Prediction of Working Memory Performance in Schizophrenia by Plasma Ratio of Clozapine to N-Desmethylclozapine
Abstract
Objective:
Clozapine’s potent antagonism of muscarinic M1 receptors is thought to worsen working memory deficits associated with schizophrenia. In contrast, its major metabolite, N-desmethylclozapine (NDMC), is thought to enhance working memory via its M1 receptor agonist activity. The authors hypothesized that the ratio of serum clozapine and NDMC concentrations would be inversely associated with working memory performance in schizophrenia.
Method:
Thirty patients with schizophrenia or schizoaffective disorder who were receiving clozapine monotherapy at bedtime completed the MATRICS Consensus Cognitive Battery (MCCB) on the day their blood was collected to assess concentrations of clozapine and NDMC as well as serum anticholinergic activity.
Results:
The clozapine/NDMC ratio was significantly and negatively associated with working memory performance after controlling for age, gender, education, and symptom severity. No significant associations were found between individual clozapine and NDMC concentrations and working memory performance. Serum anticholinergic activity was significantly associated with clozapine concentration, but not with working memory performance or NDMC concentration. No significant associations were found between any pharmacological measure and performance on other MCCB cognitive domains.
Conclusions:
This hypothesis-driven study confirms that clozapine/NDMC ratio is a strong predictor of working memory performance in patients with schizophrenia. This finding suggests that manipulating the clozapine/NDMC ratio could enhance cognition in patients with schizophrenia treated with clozapine. It also supports the study of procholinergic agents, such as M1 receptor-positive allosteric modulators, to enhance cognition in schizophrenia.
Deficits in working memory are a core feature in patients with schizophrenia (1). They are a strong predictor of functional outcomes (2), and they are not improved by currently available pharmacologic agents. Abnormal cholinergic neurotransmission has been hypothesized to be a mechanism underlying working memory dysfunction in patients with schizophrenia (3, 4). Notwithstanding the role of the dopaminergic system (especially through D1 receptors) and the nicotinic system, muscarinic neurotransmission supports working memory through several mechanisms: enhanced discrimination of signal from noise, enhancement of dopaminergic neurotransmission, and dilation of cerebral blood vessels (see reference 5 for a recent review). However, to date, studies aimed at improving cholinergic neurotransmission have failed to improve cognition in patients with schizophrenia (6, 7). The large variability in the cholinergic status of participants in these studies may have been problematic. Anticholinergic burden has been shown to be associated with cognitive impairment (8), including working memory deficits (9), in patients with schizophrenia. It has also been shown to prevent these patients from benefiting from procognitive interventions (8).
This variability in cholinergic status could explain the mixed findings in the literature on the effects of clozapine on cognition or working memory performance in patients with schizophrenia. Studies assessing clozapine’s impact on cognition have shown mixed results, ranging from deleterious (10–12) to neutral (13, 14) to beneficial effects (15, 16). The differing actions of clozapine and its major active metabolite, N-desmethylclozapine (NDMC), on the cholinergic system have been proposed as the explanation for these varying effects on cognition. Clozapine is an antagonist at muscarinic M1, M3, and M5 receptors (17, 18); thus, it is expected to have a detrimental effect on cognition, especially frontal working memory. In contrast, NDMC is a potent partial agonist at these sites and is expected to enhance working memory performance (19, 20) (see Table 1 for clozapine and NDMC affinities to various receptors). Notably, plasma concentrations of NDMC can vary from 20% to 150% of clozapine concentrations (21). Thus, the cholinergic tone of individual patients and consequently their cognitive function should be more affected by the ratio of clozapine to NDMC than by either concentration alone.
| Biding Affinity (nM) | ||
|---|---|---|
| Receptor | Clozapine | NDMC |
| 5-HT1A | 123.7 | 13.9 |
| 5-HT1B | 519.0 | 406.8 |
| 5-HT1D | 130.0 | 476.2 |
| 5-HT2A | 8.6 | 10.9 |
| 5-HT2B | 5.9 | 2.8 |
| 5-HT2C | 16.5 | 11.9b |
| 5-HT3 | 241.0 | 272.2 |
| 5-HT transporter | 1,624.0 | 316.6 |
| D1 | 266.3 | 14.3 |
| D2 | 164.0 | 101.4 |
| D3 | 316.2 | 153 |
| D4 | 24.06 | 63.94 |
| D5 | 255.3 | 283.6 |
| Dopamine transporter | >10,000 | >10,000 |
| H1 | 1.43 | 3.4 |
| M1 | 17.8 | 67.6 |
| M2 | 148.2 | 414.5 |
| M3 | 62.1 | 95.7 |
| M4 | 19.6 | 169.9 |
| M5 | 26.1 | 35.4 |
| Norepinephrine transporter | 3,168.0 | 493.9 |
| α1A | 1.6 | 104.8 |
TABLE 1. Receptor Binding Affinities of Clozapine and N-Desmethylclozapine (NDMC)a
Two published retrospective secondary data analyses support this hypothesis. One analysis (22) assessed the relationship between executive function and exposure to clozapine and NDMC in patients with schizophrenia who had participated in two clinical trials. The investigators found that a low clozapine/NDMC ratio was associated with improvement in attention and working memory performance, and to a lesser degree executive function, while each concentration alone was not. In the other analysis (23), the clozapine/NDMC ratio was more strongly associated with cognitive impairment, as assessed by a clinical measure of global cognition, than was either concentration alone. Thus, based on these results and on the role of muscarinic neurotransmission in working memory, we conducted a hypothesis-driven study designed to assess whether clozapine/NDMC ratio is a stronger determinant of cognition than clozapine and NDMC concentrations alone. We hypothesized that clozapine/NDMC ratio would predict working memory performance after controlling for age, gender, education, and symptom severity. We then tested the hypothesis of whether clozapine or NDMC concentration alone predicts working memory performance.
To assess the relationships between clozapine, NDMC, and the cholinergic burden, we explored the relationships between serum anticholinergic activity (SAA), clozapine concentration, NDMC concentration, and cognition.
Method
Participants
This cross-sectional study was conducted between February 2010 and December 2013 at the Centre for Addiction and Mental Health, a large academic psychiatric hospital that provides care to an urban catchment area and serves as a referral center for a large suburban and rural population in the Greater Toronto Area. Participants met DSM-IV-TR criteria for schizophrenia or schizoaffective disorder, were at least 18 years of age, and were clinically stable on clozapine monotherapy, taken once a day at bedtime. Clinical stability was operationalized as both not having been hospitalized within the past 3 months and having had no change in clozapine dosage within the past 4 weeks. The study was approved by the Centre for Addiction and Mental Health Research Ethics Board, and all participants provided written informed consent.
Assessments
On the morning that participants underwent their routine blood draw for white blood cell and absolute neutrophil count, their clinical and cognitive states were assessed and we collected a blood sample to measure clozapine and NDMC concentration and SAA.
Clinical and Cognitive Assessments
The Mini International Neuropsychiatric Interview (24) was used to confirm diagnosis. Clinical symptoms were assessed with the Positive and Negative Syndrome Scale (PANSS) (25). Cognition was assessed on the day the blood was drawn with the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery (MCCB) (26). The MCCB includes 10 tests that assess seven cognitive domains: 1) processing speed (the symbol coding task from the Brief Assessment of Cognition in Schizophrenia, the category fluency test [animal naming], and the Trail Making Test, part A); 2) attention/vigilance (the Continuous Performance Test, Identical Pairs version); 3) working memory (the letter-number span test and the spatial span subtest of the WMS-III); 4) verbal learning (the Hopkins Verbal Learning Test–Revised); 5) visual learning (the Brief Visuospatial Memory Test–Revised); 6) reasoning and problem solving (the mazes test from the Neuropsychological Assessment Battery); and 7) social cognition (the managing emotions test from the Mayer-Salovey-Caruso Emotional Intelligence Test). Each domain T-score was used individually as an outcome measure.
Clozapine and NDMC Concentrations
Concentrations of clozapine and NDMC were determined from heparinized plasma by high-performance liquid chromatography with isocratic elution and ultraviolet detection at 245 nm (Waters instrumentation) using a procedure developed in-house as an adaptation from established methods (27). The limit of quantitation was 100 nmol/L for both clozapine and NDMC, with a coefficient of variation for both of 8%–10%.
Serum Anticholinergic Activity (SAA)
SAA was determined using established procedures (28). The limit of detection for SAA in this study was 0.25 pmol/mL with a standard linear curve (r=0.99) from 0.50 to 25.00 pmol/mL and a coefficient of variation <12%.
Statistical Analysis
Our primary analyses assessed the relationship between working memory performance and demographic, clinical, and pharmacological variables. We first computed a series of Spearman correlations between working memory performance (T-scores) on the one hand, and age, gender, education, PANSS score, clozapine concentration, NDMC concentration, and clozapine/NDMC ratio on the other. We then conducted an initial standard multiple regression analysis in which working memory performance was the dependent variable and age, gender, education, PANSS score, and clozapine/NDMC ratio were entered simultaneously as the independent variables. Finally, we conducted two further multiple regression analyses, replacing clozapine/NDMC ratio with clozapine concentration in one and NDMC concentration in the other.
Our first set of secondary analyses assesses whether the relationship between clozapine/NDMC ratio and working memory performance was driven by only one of the two working memory tests (letter-number span test and spatial span test) that contribute to the working memory domain in MCCB. Thus, we performed a series of analyses similar to those described above, except that working memory was replaced by each of the two tests separately.
Our second set of secondary analyses assessed the relationship between performance on the other six cognitive domains and demographic, clinical, and pharmacological variables. In these analyses, multivariate general linear models were applied. The dependent variables were speed of processing, attention/vigilance, verbal learning, visual learning, reasoning and problem solving, and social cognition. The covariates were age, gender, education, PANSS score, and clozapine concentration, NDMC concentration, or clozapine/NDMC ratio.
Our third set of secondary analyses explored whether the effects of clozapine and NDMC concentrations on cognition were related to cholinergic burden. First, we computed Spearman correlations between SAA on the one hand and clozapine and NDMC concentrations on the other, and between clozapine and NDMC concentrations. Second, we conducted a standard multiple regression analysis in which SAA was the dependent variable and age, gender, clozapine concentration, and NDMC concentration were entered simultaneously as the independent variables. Third, we conducted regression analyses similar to those with working memory performance and clozapine/NDMC ratio but replacing clozapine/NDMC ratio with SAA.
We used SPSS, version 15.0 for Windows (SPSS, Inc., Chicago), for all analyses.
Results
The demographic, clinical, cognitive, and pharmacological characteristics of the 30 participants who completed the study are summarized in Table 2.
| Measure | |||
|---|---|---|---|
| Demographic and clinical characteristics | |||
| N | % | ||
| Male | 18 | 60 | |
| Diagnosis | |||
| Schizophrenia | 25 | 83 | |
| Schizoaffective disorder | 5 | 17 | |
| Mean | SD | Range | |
| Age (years) | 38.6 | 15.4 | 21–70 |
| Education (years) | 12.2 | 2.6 | 6–19 |
| Clozapine dosage (mg/day) | 352.7 | 105.1 | 150–550 |
| Clozapine concentration (nmol/L) | 2,317.8 | 1,432.2 | 498–5,864 |
| NDMC concentration (nmol/L) | 1,608.0 | 1,026.6 | 221–4,765 |
| Clozapine/NDMC concentration ratio | 1.64 | 0.70 | 0.50–3.07 |
| Serum anticholinergic activity | 21.1 | 12.0 | 2.9–47.8 |
| Positive and Negative Syndrome Scale | |||
| Total score | 53.7 | 9.8 | 35–73 |
| Positive score | 13.8 | 4.5 | 7–28 |
| Negative score | 13.8 | 5.0 | 7–28 |
| General score | 26.2 | 5.6 | 17–41 |
| MATRICS Consensus Cognitive Battery (T-scores) | |||
| Processing speed | 36.4 | 13.6 | 12–68 |
| Symbol coding task (Brief Assessment of Cognition in Schizophrenia) | 39.3 | 12.0 | 19–62 |
| Category fluency (animal naming) | 40.6 | 11.9 | 21–71 |
| Trail Making Test, part A | 37.7 | 14.5 | 6–71 |
| Attention/vigilance | 35.2 | 11.9 | 17–60 |
| Continuous Performance Test, Identical Pairs version | 35.2 | 11.9 | 17–60 |
| Working memory | 37.3 | 8.5 | 19–49 |
| Letter-number span test | 37.1 | 9.0 | 18–50 |
| Spatial span test (WMS-III) | 41.4 | 8.2 | 27–54 |
| Verbal learning | 35.4 | 9.3 | 23–63 |
| Hopkins Verbal Learning Test–Revised | 35.4 | 9.3 | 23–63 |
| Visual learning | 33.3 | 12.0 | 16–55 |
| Brief Visuospatial Memory Test–Revised | 33.3 | 12.0 | 16–55 |
| Reasoning and problem solving | 37.5 | 6.9 | 28–55 |
| Mazes test (Neuropsychological Assessment Battery) | 37.5 | 6.9 | 28–55 |
| Social cognition | 38.5 | 12.8 | 19–65 |
| Managing emotions test (Mayer-Salovey-Caruso Emotional Intelligence Test) | 38.5 | 12.8 | 19–65 |
| Overall | 27.9 | 11.8 | 9–57 |
TABLE 2. Demographic, Clinical, and Cognitive Characteristics of Participants in a Study of Clozapine-to-N-Desmethylclozapine (NDMC) Plasma Ratio and Working Memory Performance in Schizophrenia
Working memory performance was negatively correlated with clozapine/NDMC ratio (Figure 1) but was not correlated with age, gender, education, PANSS score, clozapine concentration, or NDMC concentration (Table 3).
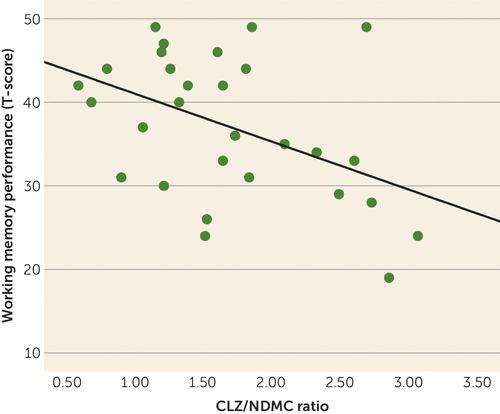
FIGURE 1. Relationship Between Working Memory Performance and Clozapine-to-N-Desmethylclozapine (NDMC) Plasma Ratio in Schizophreniaa
a Spearman’s rho=−0.40, p=0.03.
| Measure | Working Memory | Letter-Number Span Test | Spatial Span Test | |||
|---|---|---|---|---|---|---|
| t | p | t | p | t | p | |
| Gender | 0.26 | 0.80 | 0.11 | 0.91 | 0.32 | 0.75 |
| rs | p | rs | p | rs | p | |
| Age | –0.34 | 0.06 | –0.09 | 0.65 | –0.39 | 0.04 |
| Education | –0.09 | 0.65 | –0.05 | 0.80 | –0.05 | 0.79 |
| Positive and Negative Syndrome Scale score | –0.01 | 0.95 | –0.16 | 0.40 | 0.15 | 0.44 |
| Clozapine concentration | –0.28 | 0.13 | –0.15 | 0.43 | –0.26 | 0.16 |
| NDMC concentration | –0.02 | 0.90 | 0.04 | 0.86 | 0.00 | 1.00 |
| Clozapine/NDMC ratio | –0.40 | 0.03 | –0.26 | 0.17 | –0.41 | 0.02 |
| Serum anticholinergic activity | –0.06 | 0.76 | –0.07 | 0.71 | 0.00 | 0.99 |
TABLE 3. Correlation Matrix With Working Memory and Working Memory Tests in a Study of Clozapine-to-N-Desmethylclozapine (NDMC) Plasma Ratio and Working Memory Performance in Schizophrenia
As shown in Table 4, the results of the first multiple regression showed that working memory performance was associated with clozapine/NDMC ratio but not with age, gender, education, or PANSS score. In contrast, the results of the two multiple regressions with clozapine or NDMC concentration showed that working memory performance was not associated with either alone.
| Model and Variable | B | SE B | β | p |
|---|---|---|---|---|
| Clozapine/NDMC ratio (R2=0.32, p=0.040) | ||||
| Age | –0.16 | 0.10 | –0.30 | 0.11 |
| Gender | 0.52 | 2.97 | 0.03 | 0.86 |
| Education | –0.23 | 0.59 | –0.07 | 0.70 |
| PANSS score | –0.06 | 0.15 | –0.07 | 0.71 |
| Clozapine/NDMC ratio | –5.49 | 2.18 | –0.45 | 0.019 |
| Clozapine concentration (R2=0.27, p=0.16) | ||||
| Age | –0.24 | 0.10 | –0.42 | 0.027 |
| Gender | 1.18 | 3.12 | 0.07 | 0.72 |
| Education | 0.18 | 0.59 | 0.06 | 0.76 |
| PANSS | –0.04 | 0.16 | –0.04 | 0.81 |
| Clozapine concentration | –0.002 | 0.001 | –0.37 | 0.06 |
| NDMC concentration (R2=0.15, p=0.55) | ||||
| Age | –0.21 | 0.11 | –0.38 | 0.07 |
| Gender | –0.83 | 3.32 | –0.05 | 0.80 |
| Education | 0.14 | 0.66 | 0.04 | 0.89 |
| PANSS score | –0.02 | 0.17 | –0.03 | 0.89 |
| NDMC | <0.001 | 0.002 | 0.01 | 0.96 |
TABLE 4. Multiple Linear Regressions With Working Memory in a Study of Clozapine-to-N-Desmethylclozapine (NDMC) Plasma Ratio and Working Memory Performance in Schizophreniaa
The multiple regressions with each of the working memory tests showed that there could be a trend toward significant associations between clozapine/NDMC ratio and verbal working memory performance (as assessed by the letter-number span test) or visual working memory performance (as assessed by the spatial span test), separately (letter-number span test: B=−4.68, SE B=2.49, β=−0.37, p=0.072; spatial span test: B=−4.12, SE B=2.20, β=−0.35, p=0.074). Also, they were not associated with clozapine concentration.
The multivariate general regression models assessing the relationships with the other six cognitive domains revealed no significant effects of clozapine concentration, NDMC concentration, clozapine/NDMC ratio, or SAA on performance in any of these six cognitive domains.
As expected, SAA was highly correlated with clozapine concentration (Spearman’s rho [rs]=0.83, p<0.001) and NDMC concentration (rs=0.66, p<0.001). Clozapine and NDMC concentrations were also highly correlated (rs=0.73, p<0.001). The multiple regression revealed an association of SAA with clozapine concentration (B=0.006, SE B=0.001, β=0.71, p<0.001) but not with NDMC concentration. The results of the multiple regression that included working memory performance as the dependent variable and SAA as independent variable revealed that SAA was not significantly associated with working memory performance.
Discussion
This study confirmed the hypothesis that clozapine/NDMC ratio is strongly and negatively associated with working memory performance in patients with schizophrenia. This association was independent of age, gender, and symptom severity. We also did not find such a relationship with any other cognitive domain. Finally, we found a strong association between clozapine concentration and SAA but not between working memory performance and SAA.
Our findings are consistent with the results of two previous retrospective studies assessing the relationship between clozapine and NDMC concentrations and cognition. Our study confirms these preliminary findings in a well-characterized sample of adult patients with schizophrenia receiving clozapine monotherapy and recruited in a study designed to assess this relationship. Our study also shows that the association between clozapine/NDMC ratio and cognition is specific to working memory, as predicted by the role of cholinergic neurotransmission in cognitive function.
NDMC is a major metabolite of clozapine and is produced via N-desmethylation of clozapine by CYP1A2, CYP2C19, and CYP3A4 (29, 30). It has M1 receptor agonist activity (19) and potentiates hippocampal NMDA receptor currents through M1 receptor activation (20). NDMC could affect cognition through these interactions with NMDA receptor-dependent neuroplasticity. It could also improve cognition by increasing dopaminergic and cholinergic neurotransmission in the medial prefrontal cortex and hippocampus, both of which are blocked by an M1 receptor antagonist (31). Finally, NDMC could have cognitive effects by decreasing inhibitory synaptic transmission mediated by postsynaptic GABAA receptors (32). These effects could be further complicated by baseline dopaminergic tone in the frontal cortex as determined by each person’s unique characteristics, such as being affected by schizophrenia, having been exposed to antipsychotic medication for several years, and genetic makeup, such as having catechol-O-methyltransferase (COMT) Val158Met polymorphism.
The effects of clozapine and NDMC on cognition via mechanisms mediated by the M1 receptor have been demonstrated in animal studies. In one rodent study (33), clozapine was found to impair performance on a working memory task while NDMC enhanced it. In the same study, the procognitive effects of NDMC were found to be reversed by an M1 receptor antagonist. In a study in phospholipase C-β1 knockout mice known to have M1 muscarinic receptor dysfunction (34), behavioral deficits were reversed after administration of NDMC. Other studies have demonstrated positive behavioral effects of NDMC via muscarinic receptor neurotransmission activation (35) or an NMDA receptor-dependent mechanism (36). Still, in another rodent study, NDMC had no effects on the novel object recognition deficits induced by a hypoglutamatergic state, while clozapine caused further impairment (37). Taken together, these studies and the present findings suggest that NDMC could have a procognitive effect, or it could merely offset the deleterious effect of clozapine. Furthermore, the opposing effects of clozapine and NDMC on cognition are probably the reason why clozapine and NDMC, separately, are not associated with cognition in patients with schizophrenia. In contrast, clozapine/NDMC ratio is a stronger predictor of cognition because it accounts for these opposing effects.
We did not find any significant relationship between clozapine/NDMC ratio and any cognitive domain other than working memory. We believe this is because the cholinergic system is robustly and selectively critical in performing tasks that depend heavily on complex attentional processes (38). The MCCB working memory tests are highly dependent on complex attentional processes and therefore on cholinergic neurotransmission. The MCCB tests of other cognitive domains (e.g., episodic memory and reasoning and problem solving) are likely to depend on attentional processes, encoding of information, and short-term retrieval in episodic memory and planning and inhibitory mechanisms in problem solving, which are as dependent on other neurochemical systems as on the cholinergic system, and possibly more so.
As expected, SAA was strongly correlated with clozapine and NDMC concentrations. However, the association with NDMC concentration did not remain significant after we controlled for the association with clozapine concentration, which suggests that SAA is more reflective of clozapine curtailing the binding of NDMC. The absence of association between NDMC concentration and SAA after controlling for clozapine concentration could also be due to collinearity between clozapine and NDMC concentrations. Also as expected, we found that SAA was not associated with working memory performance. This is most likely due to the fact that there is no association between clozapine concentration alone and working memory performance and that SAA does not account for NDMC and its procognitive effects.
These findings seem to contradict those of a recent study in patients with schizophrenia in which SAA was inversely associated with both working memory performance and frontal gray matter density (9). However, in that study, patients were receiving various conventional and atypical antipsychotic medications and some anticholinergic medications; only seven of the 47 patients were being treated with clozapine. Thus, in that study, cognition was most likely affected mainly by the antimuscarinic burden of medications and not likely by NDMC, and consequently SAA, reflecting this antimuscarinic burden, would be expected to be associated with cognition. This is consistent with a study in which SAA was not related to cognition in patients receiving only clozapine (39) and with a follow-up study that included subjects receiving either clozapine or olanzapine (40) in which SAA was negatively associated with frontal executive control. It is also consistent with the fact that the majority of subjects who demonstrated a lack of cognitive improvement associated with high SAA during cognitive training were receiving olanzapine rather than clozapine (8). Taken together, these findings suggest that these other antipsychotics do not produce metabolites that could offset their anticholinergic burden and its negative impact on cognition.
Our study has some limitations. Because our design was cross-sectional, we did not assess the impact of changing clozapine/NDMC ratio on working memory. Such an intervention would be a logical follow-up to our study. While the MCCB was designed to assess the effects of interventions on cognition, it is possible that it is not sensitive enough to detect effects of clozapine and NDMC concentrations on cognitive domains other than working memory. Similarly, given the small size of our sample, we may have missed small effects. This limitation is partly offset by the homogeneity of our sample, which consisted exclusively of patients treated with clozapine monotherapy and a bedtime regimen. Still, replication in a larger sample is warranted.
Notwithstanding these limitations, our results have several potential implications both for future research and clinical practice. First, they highlight the importance of assessing the state of a target neurochemical system when attempting to intervene with agents that alter this system to enhance cognition. Our study specifically illustrates the need to take into account the effects of both a parent compound and its active metabolites when assessing a potential procognitive agent. Second, our study supports recent efforts to target M1 receptor neurotransmission to enhance cognition: such agents could serve as adjunctive cognitive enhancers in the treatment of patients of schizophrenia. Finally, our study demonstrates the cognitive cost of a high clozapine/NDMC ratio. In fact, given the high anticholinergic burden associated with clozapine (18), one would expect patients to be almost delirious. Our results suggest that NDMC offsets such high cognitive costs. This is an important consideration, given the recent interest in increasing the clozapine/NDMC ratio to increase the efficacy and tolerability of clozapine (41). Conversely, it suggests that cognitive benefits could be obtained by reducing a high dosage of clozapine, given that the clozapine concentration is likely to decline before the NDMC concentration if N-desmethylation is saturated (42). Alternatively, an agent that inhibits the metabolism of NDMC but not that clozapine (or does so to a lesser extent) could possibly improve cognition without worsening psychotic symptoms.
Taken together, our findings suggest that the clozapine/NDMC ratio should be monitored routinely. This is particularly true for patients undergoing pharmacological or other clinical changes that affect clozapine metabolism or those in whom cognitive change is noticed. A cognitive decline that is associated with an increase in the clozapine/NDMC ratio should warrant an evaluation of what could have caused the increase and an adjustment of the cause or of the clozapine dosage.
1 : Neurocognitive deficit in schizophrenia: a quantitative review of the evidence. Neuropsychology 1998; 12:426–445Crossref, Medline, Google Scholar
2 : Longitudinal studies of cognition and functional outcome in schizophrenia: implications for MATRICS. Schizophr Res 2004; 72:41–51Crossref, Medline, Google Scholar
3 : Cholinergic contributions to the cognitive symptoms of schizophrenia and the viability of cholinergic treatments. Neuropharmacology 2012; 62:1544–1553Crossref, Medline, Google Scholar
4 : Cholinergic pathways and cognition in patients with schizophrenia: a pilot study. Schizophr Res 2012; 139:46–52Crossref, Medline, Google Scholar
5 : Leveraging the cortical cholinergic system to enhance attention. Neuropharmacology 2013; 64:294–304Crossref, Medline, Google Scholar
6 : Galantamine for the treatment of cognitive impairments in people with schizophrenia. Am J Psychiatry 2008; 165:82–89Link, Google Scholar
7 : Efficacy and safety of donepezil in patients with schizophrenia or schizoaffective disorder: significant placebo/practice effects in a 12-week, randomized, double-blind, placebo-controlled trial. Neuropsychopharmacology 2008; 33:1217–1228Crossref, Medline, Google Scholar
8 : The cognitive cost of anticholinergic burden: decreased response to cognitive training in schizophrenia. Am J Psychiatry 2009; 166:1055–1062Link, Google Scholar
9 : Prefrontal gray matter morphology mediates the association between serum anticholinergicity and cognitive functioning in early course schizophrenia. Psychiatry Res 2012; 204:61–67Crossref, Medline, Google Scholar
10 : Effectiveness of second-generation antipsychotics in patients with treatment-resistant schizophrenia: a review and meta-analysis of randomized trials. Am J Psychiatry 2001; 158:518–526Link, Google Scholar
11 : The effect of clozapine on cognition and psychiatric symptoms in patients with schizophrenia. Br J Psychiatry 1993; 162:43–48Crossref, Medline, Google Scholar
12 : The effects of clozapine on symptom reduction, neurocognitive function, and clinical management in treatment-refractory state hospital schizophrenic inpatients. Neuropsychopharmacology 1996; 15:361–369Crossref, Medline, Google Scholar
13 : Effects of clozapine, fluphenazine, and placebo on reaction time measures of attention and sensory dominance in schizophrenia. Schizophr Res 1994; 13:133–144Crossref, Medline, Google Scholar
14 : Clinical and neurocognitive effects of clozapine and risperidone in treatment-refractory schizophrenic patients: a prospective study. J Clin Psychiatry 1998; 59:521–527Crossref, Medline, Google Scholar
15 : The comparative efficacy and long-term effect of clozapine treatment on neuropsychological test performance. Biol Psychiatry 1994; 36:717–725Crossref, Medline, Google Scholar
16 : Improvement of cognitive function in schizophrenic patients receiving clozapine or zotepine: results from a double-blind study. Pharmacopsychiatry 1997; 30:35–42Crossref, Medline, Google Scholar
17 : Antischizophrenic drugs and brain cholinergic receptors: affinity for muscarinic sites predicts extrapyramidal effects. Arch Gen Psychiatry 1974; 31:58–61Crossref, Medline, Google Scholar
18 : Anticholinergic activity of 107 medications commonly used by older adults. J Am Geriatr Soc 2008; 56:1333–1341Crossref, Medline, Google Scholar
19 : The highly efficacious actions of N-desmethylclozapine at muscarinic receptors are unique and not a common property of either typical or atypical antipsychotic drugs: is M1 agonism a pre-requisite for mimicking clozapine’s actions? Psychopharmacology (Berl) 2005; 178:451–460Crossref, Medline, Google Scholar
20 : N-desmethylclozapine, an allosteric agonist at muscarinic 1 receptor, potentiates N-methyl-D-aspartate receptor activity. Proc Natl Acad Sci USA 2003; 100:13674–13679Crossref, Medline, Google Scholar
21 : Determination of clozapine and its N-demethylated metabolite in plasma by use of gas chromatography-mass spectrometry with single ion detection. Psychopharmacology (Berl) 1988; 95:472–475Crossref, Medline, Google Scholar
22 : The role of M1 muscarinic receptor agonism of N-desmethylclozapine in the unique clinical effects of clozapine. Psychopharmacology (Berl) 2004; 177:207–216Crossref, Medline, Google Scholar
23 : Clozapine and global cognition in schizophrenia. J Clin Psychopharmacol 2010; 30:431–436Crossref, Medline, Google Scholar
24 : The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998; 59:(suppl 20):22–33Medline, Google Scholar
25 : Reliability and validity of the Positive and Negative Syndrome Scale for schizophrenics. Psychiatry Res 1988; 23:99–110Crossref, Medline, Google Scholar
26 : The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry 2008; 165:203–213Link, Google Scholar
27 : Solid-phase extraction and high-performance liquid chromatographic analysis of clozapine and norclozapine in human plasma. Ther Drug Monit 1996; 18:688–692Crossref, Medline, Google Scholar
28 : Serum anticholinergic activity in a community-based sample of older adults: relationship with cognitive performance. Arch Gen Psychiatry 2003; 60:198–203Crossref, Medline, Google Scholar
29 : The involvement of CYP1A2 and CYP3A4 in the metabolism of clozapine. Br J Clin Pharmacol 1997; 44:439–446Crossref, Medline, Google Scholar
30 : Metabolism and bioactivation of clozapine by human liver in vitro. J Pharmacol Exp Ther 1995; 272:984–990Medline, Google Scholar
31 : N-desmethylclozapine, a major metabolite of clozapine, increases cortical acetylcholine and dopamine release in vivo via stimulation of M1 muscarinic receptors. Neuropsychopharmacology 2005; 30:1986–1995Crossref, Medline, Google Scholar
32 : Effects of clozapine and N-desmethylclozapine on synaptic transmission at hippocampal inhibitory and excitatory synapses. Brain Res 2011; 1421:66–77Crossref, Medline, Google Scholar
33 McFarland K, Johnson RW, Son T: N-Desmethlyclozapine demonstrated pro-cognitive actions in experimental models. Paper presented at the 11th International Congress on Schizophrenia Research. Colorado Springs, Colo, 2007Google Scholar
34 : Role of muscarinic receptors in the activity of N-desmethylclozapine: reversal of hyperactivity in the phospholipase C knockout mouse. Behav Pharmacol 2008; 19:543–547Crossref, Medline, Google Scholar
35 : Behavioral effects of N-desmethylclozapine on locomotor activity and sensorimotor gating function in mice: possible involvement of muscarinic receptors. Brain Res 2011; 1418:111–119Crossref, Medline, Google Scholar
36 : Ameliorative effect of N-desmethylclozapine in animal models of social deficits and cognitive functions. Brain Res Bull 2011; 86:146–151Crossref, Medline, Google Scholar
37 : The influence of N-desmethylclozapine and clozapine on recognition memory and BDNF expression in hippocampus. Brain Res Bull 2011; 84:144–150Crossref, Medline, Google Scholar
38 : Modes and models of forebrain cholinergic neuromodulation of cognition. Neuropsychopharmacology 2011; 36:52–73Crossref, Medline, Google Scholar
39 : Relation of serum anticholinergicity to cognitive status in schizophrenia patients taking clozapine or risperidone. J Clin Psychiatry 1998; 59:184–188Crossref, Medline, Google Scholar
40 : Anticholinergicity and cognitive processing in chronic schizophrenia. Biol Psychol 2001; 56:1–22Crossref, Medline, Google Scholar
41 : Increasing the clozapine:norclozapine ratio with co-administration of fluvoxamine to enhance efficacy and minimize side effects of clozapine therapy. Med Hypotheses 2013; 80:689–691Crossref, Medline, Google Scholar
42 : Plasma clozapine, norclozapine, and the clozapine:norclozapine ratio in relation to prescribed dose and other factors: data from a therapeutic drug monitoring service, 1993–2007. Ther Drug Monit 2010; 32:438–447Crossref, Medline, Google Scholar



