dimension of the C-SSRS is scored 1–5, with 5 representing suicidal ideation with a plan and intent. In addition to tracking the intensity of suicidal ideation, the C-SSRS was also used to assess participants’ sense of control over their suicidal thinking, the presence of deterrents that inhibited suicide, and the motivation behind their suicidal thinking (i.e., getting attention and producing a reaction in other people compared with the desire to escape mental pain) (18, 24).
Secondary Outcome Variables
Sleep metrics.
The Insomnia Severity Index was administered at each visit. This instrument is a self-report measure of insomnia severity, with seven items each scored 0–4; higher scores indicate greater insomnia, and scores ≤7 indicate no insomnia (15). Participants completed a daily sleep diary in the morning during randomized treatment to record self-reported sleep latency, wake after sleep onset, number of awakenings, and total sleep time. Sleep diaries were not kept prior to randomization. The Epworth Sleepiness Scale was recorded once at baseline (25). The Dysfunctional Beliefs and Attitudes About Sleep Scale was used to capture distorted beliefs about sleep (26). The total possible scores on this self-administered 16-item scale range from 0 to 10, with higher scores indicating more dysfunctional beliefs and attitudes (26).
The frequency and intensity of disturbing dreams and nightmares were measured with the self-administered Disturbing Dream and Nightmare Severity Index, as nightmares mediate some of the effect of insomnia on suicidal ideation (27). Possible scores range from 0 to 37, with higher scores indicating greater problems with nightmares, and scores >10 are predictive of a nightmare disorder (28).
Depression severity.
Depression severity was measured by administration of the HAM-D by study staff at each visit (14).
Hopelessness.
Hopelessness was measured at each visit with the self-administered Beck Hopelessness Scale, which has 20 items and assesses pessimism and negative expectancies for the future with demonstrated validity across a wide age range (29, 30). Among adolescents and adults, it can predict future suicidal behavior (31–33). The range of possible scores on the Beck Hopelessness Scale is 0 to 20, with higher scores indicating more hopelessness.
Health-related quality of life.
Two subscales of the Behavior and Symptom Identification Scale were recorded at each visit: the daily living and role functioning subscale and the relationship to self and others subscale (34). The range of possible scores for each subscale is 0 to 4, with higher scores indicating a poorer self-reported health-related quality of life.
Overall status and treatment response.
The participants’ overall clinical status and response to treatment were assessed with the Clinical Global Impressions Scale severity scale (CGI-S), completed by a study psychiatrist at all visits. The CGI-S is scored from 1 to 7, with lower scores indicating less illness (35). The improvement scale of the CGI (CGI-I) was completed by a study psychiatrist at all postrandomization visits, with lower scores indicating more improvement. CGI scales were completed using all available data.
Antidepressant treatment resistance.
The participants’ response or resistance to pharmacotherapy in the present episode of illness was measured at baseline by the Antidepressant Treatment History Form (36).
Adverse events.
Each postrandomization visit included a standardized assessment of the presence or absence of drowsiness while driving or operating dangerous machinery, as well as accidents or injuries. If any injuries or accidents occurred, the time of day and relationship to the timing of the last dose of study drug was also recorded. In addition, the study psychiatrist elicited spontaneous reports of adverse events and serious adverse events at each treatment visit.
Actigraphy.
The participants wore an actigraph on their nondominant wrist for the duration of the randomized treatment to monitor rest and activity cycles. Actigraphy results will be described in a subsequent report.
Statistical Analyses
Recruitment targets were picked to achieve 80% power to detect a between-group difference of two points in the mean postrandomization score on the Scale for Suicide Ideation, assuming a standard deviation of 3.1, at the 5% two-sided level of significance, and also assuming two interim assessments and a dropout rate of 20%. A difference of two points on the Scale for Suicide Ideation corresponds to an increased risk of suicide death over a subsequent period of 20 years (23). These assumptions led to a target of 103 randomly assigned participants. The primary hypothesis was tested using a mixed-model analysis of covariance (ANCOVA) to assess treatment differences between the two study arms in mean posttreatment scores after adjusting for baseline scores and the design parameters: clinic site, sex, and history of suicide attempts. An autoregressive covariance structure was used to model the within-patient correlation over time. Visit was considered a class variable, and a linear contrast was used to assess the treatment effect on the mean score across time. The C-SSRS was similarly examined as a second indicator of suicidal ideation. This same strategy was used to assess the effect of zolpidem-CR on other outcomes, except that the covariance structure was chosen to minimize the Bayesian information criterion. Separate mixed-effects repeated measures ANCOVA models, including additional covariates and their interactions with zolpidem-CR, were also evaluated. Insomnia (the Insomnia Severity Index) and suicidal ideation (the Scale for Suicide Ideation) were assessed for 2 weeks after the end of treatment, and linear contrasts within a mixed-effects model containing all times were used to compare week 8 against the mean of weeks 9 and 10. A Wilcoxon rank sum test was used to assess group differences in compliance because of the skewed distribution.
Results
Baseline
The screening of participants began in November 2012, and the last randomly assigned participant exited the study in June 2017 (Figure 1). A total of 103 participants underwent randomized assignment. Participant characteristics are summarized in Table 1, and there were no significant differences between treatment groups at baseline. The majority of participants were women, and 39% of the sample represented minorities. Generalized anxiety disorder and posttraumatic stress disorder were present in 40% and 28% of the participants, respectively, at baseline. Fifty-seven percent of the sample were naive to antidepressant medication in this episode of major depressive disorder, and 30% had a past suicide attempt. The intensity of suicidal ideation, as measured by the Scale for Suicide Ideation, was moderate (37). At baseline, only 18% of participants described “a lot” of difficulty or an inability to control their suicidal thoughts, and 76% described deterrents that played a major role in preventing a suicidal act. Overall depression severity and insomnia severity were high (Table 1).
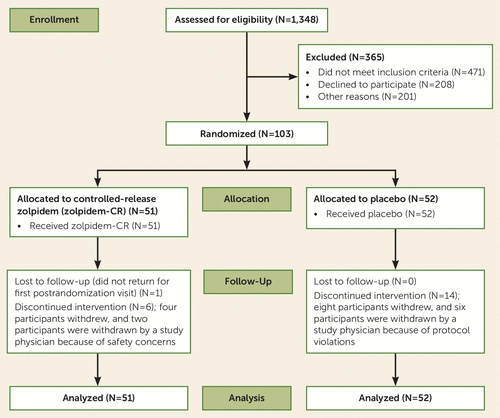
FIGURE 1. CONSORT diagram of participants in the Reducing Suicidal Ideation Through Insomnia Treatment study
| Characteristic | Controlled-Release Zolpidem (N=51) | Placebo (N=52) | Overall (N=103) | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| Age (years) | 39.7 | 14.5 | 41.2 | 12.0 | 40.5 | 13.2 |
| Body mass index | 28.3 | 6.4 | 28.2 | 5.6 | 28.2 | 6.0 |
| N | % | N | % | N | % | |
| Female | 32 | 63 | 32 | 62 | 64 | 62 |
| Clinic | ||||||
| 1 | 23 | 45 | 27 | 52 | 50 | 49 |
| 2 | 15 | 29 | 15 | 29 | 30 | 29 |
| 3 | 13 | 25 | 10 | 19 | 23 | 22 |
| Race or ethnicity | ||||||
| Caucasian | 30 | 59 | 33 | 63 | 63 | 61 |
| African American | 12 | 24 | 16 | 31 | 28 | 27 |
| Hispanic | 4 | 8 | 1 | 2 | 5 | 5 |
| Other | 5 | 10 | 2 | 4 | 7 | 7 |
| Antidepressant trials in current episode | ||||||
| Zero | 29 | 57 | 30 | 58 | 59 | 57 |
| One | 14 | 27 | 12 | 23 | 26 | 25 |
| Two or more | 8 | 16 | 10 | 19 | 18 | 17 |
| Lifetime suicide attempt(s) | 15 | 29 | 16 | 31 | 31 | 30 |
| Posttraumatic stress disorder | 13 | 25 | 16 | 31 | 29 | 28 |
| Obsessive-compulsive disorder | 2 | 4 | 3 | 6 | 5 | 5 |
| Generalized anxiety disorder | 20 | 39 | 21 | 41 | 41 | 40 |
| Panic disorder with agoraphobia | 9 | 18 | 8 | 15 | 17 | 17 |
| Panic disorder without agoraphobia | 10 | 20 | 7 | 14 | 17 | 17 |
| Mean | SD | Mean | SD | Mean | SD | |
| Scale for Suicide Ideation: suicide ideation score | 12.2 | 5.3 | 11.8 | 5.3 | 12.0 | 5.3 |
| N | % | N | % | N | % | |
| Scale for Suicide Ideation: suicide attempts | ||||||
| Never | 36 | 71 | 36 | 69 | 72 | 70 |
| Once | 8 | 16 | 8 | 15 | 16 | 16 |
| Two or more times | 7 | 14 | 8 | 15 | 15 | 15 |
| Scale for Suicide Ideation: death wish | ||||||
| No suicide attempt | 36 | 71 | 36 | 69 | 72 | 70 |
| Low | 3 | 6 | 3 | 6 | 6 | 6 |
| Moderate | 6 | 12 | 4 | 8 | 10 | 10 |
| High | 6 | 12 | 9 | 17 | 15 | 15 |
| Mean | SD | Mean | SD | Mean | SD | |
| Columbia–Suicide Severity Rating Scale (C-SSRS) past-week ideation intensity | 1.71 | 1.03 | 1.58 | 1.02 | 1.64 | 1.02 |
| N | % | N | % | N | % | |
| C-SSRS: a lot of difficulty or inability to control thoughts | 6 | 12 | 12 | 23 | 18 | 18 |
| C-SSRS: deterrents probably or definitely stopped suicide attempt | 43 | 86 | 35 | 67 | 78 | 76 |
| Clinical Global Impressions Scale, severity scale | ||||||
| Mildly ill | 3 | 6 | 0 | 0 | 3 | 3 |
| Moderately ill | 33 | 65 | 32 | 62 | 65 | 63 |
| Markedly ill | 13 | 25 | 18 | 35 | 31 | 30 |
| Severely ill | 2 | 4 | 2 | 4 | 4 | 4 |
| Mean | SD | Mean | SD | Mean | SD | |
| Hamilton Depression Rating Scale score | 28.7 | 4.7 | 29.6 | 7.0 | 29.1 | 5.9 |
| Insomnia Severity Index score | 20.7 | 4.0 | 21.0 | 4.3 | 20.9 | 4.1 |
| Beck Hopelessness Scale score | 13.4 | 4.8 | 12.7 | 4.6 | 13.1 | 4.7 |
| Disturbing Dream and Nightmare Severity Index score | 10.5 | 8.8 | 10.2 | 7.7 | 10.3 | 8.2 |
| Dysfunctional Beliefs and Attitudes About Sleep Scale score | 6.7 | 1.6 | 6.7 | 1.5 | 6.7 | 1.6 |
| Behavior and Symptom Identification Scale 32, daily living and role functioning score | 2.4 | 0.6 | 2.2 | 0.7 | 2.3 | 0.6 |
| Behavior and Symptom Identification Scale 32, relationship to self and others score | 2.5 | 0.6 | 2.2 | 0.7 | 2.3 | 0.7 |
| Mini-Mental State Examination score | 29.6 | 0.8 | 29.3 | 1.0 | 29.4 | 0.9 |
| Epworth Sleepiness Scale score | 7.9 | 5.0 | 8.5 | 4.7 | 8.2 | 4.9 |
TABLE 1. Baseline characteristics of participants in the Reducing Suicidal Ideation Through Insomnia Treatment study
Randomized Treatment
Eighty-six percent of participants assigned to zolpidem-CR completed all scheduled visits after randomization, compared with 73% of placebo participants. Overall, 90% of the scheduled visits were completed. Participants took 91% of all prescribed doses of the study drug and 94% of all prescribed doses of the SSRI, with no significant differences between groups (p=0.11 and p=0.29, respectively).
In the zolpidem-CR arm, 73% of patients received an increase in daily dose from 6.25 mg to 12.5 mg at some time, while 72% of the placebo arm received an increase (p=0.92). Also, 36% of participants in the zolpidem-CR arm received a doubling of their SSRI dosage, while 29% of those in the placebo arm received an increase (p=0.44).
Outcomes
Consistent with its known hypnotic effect, the zolpidem-CR group immediately showed more improvement in Insomnia Severity Index scores compared with the placebo group, with a significant advantage during the period of randomized treatment (Figure 2). On average, the least squares mean score on the Insomnia Severity Index over the 8-week period was 11.2 (SE=0.6) for the zolpidem-CR group and 13.8 (SE=0.6) for the placebo group (p=0.004). When the sample was split by severe insomnia (Insomnia Severity Index score >21, N=50) relative to mild to moderate insomnia (Insomnia Severity Index scores 8–21, N=53), the zolpidem-CR intervention provided no additional improvement in the scores for mild to moderate insomnia (mean=−0.14, SE=1.18; p>0.05). In contrast, for those participants with severe insomnia, the zolpidem-CR intervention produced a significant advantage in improvement in Insomnia Severity Index scores (mean=−5.23, SE=1.43; p<0.001). Among the participants who completed at least 50% of their sleep diaries, zolpidem-CR showed numerical advantages across 8 weeks for sleep latency (mean=−13.2 minutes, SE=8.0), wake after sleep onset (mean=−20.7 minutes, SE=11.5), and total sleep time (mean=12.8 minutes, SE=15.0).
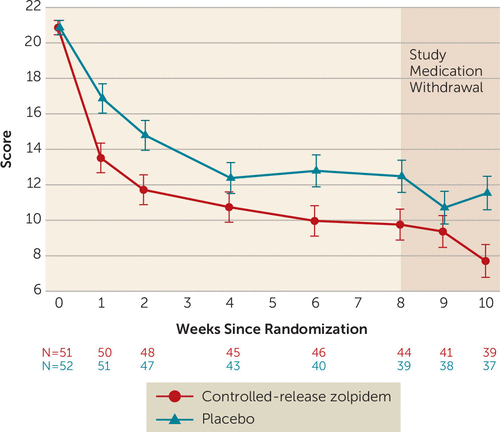
FIGURE 2. Least squares mean scores on the Insomnia Severity Index for participants in the Reducing Suicidal Ideation Through Insomnia Treatment studya
a Error bars indicate standard errors.
There was no significant effect of treatment on suicidal ideation as quantified by the Scale for Suicide Ideation (p=0.50). The least squares mean estimate of the treatment effect, as measured by the Scale for Suicide Ideation, was −0.56 (SE=0.83; 95% CI=−2.19, 1.08; Cohen’s d=−0.11), with no effect of site (Figure 3). However, we found a significant longitudinal linear association between scores on the Insomnia Severity Index and the Scale for Suicide Ideation across treatment that remained after adding a covariate for depression scores with the sleep and suicide items removed (beta=0.12, SE=0.04, p=0.002). There was a significant decrease in scores on the Scale for Suicide Ideation over time in both groups. At the end of randomized treatment, 61% of the zolpidem-CR patients and 57% of the placebo patients had achieved a score of 0 on the Scale for Suicide Ideation (59% overall). Past treatment with an antidepressant medication in the index episode of major depressive disorder was not a significant moderator of the effect of zolpidem-CR on scores on the Scale for Suicide Ideation.
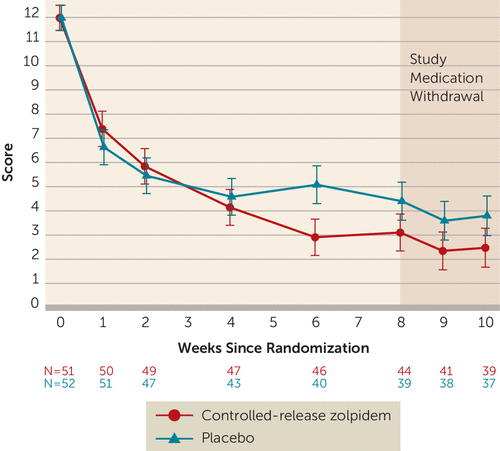
FIGURE 3. Least squares mean scores on the Scale for Suicide Ideation for participants in the Reducing Suicidal Ideation Through Insomnia Treatment studya
a Error bars indicate standard errors.
As a second indicator of suicidal ideation, the suicidal ideation score of the C-SSRS showed an advantage for zolpidem-CR (p=0.035). The least squares mean estimate of the treatment effect was −0.26 (SE=0.12) (95% CI=−0.50, −0.02; Cohen’s d=−0.26) (Figure 4). The zolpidem-CR intervention was associated with a numerically greater reduction in C-SSRS suicidal ideation scores in those participants with severe baseline insomnia (mean=−0.41, SE=0.21) relative to those with mild to moderate baseline insomnia (mean=−0.08, SE=0.15).
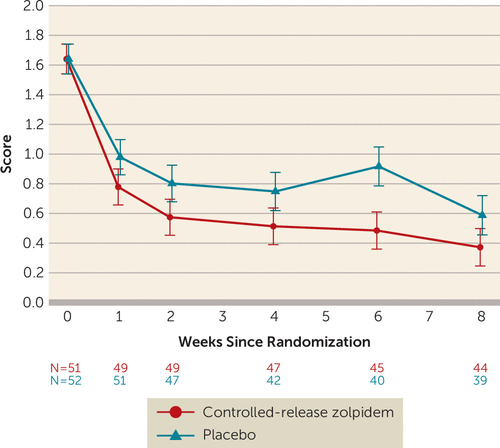
FIGURE 4. Least squares mean scores for suicidal ideation on the Columbia–Suicide Severity Rating Scale for participants in the Reducing Suicidal Ideation Through Insomnia Treatment studya
a Error bars indicate standard errors.
There were significant improvements over time in depression scores (p<0.001), but no significant differences between treatments were found. Scores on the CGI-S indicated significant improvement in both groups over time (p<0.001), but the advantage for the zolpidem-CR group fell short of significance (mean change=−0.25, 95% CI=−0.53, 0.02, p=0.07). Scores on the CGI-I also indicated improvement for both groups across time (p<0.001), but with a significant advantage for the zolpidem-CR group (mean change=−0.28, 95% CI=−0.52, −0.04, p=0.022) that remained after controlling for baseline CGI-S scores (mean change=−0.25, 95% CI=−0.49, −0.01, p<0.05). There were significant improvements over time in hopelessness scores (p<0.001), quality of life subscales (both p<0.001), dysfunctional beliefs and attitudes about sleep (p<0.001), and nightmares (p<0.001) overall. However, there were no differences between treatment groups for any of these secondary outcomes.
Follow-Up
Weekly telephone follow-up for the 2 weeks after the conclusion of randomized treatment showed no loss of effect, as measured by scores on the Insomnia Severity Index or the Scale for Suicide Ideation, in either group (Figure 2 and Figure 3).
Adverse Events
There were no deaths and no suicide attempts during any phase of the study. Pregnancy and subsequent miscarriage in one participant in the zolpidem-CR group was the sole serious adverse event observed, and it was judged not to be related to study participation. Regarding nonserious adverse events, 61% of the zolpidem-CR group and 71% of the placebo group experienced an adverse event, with the most common categories being cold symptoms and flu, nausea and diarrhea, and headache in descending rank order.
Discussion
This investigation found that the addition of zolpidem-CR to an SSRI was superior to placebo plus an SSRI in reducing insomnia symptoms in suicidal patients with major depressive disorder. This finding is similar to what we have described in nonsuicidal samples of patients with major depression and insomnia (38). The advantage produced by zolpidem-CR was found principally in those patients with severe insomnia at baseline. We did not find that adding zolpidem-CR to an SSRI provided an advantage in decreasing suicidal ideation scores on the Scale for Suicide Ideation, but we did find an advantage for zolpidem-CR in the suicide ideation score on the C-SSRS. As with the effect in insomnia scores, the effect of zolpidem-CR on C-SSRS suicide ideation scores was numerically greater in those patients with severe baseline insomnia. The clinical significance of the advantage seen for suicidal ideation scores on the C-SSRS in the zolpidem-CR group was modest (mean=−0.41, SE=0.21), even in the severe insomnia group, and this is consistent with a 50% decrease (half-point drop) between the item anchors “Have you actually had any thoughts of killing yourself?” and “Have you wished you were dead or wished you could go to sleep and not wake up?” (18, 24).
It is noteworthy that one measure of suicidal ideation (the Scale for Suicide Ideation) failed to show a treatment effect of zolpidem-CR, while another measure of suicidal ideation (the C-SSRS) did show an effect. However, there are no published studies on the relative performance of the two scales in randomized clinical trials, leaving no guidance on which measure should be preferred in randomized clinical trials of suicidality. We found a significant correlation between the two measures of intensity of suicidal ideation (see Table S1 in the online supplement), but the structure of the scoring is sufficiently different that the scales may be measuring different constructs of intensity. The improvements in suicidality and insomnia that were seen during the randomized phase persisted after the discontinuation of bedtime medication into the 2-week telephone follow-up. This finding is similar to what we reported with the addition of eszopiclone to SSRIs in depressed patients with insomnia (39).
Still, significant improvements in insomnia intensity and suicidal ideation were also seen in the fluoxetine plus placebo group. Although SSRIs are known to have adverse effects on objective sleep continuity (40, 41), patients nevertheless may report perceived improvements in insomnia even as the objective measures worsen (42). This may be related to a more positive appraisal of sleep as the depression lifts or may be related to placebo effects (43).
In our view, the safety findings of the study are as important as the efficacy findings. First, we found that it is possible to selectively recruit and safely retain suicidal outpatients with major depressive disorder in a pharmacotherapy randomized clinical trial and that the participants were highly adherent to the trial’s schedule of events (44). As a result, this study establishes methods that may be generalized to carry out double-blind randomized trials of treatments for suicidality in outpatients. Second, we detected no worsening of suicidal ideation or the emergence of suicidal behavior under the controlled circumstance of a randomized clinical trial, leading us to conclude that there are no reasons to systematically exclude suicidal outpatients from psychotropic randomized clinical trials (45).
The REST-IT trial has strengths and limitations. Strengths include the innovation of a sample entirely comprising patients with ongoing suicidal ideation (albeit without plan or intent) and with a relatively high proportion of minority participants, successful safety planning, and good retention. Limitations include the exclusion of suicidal patients with an active plan and imminent intent for suicide, as those patients would not be suitable for an outpatient study. Patients with bipolar disorder, primary psychotic disorders, and substance abuse were also excluded, as were patients older than age 65. The study did not include a specific measure of anxiety severity. The periods of randomized treatment and follow-up were short, and polysomnography was not an outcome measure. Also, because the C-SSRS was our second indicator of suicidality, if a Bonferroni correction were applied with alpha=0.025, then the C-SSRS finding would be judged as having fallen short of significance. The study did not compare a hypnotic against cognitive-behavioral therapy for insomnia (CBT-I), and CBT-I has proven helpful for insomnia in patients with depression (46). Indeed, an open-label study indicated that CBT-I was associated with reductions of suicidal ideation in depressed patients with insomnia (47). Clinical guidelines for chronic insomnia endorse CBT-I as a first-line treatment (48, 49). However, the onset of treatment effect of CBT-I is likely to be slower than that of a hypnotic, and the time to onset of effect may be an important consideration in the beginning phases of treatment for suicidal patients. In addition, the relationship between insomnia and suicidality must be understood in light of the possibility that underlying circadian dysfunction can explain the results (50, 51).
Conclusions
Insomnia symptoms are a driver of suicidal ideation (3). The REST-IT study shows that hypnotic medications are effective for insomnia in suicidal outpatients with major depressive disorder and that the resolution of suicidal ideation positively covaries with resolution of insomnia symptoms. Correspondingly, the addition of zolpidem-CR produced greater reductions in suicidality compared with placebo, as measured by the C-SSRS. Still, SSRI monotherapy was associated with some improvement in both insomnia and suicidality, and hence routine prescription of hypnotics as a means of mitigating suicidal ideation may not be necessary in all outpatients with major depression. However, our results do suggest that, with proper safety procedures, it is possible to provide short-term relief of insomnia and more rapid reduction of suicidal ideation by time-limited prescribing of small quantities of hypnotics, without incurring major risk of emergent suicidal ideation, especially in those suicidal outpatients with major depression with the most severe insomnia (11).
1 : Suicide trends among and within urbanization levels by sex, race/ethnicity, age group, and mechanism of death: United States, 2001–2015. MMWR Surveill Summ 2017; 66:1–16Crossref, Medline, Google Scholar
2 : The link between suicide and insomnia: theoretical mechanisms. Curr Psychiatry Rep 2013; 15:389Crossref, Medline, Google Scholar
3 : Insomnia symptoms drive changes in suicide ideation: a latent difference score model of community adults over a brief interval. J Abnorm Psychol 2017; 126:739–749Crossref, Medline, Google Scholar
4 : Insomnia severity is an indicator of suicidal ideation during a depression clinical trial. Sleep Med 2010; 11:822–827Crossref, Medline, Google Scholar
5 : Insomnia and daytime cognitive performance: a meta-analysis. Sleep Med Rev 2012; 16:83–94Crossref, Medline, Google Scholar
6 : Problem-solving in suicide attempters. Psychol Med 2004; 34:163–167Crossref, Medline, Google Scholar
7 : Neuropsychological function and suicidal behavior: attention control, memory and executive dysfunction in suicide attempt. Psychol Med 2013; 43:539–551Crossref, Medline, Google Scholar
8 : Attention deficit in depressed suicide attempters. Psychiatry Res 2008; 159:7–17Crossref, Medline, Google Scholar
9 : Neuropsychological dysfunction in depressed suicide attempters. Am J Psychiatry 2001; 158:735–741Link, Google Scholar
10 : Hypnotic medications and suicide: risk, mechanisms, mitigation, and the FDA. Am J Psychiatry 2017; 174:18–25Link, Google Scholar
11 : A multi-site randomized clinical trial to reduce suicidal ideation in suicidal adult outpatients with major depressive disorder: development of a methodology to enhance safety. Clin Trials 2015; 12:189–198Crossref, Medline, Google Scholar
12 Spitzer RL, Williams JBW, Gibbon M, et al: Structured Clinical Interview for DSM-IV (SCID). New York, New York State Psychiatric Institute, Biometrics Research, 1995Google Scholar
13 : Derivation of research diagnostic criteria for insomnia: report of an American Academy of Sleep Medicine work group. Sleep 2004; 27:1567–1596Crossref, Medline, Google Scholar
14 : A rating scale for depression. J Neurol Neurosurg Psychiatry 1960; 23:56–62Crossref, Medline, Google Scholar
15 : Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med 2001; 2:297–307Crossref, Medline, Google Scholar
16 : “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12:189–198Crossref, Medline, Google Scholar
17 : Assessment of suicidal intention: the Scale for Suicide Ideation. J Consult Clin Psychol 1979; 47:343–352Crossref, Medline, Google Scholar
18 : Risk factors for suicidal behavior: utility and limitations of research instruments, in Standardized Evaluation in Clinical Practice. Edited by First MB. Washington, DC, American Psychiatric Association Publishing, 2003, pp 103–130Google Scholar
19 : Prevalence of obstructive sleep apnea in suicidal patients with major depressive disorder. J Psychiatr Res 2019; 116:147–150Crossref, Medline, Google Scholar
20 Food and Drug Administration (FDA), US Department of Health and Human Services: FDA requiring lower recommended dose for certain sleep drugs containing zolpidem. 2013Google Scholar
21 : Psychometric characteristics of the Scale for Suicide Ideation with psychiatric outpatients. Behav Res Ther 1997; 35:1039–1046Crossref, Medline, Google Scholar
22 : Suicide ideation at its worst point: a predictor of eventual suicide in psychiatric outpatients. Suicide Life Threat Behav 1999; 29:1–9Medline, Google Scholar
23 : Risk factors for suicide in psychiatric outpatients: a 20-year prospective study. J Consult Clin Psychol 2000; 68:371–377Crossref, Medline, Google Scholar
24 : Columbia Classification Algorithm of Suicide Assessment (C-CASA): classification of suicidal events in the FDA’s pediatric suicidal risk analysis of antidepressants. Am J Psychiatry 2007; 164:1035–1043Link, Google Scholar
25 : A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 1991; 14:540–545Crossref, Medline, Google Scholar
26 : Sleep patterns and aging: comparison of older adults with and without insomnia complaints. Psychol Aging 1989; 4:290–294Crossref, Medline, Google Scholar
27 : Nightmares and dysfunctional beliefs about sleep mediate the effect of insomnia symptoms on suicidal ideation. J Clin Sleep Med 2013; 9:135–140Medline, Google Scholar
28 : Sleep dynamic therapy for Cerro Grande Fire evacuees with posttraumatic stress symptoms: a preliminary report. J Clin Psychiatry 2002; 63:673–684Crossref, Medline, Google Scholar
29 : Beck Hopelessness Scale Manual. New York, Psychological Corporation, 1988Google Scholar
30 : Use of the Beck Depression Inventory, hopelessness scale, scale for suicidal ideation, and suicidal intent scale with adolescents. Adv Adolesc Mental Health 1988; 3:219–231Google Scholar
31 : Relationship between hopelessness and ultimate suicide: a replication with psychiatric outpatients. Am J Psychiatry 1990; 147:190–195Link, Google Scholar
32 : Cognitive risk factors and suicide attempts among formerly hospitalized adolescents: a prospective naturalistic study. J Am Acad Child Adolesc Psychiatry 2001; 40:91–99Crossref, Medline, Google Scholar
33 : Individual risk factors for early repetition of deliberate self-harm. Br J Med Psychol 1997; 70:387–393Crossref, Medline, Google Scholar
34 : Reliability and validity of a brief patient-report instrument for psychiatric outcome evaluation. Hosp Community Psychiatry 1994; 45:242–247Abstract, Google Scholar
35 : ECDEU Assessment Manual for Psychopharmacology. Rockville, Md, US Department of Health, Education and Welfare, 1976Google Scholar
36 : Medication resistance and clinical response to electroconvulsive therapy. Psychiatry Res 1990; 31:287–296Crossref, Medline, Google Scholar
37 : A psychometric cut-point to separate emergently suicidal depressed patients from stable depressed outpatients. Indian J Psychiatry 2013; 55:283–286Crossref, Medline, Google Scholar
38 : Zolpidem for persistent insomnia in SSRI-treated depressed patients. J Clin Psychiatry 1999; 60:668–676Crossref, Medline, Google Scholar
39 : Evaluation of eszopiclone discontinuation after cotherapy with fluoxetine for insomnia with coexisting depression. J Clin Sleep Med 2007; 3:48–55Medline, Google Scholar
40 : Effects of paroxetine on human sleep. Br J Clin Pharmacol 1986; 22:97–99Medline, Google Scholar
41 : Fluoxetine-induced sleep disturbance in depressed patients. Neuropsychopharmacology 1996; 14:437–442Crossref, Medline, Google Scholar
42 : A comparison of nefazodone and fluoxetine on mood and on objective, subjective, and clinician-rated measures of sleep in depressed patients: a double-blind, 8-week clinical trial. J Clin Psychiatry 1997; 58:185–192Crossref, Medline, Google Scholar
43 : Placebo effects in primary insomnia. Sleep Med Rev 2005; 9:381–389Crossref, Medline, Google Scholar
44 : The effect of participating in suicide research: does participating in a research protocol on suicide and psychiatric symptoms increase suicide ideation and attempts? Suicide Life Threat Behav 2010; 40:535–543Crossref, Medline, Google Scholar
45 : Implications of zero suicide for suicide prevention research. JAMA 2018; 320:1633–1634Crossref, Medline, Google Scholar
46 : Efficacy of cognitive-behavior therapy for insomnia combined with antidepressant pharmacotherapy in patients with comorbid depression and insomnia: a randomized controlled trial. J Clin Psychiatry 2016; 77:e1316–e1323Crossref, Medline, Google Scholar
47 : Effects of cognitive behavioral therapy for insomnia on suicidal ideation in veterans. Sleep (Basel) 2015; 38:259–265Crossref, Medline, Google Scholar
48 : Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med 2016; 165:125–133Crossref, Medline, Google Scholar
49 : Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med 2017; 13:307–349Crossref, Medline, Google Scholar
50 : The relationship of person-specific eveningness chronotype, greater seasonality, and less rhythmicity to suicidal behavior: a literature review. J Affect Disord 2018; 227:721–730Crossref, Medline, Google Scholar
51 : A rest-activity biomarker to predict response to SSRIs in major depressive disorder. J Psychiatr Res 2015; 64:19–22Crossref, Medline, Google Scholar




