Brain Imaging of the Cortex in ADHD: A Coordinated Analysis of Large-Scale Clinical and Population-Based Samples
Abstract
Objective:
Neuroimaging studies show structural alterations of various brain regions in children and adults with attention deficit hyperactivity disorder (ADHD), although nonreplications are frequent. The authors sought to identify cortical characteristics related to ADHD using large-scale studies.
Methods:
Cortical thickness and surface area (based on the Desikan–Killiany atlas) were compared between case subjects with ADHD (N=2,246) and control subjects (N=1,934) for children, adolescents, and adults separately in ENIGMA-ADHD, a consortium of 36 centers. To assess familial effects on cortical measures, case subjects, unaffected siblings, and control subjects in the NeuroIMAGE study (N=506) were compared. Associations of the attention scale from the Child Behavior Checklist with cortical measures were determined in a pediatric population sample (Generation-R, N=2,707).
Results:
In the ENIGMA-ADHD sample, lower surface area values were found in children with ADHD, mainly in frontal, cingulate, and temporal regions; the largest significant effect was for total surface area (Cohen’s d=−0.21). Fusiform gyrus and temporal pole cortical thickness was also lower in children with ADHD. Neither surface area nor thickness differences were found in the adolescent or adult groups. Familial effects were seen for surface area in several regions. In an overlapping set of regions, surface area, but not thickness, was associated with attention problems in the Generation-R sample.
Conclusions:
Subtle differences in cortical surface area are widespread in children but not adolescents and adults with ADHD, confirming involvement of the frontal cortex and highlighting regions deserving further attention. Notably, the alterations behave like endophenotypes in families and are linked to ADHD symptoms in the population, extending evidence that ADHD behaves as a continuous trait in the population. Future longitudinal studies should clarify individual lifespan trajectories that lead to nonsignificant findings in adolescent and adult groups despite the presence of an ADHD diagnosis.
Attention deficit hyperactivity disorder (ADHD) is a common neuropsychiatric disorder characterized by age-inappropriate levels of inattention and/or hyperactivity and impulsivity. ADHD occurs in around 5%−7% of children and 2.5% of adults (1, 2). ADHD can have a negative effect on multiple aspects of daily life for patients, and it represents a major public health challenge (3). Neuroimaging studies in ADHD show differences between the brains of people with ADHD and those of healthy individuals in structure (4–9), function (8, 10, 11), and connectivity (12–14), albeit with small effect sizes (9). While informative, existing studies have several major limitations. First, most ADHD neuroimaging studies have been cross-sectional and were performed in children; studies that either consider ADHD throughout the lifespan or have a longitudinal design are rare. In one lifespan study, we recently showed that differences in intracranial volume and subcortical volumes between patients and healthy individuals were largely restricted to childhood (9). An earlier longitudinal study showed slower, delayed development of cortical thickness and surface area in children with ADHD, especially in frontal-temporal regions (15). Nonetheless, large-scale studies of cerebral cortical architecture throughout the lifespan are lacking.
A second major limitation in the neuroimaging literature is that most studies of ADHD have small sample sizes and show limited reproducibility (16). Combining data from existing research by means of meta- or mega-analysis can produce more reliable results. For ADHD, meta- or mega-analyses of structural brain phenotypes are available for subcortical structures (9, 17), but the cortex has only been assessed in meta-analyses of brain-wide voxel-based morphometry (VBM) studies (5–8). The largest VBM study (931 patients and 822 control subjects) reported case-control differences for the anterior cingulate, medial prefrontal cortex, ventromedial orbitofrontal cortex, and insula (8). Here, we further the research by providing the first large-scale mega-analytic examination of cortical measures across the lifespan in ADHD. We analyzed cortical surface area and thickness separately, as recent large-scale studies show that the biological mechanisms underlying such measures overlap only partially (18). Our large sample size also provides the statistical power needed to examine clinical factors such as common comorbid disorders.
Neuroimaging analyses of ADHD have also largely not addressed a major question: Are the observed brain differences a consequence of living with the disorder, or do the brain differences reflect underlying risk for the disorder? Different study designs can help us begin to address this question. Family-based studies can indicate whether cortical changes are present in unaffected siblings of case subjects to indicate the involvement of shared genetic and/or environmental risk factors that underlie the cortical characteristics associated with the disorder. Several family studies (e.g., 19) suggest that at least some of the brain alterations seen in patients are also present in their unaffected siblings and are associated with symptom severity in healthy individuals. Population-based studies can determine whether individuals with traits of ADHD show cortical differences similar to those associated with the full syndrome. The largest population study published to date (N=776 children) showed that higher levels of ADHD symptoms were associated with a thinner cortex in caudal middle frontal, temporal, and occipital regions (20). Although this and similar studies (21) showed that brain alterations extend beyond the clinical disorder, no attempts have yet been made to directly assess the overlap between studies in clinical samples and the general population. Combined, family- and population-based findings suggest that the brain differences seen in individuals with ADHD are not simply markers of the disorder, but larger studies directly comparing brain phenotypes across different informative study designs are needed to shed more light on this.
Here, we present a mega-analysis of cortical thickness and surface area in participants with ADHD and healthy control subjects across the lifespan from the ENIGMA-ADHD Working Group, a worldwide collaboration aiming to characterize the brain characteristics of people with ADHD. All partners used standardized methods (segmentation protocols and quality control procedures), limiting methodological heterogeneity more than in previous meta-analyses. In addition to assessing case-control differences in children, adolescents, and adults, we investigated cortical brain correlates of clinical features, assessed familiality of effects, and mapped the dimensionality of affected cortical regions in the large, independent pediatric Generation-R population study (22).
Methods
Contributing Studies
The ENIGMA-ADHD Working Group currently consists of 36 cohorts from around the world (http://enigma.ini.usc.edu/ongoing/enigma-adhd-working-group/). All cohorts have structural imaging data available for individuals with an ADHD diagnosis, and most sites also include data from healthy control subjects. An overview of the sites is provided in Table ST1 in the online supplement; details of image acquisition and study protocols are provided in Table ST2 and Appendix SA1 in the online supplement. The data set for the cortical analysis comprised 4,180 individuals: 2,246 people with ADHD (mean age, 19.22 years [SD=11.31, range=4–62]; 74.1% males) and 1,934 healthy control subjects (mean age, 18.05 years [SD=11.26, range=4–63]; 59.8% males).
For the analysis of dimensionally assessed ADHD traits in the general population, we used data from 2,707 individuals (mean age, 10.11 years [SD=0.57, range=8.5–11.9]; 49.4% males; see Table ST3 in the online supplement) from the Generation-R cohort (22).
For all participating cohorts, approval for the analysis was available from the responsible ethics committees.
Neuroimaging
Structural T1-weighted brain MRI data were acquired and processed at the individual sites. The images were analyzed using standardized protocols to harmonize analysis and quality control processes (see http://enigma.ini.usc.edu/protocols/imaging-protocols/; see also Appendix SA2 in the online supplement) (23–25). Fully automated and validated neuroimaging segmentation algorithms based on FreeSurfer, version 5.1 or 5.3, were used (see Table ST2 in the online supplement). Regions based on the Desikan-Killiany atlas were segmented, which resulted in cortical thickness and surface area values for 34 left and 34 right hemisphere regions. Two whole-hemisphere values for average thickness and average surface area were also computed. For further analysis, we used the mean of the right and left values.
The Generation-R data were collected using a single, study-dedicated MRI scanner and processed using FreeSurfer, version 6.0, on a high-performance computing system (Cartesius, surfsara.nl) (for the scanner sequence, see Appendix SA3 in the online supplement). All imaging data were visually inspected for inaccuracies in the surface-based reconstruction. Data not suitable for analysis were excluded (for a flowchart, see Figure SF1 in the online supplement), resulting in an N of 2,707. For a nonresponse analysis, see Appendix SA4 in the online supplement.
Case-Control Differences in Cortical Thickness and Surface Area in Children, Adolescents, and Adults
Based on the age specificity of earlier findings (9), three age groups were assessed: children ages 4–14 (1,081 case subjects and 1,048 control subjects), adolescents ages 15–21 (432 case subjects and 347 control subjects), and adults ages 22–63 (733 case subjects and 539 control subjects). Because there are marked developmental changes across the 4- to 14-year age range, we also performed supplemental analyses on age tertiles of the childhood group. For each of the age groups, we determined differences between participants with ADHD and healthy control subjects using mixed-effect models with site as a random factor in the nlme package in R. Age and sex were included as additional covariates; for the surface area analysis, intracranial volume (ICV) was also added, because surface area scales with head size (24–26). We also included analyses without ICV as a covariate given the debate over whether it should be included or not (see Appendix SA5 in the online supplement). To calculate Cohen’s d effect size estimates, adjusting for the appropriate covariates, we used the t-statistic from the diagnosis (ADHD=1, control=0) predictor in the equation (27). To correct for multiple comparisons, we used a false discovery rate at q=0.05.
Split-Half Validation of Case-Control Findings
To ensure stability of effects, we performed a validation of our mega-analysis in age groups with significant results. The data were split into two halves, statistically matched for age, sex, and ICV within each site. Validation was defined as a false-discovery-rate-corrected p<0.05 in the first half and an uncorrected p<0.05 in the second half, with matching effect directions (28).
Exploration of the Influence of Sex, IQ, and Clinical Factors on Cortical Regions Affected in ADHD
For regions and age groups showing validated case-control differences, we examined potential effects of sex, IQ, comorbid disorders, medication use, and ADHD symptoms (severity) (for details, see Appendix SA6 in the online supplement). Given the exploratory nature of these analyses, we report uncorrected p values in the Results section.
Family Study
Two subsets of the ENIGMA-ADHD sample (NeuroIMAGE Amsterdam and Nijmegen [29]) collected brain data from patients (N=211), their unaffected siblings (N=175), and unrelated control subjects (N=120). To determine familial effects on ADHD-affected cortical regions, unaffected siblings were compared with healthy control subjects in those cortical regions. Levels of ADHD symptoms in the unaffected siblings had been shown not to differ from those of control subjects (19). Correction for multiple comparisons was performed based on the effective number of independent tests (Meff) (30); differences between unaffected siblings and control subjects were considered significant at p<0.01 (Meff=5; for details, see Appendix SA7 in the online supplement).
Association Between ADHD Symptoms and the Cortex in the General Population
ADHD symptoms were assessed in children from Generation-R using the Child Behavior Checklist (CBCL) (31). Both attention problems (on the CBCL attention problems syndrome scale) and ADHD problems (on the CBCL DSM-oriented scale) were examined for associations with surface area or thickness in regions with validated case-control differences in ENIGMA-ADHD. The R package, version 3.3.3, was used to fit multiple linear regressions to model these associations. Primary analyses were adjusted for age at MRI scan, sex, ICV, and ethnicity. In supplemental analyses, models were additionally adjusted for nonverbal IQ, ADHD medication status, MRI scanner software version, and motion during scanning (see Appendix SA8 in the online supplement).
Results
Case-Control Differences in Cortical Surface Area and Thickness in Children, Adolescents, and Adults
In children with ADHD compared with control children, lower values for cortical surface area were widespread, with 24 of 34 regions and total surface area being smaller in the ADHD group (Table 1, Figure 1; see also Table ST4 in the online supplement). The largest effect was found for total surface area with a Cohen’s d of −0.21 (false-discovery-rate-corrected p≤0.001). When the child group was further subdivided in post hoc analyses, this effect size increased to −0.35 (false-discovery-rate-corrected p≤0.001) in the youngest tertile (ages 4–9 years), which comprised 317 case subjects and 340 control subjects (see Table ST5 in the online supplement). More generally, the youngest group showed the largest case-control differences (see Table ST5). No case-control differences were found in the adolescent and adult groups (see Tables ST6 and ST7 in the online supplement; Table ST8 shows the combined analysis of age groups. For results of the model without ICV, see Table ST9).
| Cortical Surface Area | Control Group (N) | ADHD Group (N) | Cohen’s d | SE | 95% CI | p | p (FDR corrected) |
|---|---|---|---|---|---|---|---|
| Total surface areab | 1,048 | 1,081 | –0.21 | 0.04 | –0.29, –0.12 | <0.001 | <0.001 |
| Superior frontal gyrusb | 1,044 | 1,074 | –0.19 | 0.04 | –0.28, –0.11 | <0.001 | <0.001 |
| Lateral orbitofrontal cortexb | 1,047 | 1,081 | –0.17 | 0.04 | –0.26, –0.09 | <0.001 | <0.001 |
| Medial orbitofrontal cortex | 1,039 | 1,070 | –0.16 | 0.04 | –0.24, –0.07 | <0.001 | 0.002 |
| Posterior cingulate cortexb | 1,042 | 1,078 | –0.16 | 0.04 | –0.25, –0.08 | <0.001 | 0.002 |
| Rostral anterior cingulate cortexb | 1,041 | 1,067 | –0.16 | 0.04 | –0.25, –0.08 | <0.001 | 0.002 |
| Superior temporal gyrus | 987 | 993 | –0.15 | 0.05 | –0.24, –0.07 | <0.001 | 0.003 |
| Caudal middle frontal gyrusb | 1,046 | 1,077 | –0.15 | 0.04 | –0.23, –0.06 | <0.001 | 0.003 |
| Fusiform gyrus | 1,043 | 1,075 | –0.13 | 0.04 | –0.21, –0.04 | 0.004 | 0.01 |
| Isthmus cingulate cortex | 1,040 | 1,079 | –0.13 | 0.04 | –0.22, –0.05 | 0.002 | 0.008 |
| Middle temporal gyrusb | 1,001 | 1,024 | –0.13 | 0.04 | –0.22, –0.04 | 0.004 | 0.01 |
| Rostral middle frontal gyrus | 1,044 | 1,079 | –0.13 | 0.04 | –0.21, –0.04 | 0.004 | 0.01 |
| Supramarginal gyrus | 1,036 | 1,063 | –0.13 | 0.04 | –0.22, –0.05 | 0.002 | 0.008 |
| Inferior parietal cortex | 1,041 | 1,078 | –0.12 | 0.04 | –0.20, –0.03 | 0.009 | 0.02 |
| Inferior temporal gyrus | 1,041 | 1,064 | –0.12 | 0.04 | –0.21, –0.04 | 0.005 | 0.01 |
| Lateral occipital cortex | 1,047 | 1,078 | –0.12 | 0.04 | –0.21, –0.04 | 0.005 | 0.01 |
| Precuneus | 1,044 | 1,080 | –0.12 | 0.04 | –0.20, –0.03 | 0.008 | 0.02 |
| Superior parietal cortex | 1,045 | 1,073 | –0.12 | 0.04 | –0.21, –0.04 | 0.004 | 0.01 |
| Insula | 1,042 | 1,078 | –0.12 | 0.04 | –0.21, –0.04 | 0.006 | 0.01 |
| Banks of superior temporal sulcus | 974 | 999 | –0.10 | 0.05 | –0.19, –0.01 | 0.02 | 0.04 |
| Pars triangularis of inferior frontal gyrus | 1,048 | 1,074 | –0.10 | 0.04 | –0.18, –0.01 | 0.02 | 0.04 |
| Postcentral gyrus | 1,032 | 1,060 | –0.10 | 0.04 | –0.18, –0.01 | 0.03 | 0.05 |
| Precentral gyrus | 1,041 | 1,064 | –0.10 | 0.04 | –0.19, –0.02 | 0.02 | 0.03 |
| Temporal pole | 1,043 | 1,075 | –0.10 | 0.04 | –0.18, –0.01 | 0.03 | 0.04 |
TABLE 1. Mega-analysis of case-control cortical surface area differences in children age 14 and younger in ENIGMA-ADHDa
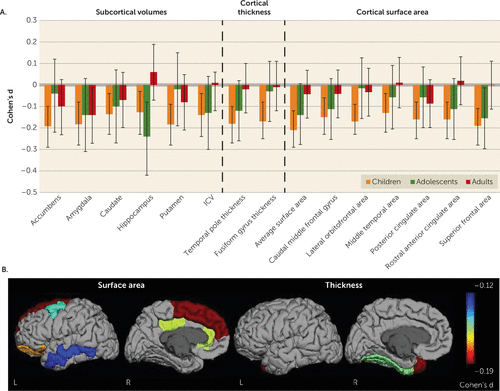
FIGURE 1. Subcortical and cortical brain differences across the lifespan between patients with ADHD and control subjectsa
a ICV=intracranial volume. In panel A, Cohen’s d effect sizes are shown, with 95% confidence intervals, for case-control differences in ENIGMA-ADHD cortical and subcortical structural features stratified by three age groups: children up to age 14, adolescents from age 15 to 21, and adults older than age 21. Structural features of all regions listed on the x-axis showed significant case-control differences in children; in analyses of cortical and subcortical features, no significant effects were seen in adolescents or adults. This is reflected in the effects sizes shown, all of which reached case-control statistical significance for children but not for the adolescent and adult groups, except for the hippocampus, which shows a significant case-control difference in the adolescent group as well. Panel B shows the heat maps of validated case-control differences in the childhood subset for both surface area and thickness in each hemisphere. (The graphs for the subcortical volumes in panel A are adapted from reference 9, used with permission from Elsevier.)
Cortical thickness was affected in four regions (the fusiform, parahippocampal, and precentral gyri and the temporal pole) in children, all being thinner in the children with ADHD than in the control children (Table 2, Figure 1; see also Table ST10 in the online supplement). Further subdivision of the child group retained significant effects for the fusiform gyrus (d=−0.31, false-discovery-rate-corrected p=0.002) and the temporal pole (d=−0.25, false-discovery-rate-corrected p=0.02) in the group of children ages 10 and 11 (356 case subjects, 365 control subjects); in younger (4–9 years) and older (12–14 years) children, effects did not survive correction for multiple comparisons (see Table ST11 in the online supplement). In adolescents and adults, no case-control differences were found (see Tables ST12 and ST13 in the online supplement; Table ST14 shows the combined analysis of age groups).
| Region | Control Group (N) | ADHD Group (N) | Cohen’s d | SE | 95% CI | p | p (FDR corrected) |
|---|---|---|---|---|---|---|---|
| Temporal poleb | 1,042 | 1,075 | –0.18 | 0.04 | –0.27, –0.10 | <0.001 | 0.001 |
| Fusiform gyrusb | 1,044 | 1,077 | –0.17 | 0.04 | –0.25, –0.08 | <0.001 | 0.003 |
| Precentral gyrus | 1,040 | 1,064 | –0.16 | 0.04 | –0.25, –0.07 | <0.001 | 0.003 |
| Parahippocampal gyrus | 1,041 | 1,076 | –0.15 | 0.04 | –0.23, –0.06 | <0.001 | 0.008 |
TABLE 2. Mega-analysis of case-control cortical thickness differences in children age 14 and younger in ENIGMA-ADHDa
Validation of Case-Control Findings
The split-half validation analysis showed seven regions for surface area and two regions for thickness to be significant in both halves (Tables 1 and 2 and Figure 1; see also Tables ST15 and ST16 in the online supplement). For all other regions, the direction of effects was the same in both split halves.
Effect sizes of the validated cortical differences across the age groups are plotted in Figure 1, together with the effect sizes of subcortical brain volumes from our earlier work (9). A post hoc analysis with the interaction of age group by diagnosis added to the main model indicated differences in effect sizes across the lifespan for surface area of the superior frontal gyrus and thickness of the fusiform gyrus (see Table ST17 in the online supplement).
Exploration of Effects of Sex, IQ, Comorbidity, Psychostimulant Medication, and ADHD Severity
Extending the main findings, we investigated several factors linked to ADHD that have been shown to influence brain volume in their own right. No significant interaction effects of diagnosis by sex were found (see Table ST18 in the online supplement). Correcting for IQ in surface area analyses produced only minor changes in the level of significance in the case-control comparisons. In all thickness analyses, IQ was a nonsignificant contributor (see Table ST19 in the online supplement).
For comorbidity analyses, we had information on case subjects of the childhood subset (N=1,081) available (comorbidity ever versus never, lifetime) for almost 50% of participants (see Table ST20 in the online supplement). In total, 194 children with ADHD (39%) were ever or currently diagnosed with a comorbid psychiatric disorder. The three most frequently co-occurring disorders were oppositional defiant disorder (in 79 case subjects [16.0%]), anxiety disorders (in 39 case subjects [8.6%]), and mood disorders (in 13 case subjects [3.0%]). Presence or absence of comorbid disorders was not related to cortical surface area; a nominal effect of ever being diagnosed with a comorbid psychiatric disorder was found for fusiform gyrus thickness, with a thinner fusiform gyrus in case subjects who had an additional current or past disorder (see Table ST21 in the online supplement).
Current stimulant use versus no current use had a nominally significant association with surface area of two regions in the frontal cortex, with children taking medication having lower surface areas (see Table ST21 in the online supplement).
Severity ratings for hyperactivity/impulsivity on the Conners questionnaires (available for 240 childhood patients), but not for inattention, showed nominally significant correlation with surface area in the rostral anterior cingulate cortex (r=−0.18, p=0.01), the superior frontal gyrus (r=−0.19, p=0.01), and total surface area (r=−0.15, p=0.03) (see Table ST22 in the online supplement).
Family Study
Among the validated ADHD-associated cortical features, surface area of the caudal middle frontal, lateral orbital frontal, and superior frontal gyrus and total surface area were significantly smaller in the unaffected siblings compared with control subjects (Figure 2; see also Table ST23 in the online supplement), indicating familial effects. A similar trend was seen for the majority of the other cortical measures (see Figure SF2 in the online supplement).
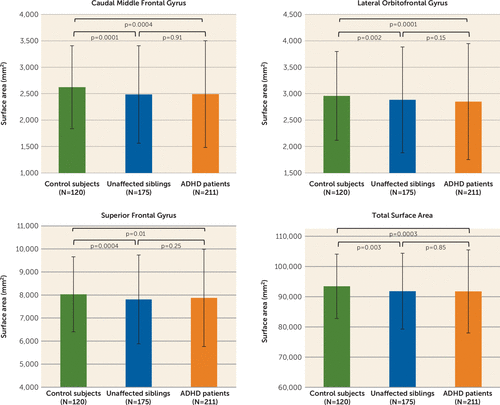
FIGURE 2. Results of familiality analyses, comparing healthy control subjects, unaffected siblings, and case subjects, in the ADHD-affected cortical regions in the NeuroIMAGE data sets (N=506)a
a Displayed are the cortical surface areas showing effects of familiality in the NeuroIMAGE data sets. For these regions, unaffected siblings differed from healthy control subjects (correction for multiple comparisons was performed based on the effective number of independent tests). Cortical values are adjusted for age, sex, and site. Error bars indicate standard deviation.
Effects of ADHD Symptoms in the General Population on the Validated Brain Phenotypes
Population-based analysis showed the caudal middle frontal gyrus, the middle temporal gyrus, and total surface area to be associated with the attention problems scale of the CBCL (Table 3; see also Figure SF3 in the online supplement); higher levels of dimensional ADHD symptoms were associated with smaller surface areas. No associations were found with the two cortical thickness measures (Table 3). To ensure that a linear fit was optimal and that the more severe end of the symptom continuum was not driving findings, models with quadratic and cubic symptom terms were also tested. Akaike and Bayesian information criteria values were highly similar across models, suggesting little or no improvement over the simpler linear term (see Table ST24 in the online supplement).
| Region | B | SE | 95% CI | β | p | FDR p |
|---|---|---|---|---|---|---|
| Surface area | ||||||
| Caudal middle frontal gyrus | –14.10 | 5.49 | –24.87, –3.33 | –0.04 | 0.01 | 0.03 |
| Lateral orbitofrontal cortex | –8.28 | 5.01 | –18.10, 1.54 | –0.02 | 0.10 | 0.11 |
| Middle temporal gyrus | –13.63 | 5.86 | –25.12, –2.14 | –0.03 | 0.02 | 0.04 |
| Posterior cingulate cortex | –5.02 | 2.42 | –9.77, –0.27 | –0.03 | 0.04 | 0.06 |
| Rostral anterior cingulate cortex | –3.50 | 1.93 | –7.29, 0.29 | –0.03 | 0.07 | 0.09 |
| Superior frontal gyrus | –7.16 | 11.93 | –30.55, 16.24 | –0.01 | 0.55 | 0.55 |
| Total surface area | –323.79 | 77.50 | –475.75, –171.82 | –0.04 | <0.001 | <0.001 |
| Total surface area (residualizedb) | –291.62 | 77.43 | –443.44, –139.79 | –0.07 | <0.001 | <0.001 |
| Thickness | ||||||
| Fusiform gyrus | 0.004 | 0.002 | 0.000, 0.01 | 0.04 | 0.05 | 0.054 |
| Temporal pole | 0.01 | 0.01 | –0.001, 0.03 | 0.04 | 0.07 | 0.07 |
TABLE 3. Associations between validated cortical regions and CBCL attention problems syndrome scale in Generation-Ra
Adding nonverbal IQ or ADHD medication status to the analysis model of the attention problems did not affect the results (see Table ST25 in the online supplement). The results also remained stable when we tested the effect of MRI scanner software version and image quality (see Table ST25). The quantitative amount of motion in the T1-weighted scan (32) did not seem to affect the analyses (see Table ST26 in the online supplement).
Discussion
Here we report the largest study to date of ADHD and cortical surface area and thickness in clinical samples and a pediatric population sample. Compared with healthy control subjects, children with ADHD showed smaller surface area in frontal, temporal, and cingulate regions, with the effects being most prominent in the youngest children (ages 4–9). Case-control differences had small effect sizes but survived validation. Differences in cortical thickness were limited to the temporal pole and the fusiform gyrus, which were thinner in children with ADHD. These differences were most prominent in the group of children 10 and 11 years old. The influence of comorbidity and symptom ratings, available from subsamples, appeared limited. None of these covariates of interest showed effects that survived correction for multiple testing. There were no significant associations between cortical alterations and either stimulant treatment or IQ. Family-based analyses revealed familial effects for four surface area regions but not for any of the thickness measures. A set overlapping with family-based analyses (caudal middle frontal gyrus, total surface area) and/or severity rating analyses (total surface area) showed associations with CBCL-based ratings of attention problems in the population-based sample; no such effects were found for thickness.
The regions affected in ADHD were widespread across the cortex. The frontal cortex differences in orbital, middle, and superior regions nicely confirm earlier work (e.g., 8, 15). These regions play a key role in cognitive processes related to reward and punishment, emotional processing, response inhibition, and attention—all known to be deficient in ADHD (33–35). Few studies have yet implicated structural differences in the cingulate cortex, an important structure linked to executive functioning and emotion (36), in ADHD (7, 37). Findings for the temporal cortex are particularly interesting, because both surface area and thickness were affected. The functions of this region are diverse, as it seems to be involved in semantic memory and processing of abstract concepts, attention, and emotion processing and regulation (38). Integrating the present findings with our earlier subcortical results (9), the multitude of findings for brain regions involved in emotion processing is intriguing. In view of this, the network of orbitofrontal cortex, cingulate, and amygdala could be particularly interesting for future research (39, 40), as it may underlie the deficient emotional self-regulation often observed in ADHD patients (33).
Effect sizes of the observed brain differences were small, which is at a level similar to our earlier findings for subcortical volumes and ICV in ADHD (Figure 1) and comparable to effect sizes seen in other psychiatric disorders studied within the ENIGMA consortium (23, 24). Whether this reflects phenotypic heterogeneity, with only a subgroup of patients showing reduced brain structure of large(r) effect size, or homogeneously small effects existing in the majority of patients, remains to be investigated. Effects were not driven by IQ. Findings in several areas seemed to scale with the severity of hyperactivity/impulsivity in patients, but the heterogeneity of assessment instruments limited the power of this analysis. As in our earlier analysis of subcortical volumes and ICV, we did not find any significant associations between psychostimulant medication and cortical dimensions, either in case-control or in population-based designs. However, given our observational design and reliance on legacy data, we would not want to draw any firm conclusions from those results.
Looking across the lifespan, all case-control differences were most pronounced in children and nonsignificant in adolescents and adults. The same phenomenon, albeit attenuated, was seen in our recent cross-sectional study of ICV and subcortical structures (9) (Figure 1). Post hoc analysis of potential differences in effect sizes across the three age groups in the present study confirmed age-related attenuation of effects for several structures. Those findings are in line with an earlier longitudinal study in which case-control differences in cortical thickness observed in children attenuated with increasing age, suggesting a delayed cortical maturation (41). An alternative explanation for the age-related differences may be the existence of subgroups; the childhood patient group is likely to consist of a mix of individuals in whom ADHD will persist and remit in adulthood, while the adult group consists largely of individuals with persistent ADHD. We cannot yet rule out low power as a reason for not detecting significant effects in the older subgroups, which were half the size of the children’s group, and these initial findings concerning apparent differences across the lifespan should be confirmed in longitudinal studies.
The case-control differences observed in the childhood sample did not seem to be influenced by comorbidity. However, we noticed that the comorbidity rate in this subset was relatively low (39%). There could be several reasons for that. First, the sample we used in our analysis of comorbidity was very young (4–14 years), as we focused only on the subsample with significant case-control differences. The relatively young age could explain the lower than expected comorbidity rate, as children may simply not yet have developed some of the frequent comorbid psychiatric disorders (e.g., substance use disorders). In comparison, Taurines et al. (42) reported in their review that 73% of 6- to 18-year-olds with ADHD had one or more comorbid disorders. A second reason could lie in the fact that we are dealing with research diagnoses, in which comorbidity assessments were often limited to checking inclusion and exclusion criteria for a specific study aim. This is a clear limitation of dealing with legacy data from multiple sites using different protocols and different instruments for assessing comorbidity and symptom severity. We adjusted our design accordingly and concentrated only on the three most frequent comorbidities, defining those as ever or never experienced.
Although our study was not designed to assess causality, our results may shed some light on the issue of whether brain differences are a consequence of living with the disorder or a risk factor for the disorder. Our family analysis showed that in unaffected siblings of case subjects—that is, those without a diagnosis and with levels of ADHD symptoms comparable to healthy control subjects—surface area differences from control subjects were similar to those of their affected siblings. In addition, the relationship between ADHD symptoms and cortical phenotypes also held in the general population. Here, the dimensional assessment of attention problems was related to brain morphology in a linear fashion, suggesting that the phenotype and underlying brain morphology are independent of clinical diagnosis, operating along a continuum. The two different approaches show cortical alterations in ADHD-related regions to occur independently of diagnosis, indicating that they are neither necessary nor sufficient to cause the disorder. The overlap between the findings from the different approaches was not complete, however. Future studies could perform more direct comparisons between case-control and population samples using, for example, conjunction analysis (43). In such a design, it would be interesting to test the liability-threshold model to better understand which factors contribute to liability for the disorder. Also, whether the observed brain differences relative to control subjects are indeed risk factors for ADHD remains to be investigated in prospective longitudinal designs. Future imaging genetics studies might further clarify the neurobiological pathways and mechanisms underlying cortical differences in ADHD. While genetic information is not available for sufficient numbers of participants from ENIGMA-ADHD, the ENIGMA Genetics Working Group recently identified genetic factors determining cortical surface area and thickness in a largely healthy population (18). Those genetic factors may in turn constitute risk factors for ADHD given recent findings of genetic overlap between the genetic contribution to ADHD and to the total surface area of the cortex. As we showed recently for subcortical volumes and intracranial volume, further work may delineate the individual genes or gene networks underlying such genetic overlap (44).
The present study has several strengths and limitations. Its major strength lies in the large sizes of both the clinical (N=4,180) and population-based (N=2,707) samples, along with the use of harmonized segmentation protocols, which provided unprecedented power to detect effects. Another strength is the split-half validation, combined with stringent multiple comparison correction, showing that our findings—despite small effect sizes—are stable. Also, results from the population study suggest little effect of motion during scanning on our cortical regions of interest. The combination of case-control with family- and population-based designs to identify mechanisms is an additional strength. A limitation is that we relied on legacy data in ENIGMA-ADHD, so the participating studies differ somewhat in their aims, methods, and assessments. Given this heterogeneity, our findings may underestimate the true effects, and we may have missed effects of comorbidity, medication, and symptom severity because of insufficient power. The limited sample size of the family study and the small effect sizes for brain differences are probably the reason the expected staircase effect was found in the family study but fell short of statistical significance.
In light of the findings from this study and the earlier ENIGMA study of ADHD (9), what should future neuroimaging studies in ADHD look like? The effect sizes observed are small (Cohen’s d=−0.21), with the largest effects for measures of total brain volume and surface area in this and our previous study (9). Also, effects are restricted to childhood despite persistent ADHD diagnosis in adolescents and adults. Future studies should answer the question of whether (regional) effect sizes are comparable in everyone, or whether subgroups exist in which certain regional effect sizes are more pronounced. This could be examined using clustering algorithms, such as community detection, and machine learning (45). An analysis of particular interest would be the comparison between children whose ADHD remits in adulthood and those whose ADHD persists. In-depth analysis of adults with persisting ADHD compared with adults whose disorder remits could add to our understanding of the null findings in adults, as it seems counterintuitive that the adults with persistent ADHD, believed to be more severely affected, show no apparent signs of brain differences in adulthood, but the childhood group, which is likely to contain a mix of individuals whose illness remits and persists in adulthood, does. Subgroups may also provide information on comorbidity and links to symptom severity in the different behavioral domains of ADHD. Most importantly, longitudinal studies are needed to identify the processes that lead to the apparent reductions of case-control effects from childhood to adolescence and adulthood; only a few longitudinal samples for ADHD are currently available (15, 29). We should also not forget that the segmentation used in the present study is based on classical neuroanatomical divisions rather than a partitioning based on biological functions (44, 46). Other cortical phenotypes, such as gyrification (47) or more sophisticated methods to define regional gray matter structure, and analyses of other brain measures to be captured by neuroimaging in large sample sizes (e.g., white matter integrity [48], resting-state functional MRI [49]) may help us find the presumed case-control differences in adults (50, 51).
In conclusion, we identify here, for the first time, cortical phenotypes affected in ADHD that are robust, and we show an association with ADHD beyond narrowly defined clinical diagnoses. Our work suggests that these phenotypes behave as endophenotypes and thus extends the evidence for ADHD as a continuous trait in the population, previously shown for behavioral measures and genetics (52) and now for neuroimaging phenotypes. Future studies should clarify individual lifespan trajectories and identify the underlying genetic and environmental factors shaping these trajectories.
1 : Attention-deficit/hyperactivity disorder. Nat Rev Dis Primers 2015; 1:15020Crossref, Medline, Google Scholar
2 : Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics 2015; 135:e994–e1001Crossref, Medline, Google Scholar
3 : Economic impact of childhood/adolescent ADHD in a European setting: the Netherlands as a reference case. Eur Child Adolesc Psychiatry 2014; 23:587–598Crossref, Medline, Google Scholar
4 : Meta-analysis of structural imaging findings in attention-deficit/hyperactivity disorder. Biol Psychiatry 2007; 61:1361–1369Crossref, Medline, Google Scholar
5 : Structural brain change in attention deficit hyperactivity disorder identified by meta-analysis. BMC Psychiatry 2008; 8:51Crossref, Medline, Google Scholar
6 : Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivity disorder indicates treatment effects. Acta Psychiatr Scand 2012; 125:114–126Crossref, Medline, Google Scholar
7 : Gray matter volume abnormalities in ADHD: voxel-based meta-analysis exploring the effects of age and stimulant medication. Am J Psychiatry 2011; 168:1154–1163Link, Google Scholar
8 : Structural and functional brain abnormalities in attention-deficit/hyperactivity disorder and obsessive-compulsive disorder: a comparative meta-analysis. JAMA Psychiatry 2016; 73:815–825Crossref, Medline, Google Scholar
9 : Subcortical brain volume differences in participants with attention deficit hyperactivity disorder in children and adults: a cross-sectional mega-analysis. Lancet Psychiatry 2017; 4:310–319Crossref, Medline, Google Scholar
10 : Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder: exploring task-specific, stimulant medication, and age effects. JAMA Psychiatry 2013; 70:185–198Crossref, Medline, Google Scholar
11 : Ventral-striatal responsiveness during reward anticipation in ADHD and its relation to trait impulsivity in the healthy population: a meta-analytic review of the fMRI literature. Neurosci Biobehav Rev 2014; 38:125–134Crossref, Medline, Google Scholar
12 : Research review: Diffusion tensor imaging studies of attention-deficit/hyperactivity disorder: meta-analyses and reflections on head motion. J Child Psychol Psychiatry 2018; 59:193–202Crossref, Medline, Google Scholar
13 : Functional decoding and meta-analytic connectivity modeling in adult attention-deficit/hyperactivity disorder. Biol Psychiatry 2016; 80:896–904Crossref, Medline, Google Scholar
14 : A systematic review and meta-analysis of tract-based spatial statistics studies regarding attention-deficit/hyperactivity disorder. Neurosci Biobehav Rev 2016; 68:838–847Crossref, Medline, Google Scholar
15 : Trajectories of cerebral cortical development in childhood and adolescence and adult attention-deficit/hyperactivity disorder. Biol Psychiatry 2013; 74:599–606Crossref, Medline, Google Scholar
16 : Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci 2013; 14:365–376Crossref, Medline, Google Scholar
17 : Corpus callosum morphology in children and adolescents with attention deficit hyperactivity disorder: a meta-analytic review. Neuropsychology 2008; 22:341–349Crossref, Medline, Google Scholar
18 : The genetic architecture of the human cerebral cortex. bioRxiv, September 3, 2018 (doi: https://doi.org/10.1101/399402)Google Scholar
19 : Voxel-based morphometry analysis reveals frontal brain differences in participants with ADHD and their unaffected siblings. J Psychiatry Neurosci 2016; 41:272–279Crossref, Medline, Google Scholar
20 : Cortical thickness and inattention/hyperactivity symptoms in young children: a population-based study. Psychol Med 2014; 44:3203–3213Crossref, Medline, Google Scholar
21 : Cortical development in typically developing children with symptoms of hyperactivity and impulsivity: support for a dimensional view of attention deficit hyperactivity disorder. Am J Psychiatry 2011; 168:143–151Link, Google Scholar
22 : Paediatric population neuroimaging and the Generation R Study: the second wave. Eur J Epidemiol 2018; 33:99–125Crossref, Medline, Google Scholar
23 : Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol Psychiatry 2017; 22:900–909Crossref, Medline, Google Scholar
24 : Cortical abnormalities associated with pediatric and adult obsessive-compulsive disorder: findings from the ENIGMA Obsessive-Compulsive Disorder Working Group. Am J Psychiatry 2018; 175:453–462Link, Google Scholar
25 : Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry 2018; 23:932–942Crossref, Medline, Google Scholar
26 : Head size, age, and gender adjustment in MRI studies: a necessary nuisance? Neuroimage 2010; 53:1244–1255Crossref, Medline, Google Scholar
27 : Effect size, confidence interval, and statistical significance: a practical guide for biologists. Biol Rev Camb Philos Soc 2007; 82:591–605Crossref, Medline, Google Scholar
28 : Mega-analysis of gray matter volume in substance dependence: general and substance-specific regional effects. Am J Psychiatry 2019; 176:119–128Link, Google Scholar
29 : The NeuroIMAGE study: a prospective phenotypic, cognitive, genetic and MRI study in children with attention-deficit/hyperactivity disorder: design and descriptives. Eur Child Adolesc Psychiatry 2015; 24:265–281Crossref, Medline, Google Scholar
30 : Adjusting multiple testing in multilocus analyses using the eigenvalues of a correlation matrix. Heredity (Edinb) 2005; 95:221–227Crossref, Medline, Google Scholar
31 : Diagnostic accuracy of the Child Behavior Checklist scales for attention-deficit hyperactivity disorder: a receiver-operating characteristic analysis. J Consult Clin Psychol 1994; 62:1017–1025Crossref, Medline, Google Scholar
32 : Automated quality assessment of structural magnetic resonance images in children: comparison with visual inspection and surface-based reconstruction. Hum Brain Mapp 2018; 39:1218–1231Crossref, Medline, Google Scholar
33 : Emotion dysregulation in attention deficit hyperactivity disorder. Am J Psychiatry 2014; 171:276–293Link, Google Scholar
34 : The impact of reinforcement contingencies on AD/HD: a review and theoretical appraisal. Clin Psychol Rev 2005; 25:183–213Crossref, Medline, Google Scholar
35 : Validity of the executive function theory of attention-deficit/hyperactivity disorder: a meta-analytic review. Biol Psychiatry 2005; 57:1336–1346Crossref, Medline, Google Scholar
36 : Anterior cingulate cortex: unique role in cognition and emotion. J Neuropsychiatry Clin Neurosci 2011; 23:121–125Crossref, Medline, Google Scholar
37 : Impulsive aggression and response inhibition in attention-deficit/hyperactivity disorder and disruptive behavioral disorders: findings from a systematic review. Neurosci Biobehav Rev 2018; 90:231–246Crossref, Medline, Google Scholar
38 : Where is the anterior temporal lobe and what does it do? J Neurosci 2013; 33:4213–4215Crossref, Medline, Google Scholar
39 : Altered gray matter organization in children and adolescents with ADHD: a structural covariance connectome study. Transl Psychiatry 2016; 6:e947Crossref, Medline, Google Scholar
40 : Integrity of amygdala subregion-based functional networks and emotional lability in drug-naïve boys with ADHD. J Atten Disord (Epub ahead of print, August 8, 2016)Google Scholar
41 : Attention-deficit/hyperactivity disorder is characterized by a delay in cortical maturation. Proc Natl Acad Sci USA 2007; 104:19649–19654Crossref, Medline, Google Scholar
42 : Developmental comorbidity in attention-deficit/hyperactivity disorder. Atten Defic Hyperact Disord 2010; 2:267–289Crossref, Medline, Google Scholar
43 : Valid conjunction inference with the minimum statistic. Neuroimage 2005; 25:653–660Crossref, Medline, Google Scholar
44 : Genetic markers of ADHD-related variations in intracranial volume. Am J Psychiatry 2019; 176:228–238Link, Google Scholar
45 : Finding community structure in networks using the eigenvectors of matrices. Phys Rev E Stat Nonlin Soft Matter Phys 2006; 74:036104Crossref, Medline, Google Scholar
46 : Genetic influences on schizophrenia and subcortical brain volumes: large-scale proof of concept. Nat Neurosci 2016; 19:420–431Crossref, Medline, Google Scholar
47 : Development of cortical surface area and gyrification in attention-deficit/hyperactivity disorder. Biol Psychiatry 2012; 72:191–197Crossref, Medline, Google Scholar
48 : Multi-site genetic analysis of diffusion images and voxelwise heritability analysis: a pilot project of the ENIGMA-DTI working group. Neuroimage 2013; 81:455–469Crossref, Medline, Google Scholar
49 : A resting state fMRI analysis pipeline for pooling inference across diverse cohorts: an ENIGMA rs-fMRI protocol. Brain Imaging Behav (Epub ahead of print, September 6, 2018)Google Scholar
50 : Brain alterations in adult ADHD: effects of gender, treatment, and comorbid depression. Eur Neuropsychopharmacol 2014; 24:397–409Crossref, Medline, Google Scholar
51 : Characterising resting-state functional connectivity in a large sample of adults with ADHD. Prog Neuropsychopharmacol Biol Psychiatry 2016; 67:82–91Crossref, Medline, Google Scholar
52 : A genome-wide association meta-analysis of attention-deficit/hyperactivity disorder symptoms in population-based pediatric cohorts. J Am Acad Child Adolesc Psychiatry 2016; 55:896–905.e6Crossref, Medline, Google Scholar



