Association of CRP Genetic Variation and CRP Level With Elevated PTSD Symptoms and Physiological Responses in a Civilian Population With High Levels of Trauma
Abstract
Objective:
Increased systemic inflammation is associated with stress-related psychopathology. Specifically, levels of the proinflammatory marker C-reactive protein (CRP) are elevated in individuals with posttraumatic stress disorder (PTSD). Furthermore, single-nucleotide polymorphisms (SNPs) in the CRP gene are associated with CRP level, risk for cardiovascular disease, and obesity. The authors examined whether polymorphisms within the CRP gene and increased CRP levels are associated with PTSD symptoms and fear physiology in a civilian population with high levels of trauma.
Method:
Cross-sectional data and DNA samples were collected from 2,698 individuals recruited from an inner-city public hospital that serves a primarily African American, low-socioeconomic-status population. A subgroup of 187 participants participated in further interviews, testing, and physiological measures; of these, 135 were assessed using the fear-potentiated startle paradigm to assess fear-related phenotypes of PTSD.
Results:
One SNP within the CRP gene, rs1130864, was significantly associated with increased PTSD symptoms (N=2,692), including “being overly alert” as the most significant individual symptom (N=2,698). Additionally, CRP genotype was associated with the odds of PTSD diagnosis (N=2,692). This SNP was also associated with increased CRP level (N=137), and high CRP levels (>3 mg/L) were positively associated with PTSD symptoms (N=187) and fear-potentiated startle to a safety signal (N=135).
Conclusions:
Together, these data indicate that genetic variability in the CRP gene is associated with serum CRP level and PTSD symptom severity, including that of hyperarousal symptoms. Elevated CRP levels were also associated with exacerbated fear-related psychophysiology and PTSD symptom ratings and diagnosis. These findings suggest a potential mechanism by which an increased proinflammatory state may lead to heightened PTSD symptoms.
Posttraumatic stress disorder (PTSD) is an anxiety disorder characterized by avoidance, re-experiencing, and hyperarousal symptoms following exposure to a traumatic life event. However, a diagnosis of PTSD has been linked to high rates of illness (1) and inflammatory disease (2), including cardiovascular (3) and metabolic disease (4). Individuals with PTSD show elevated levels of the inflammatory cytokines interleukin-6 (IL-6), IL-1β, and IL-2 (5–7), augmented nuclear factor-κB (NF-κB) gene expression (8) and activity (9), and altered immune cell sensitivity to glucocorticoids (10). Furthermore, peripheral levels of inflammatory molecules correlate with PTSD ratings (11). These data linking PTSD to a proinflammatory state further support the notion that the disorder is associated with chronic inflammation, in a manner similar to depression (12).
More recently, increased levels of the proinflammatory marker C-reactive protein (CRP) have been described in individuals with PTSD (13, 14). The directionality of the change in CRP levels in PTSD is equivocal, as other studies have shown decreased CRP levels in individuals with PTSD (15) or even a lack of association between PTSD and CRP levels (11, 16). However, a prospective study recently showed that increased baseline CRP prior to deployment was a significant predictor for PTSD, suggesting that increased inflammation is a risk factor for the disorder (17). Genetic variations that cause an increase in CRP levels could also serve as a vulnerability factor for PTSD, as genetic traits significantly account for interindividual variability in CRP level (18). Single-nucleotide polymorphisms (SNPs) are present in the CRP gene that influence CRP level (19) and confer increased individual vulnerability to cardiovascular disease (20).
In the present study, we assessed whether particular SNPs that tag the CRP gene at moderate levels of linkage disequilibrium (rs3093068, rs1205, rs3093067, rs1130864, rs3093066, and rs3091244) are associated with higher serum CRP levels, higher PTSD symptom ratings, and a higher odds ratio for likelihood of PTSD diagnosis. Furthermore, we sought to determine whether increased CRP levels are associated with PTSD in an urban population with high levels of trauma and a high lifetime prevalence of PTSD (21). We hypothesized that CRP level in highly traumatized individuals would be associated positively with PTSD symptom ratings. We also assessed whether CRP level would be associated with fear-potentiated startle, a robust psychophysiological phenotype that is increased in PTSD (22). These data will elucidate whether CRP genetic variance is associated with heightened inflammation indicated by CRP level as well as augmented PTSD symptoms in trauma-exposed individuals. Together, these data suggest a potential mechanism by which increased proinflammatory states may modulate neurophysiology and heighten PTSD symptoms.
Method
Participants
Study participants were recruited from primary care clinics at Grady Memorial Hospital in Atlanta, which serves a primarily African American, low-socioeconomic status, inner-city population (21). Participants were English-speaking men and women 18 to 65 years of age who provided written informed consent. All study procedures were reviewed and approved by the Emory Institutional Review Board and the Grady Hospital Research Oversight Committee.
All participants (N=2,698) underwent a screening interview in which DNA was collected into Oragene saliva kits (DNA Genotek, Ottawa). Demographic information was collected using a locally developed form to record age, sex, self-identified race, education, and income (21). Lifetime trauma history was determined by the 14-item Traumatic Events Inventory, which assesses the experiencing and witnessing of traumatic events separately and has been used to describe our sample population previously (21). PTSD symptoms were assessed with the PTSD Symptom Scale (23).
A subgroup of participants (N=187), representing a cross-section of the entire cohort, were available for in-depth interviews and phenotyping and returned to participate in structured clinical interviews, phlebotomy, and physiological measures. In this subgroup, the Clinician-Administered PTSD Scale (CAPS) was used to assess the categorical presence of current PTSD as well as to generate continuous scores for current PTSD symptoms (24). On the morning of the interview, blood samples were collected for assessment of CRP levels. Of these participants, 135 participated in the fear-potentiated startle paradigm (22) to assess neurophysiology, as described below. Because some participants declined to answer some questions, the total number of participants listed for subanalyses may be lower than the total of the subgroup.
CRP Genotyping and Analyses
DNA from all participants was extracted using the Qiagen M48 automated extraction system. All DNA for genotyping was quantified by gel electrophoresis using Quantity One (BioRad, Hercules, Calif.) and then normalized to 400 ng. Using the Illumina Human Omni1-Quad BeadChip (Illumina, San Diego, Calif.), genome-wide SNP genotyping was performed according to the manufacturer’s instructions.
For these analyses, we analyzed six targeted tagging SNPs that were available from Illumina’s Omni1-Quad platform and that lay within the CRP gene, as previously described (25–28): rs3093068, rs1205, rs3093067, rs1130864, rs3093066, and rs3091244. The maximum r2 between each CRP SNP in our sample of traumatized individuals is described in Table S1 in the data supplement that accompanies the online edition of this article. Quality control and association analyses via linear (or logistic) regression were conducted using PLINK, version 1.07 (29). The genotype call rate for all SNPs was >99%, and all of the SNPs in CRP were in Hardy-Weinberg equilibrium (p values, >0.1). To examine main effects in subjects with substantial trauma exposure, we included subjects in the PTSD Symptom Scale analyses if they had experienced one or more lifetime adult or childhood trauma exposures (N=2,692). All subjects, regardless of trauma level, were included in the CRP analyses (N=137), since we were not directly examining PTSD symptoms. Except where noted, we tested for association under a dominant model, where the “risk” allele carriers were coded as 1 and the “resilient” (major allele homozygote) group coded as 0. A Bonferroni threshold of p<0.0083 was used for correction for multiple testing. As a secondary analysis, we also tested for association under an additive model (see the online data supplement). Using PLINK, we verified key results with permutation tests (10,000) to ensure that our results did not depend on distributional assumptions. In each permutation, trait values were randomly shuffled across individuals, the association test was reperformed, and the t statistic was recorded. Permutation p values were then estimated as the proportion of permutations for which the t statistic exceeded the original in magnitude. To verify our findings from the continuous outcome measure of PTSD symptoms, we also conducted a logistic regression analysis using categorical PTSD diagnosis as an outcome measure using a dominant model.
We had genome-wide data on a subset of our subjects and thus had available principal components to infer axes of ancestry. Prior to principal components analysis, we used PLINK to prune the data in windows of 50 base pairs, removing one SNP from each pair of SNPs with an r2 value >0.05 to obtain a set of roughly independent SNPs. We then used the top 10 principal components as covariates in secondary analyses for PTSD outcomes (30, 31). Given the smaller sample size for the analyses of CRP level, we used only the top two principal components as covariates to prevent model overfitting.
As many more biological and genetic factors are thought to be associated with intermediate phenotypes than with the full syndrome, we separately assessed 17 specific PTSD symptoms examined in the DSM-IV criteria for PTSD via the PTSD Symptom Scale (N=2,692). Specifically, we examined the effect of the dominant model of SNP rs1130864 controlling for the principal components as above, for each symptom in question, with a Bonferroni-corrected threshold of p<0.0029.
CRP Levels
Serum samples were stored at −80°C until the time of CRP assay. Serum CRP level was measured using an immunoturbidometric assay from Sekisui Diagnostics (Lexington, Mass.) on the Beckman AU480 chemistry analyzer, with an interassay coefficient of variation (CV) of 5.2% and an intra-assay CV of 3.1%. Individuals with CRP levels >20 mg/L were excluded from the analysis. CRP levels averaged 5.14 mg/L (SD=4.77, range=0.03–18.84). As previously described (13), a cutpoint of >3 mg/L was used to distinguish individuals with low and high CRP levels.
Fear-Potentiated Startle Paradigm
Based on our previous work (22), the fear-potentiated startle protocol consisted of an initial habituation phase in which conditioned stimuli (CSs) were presented without any reinforcement. The conditioning phase of acquisition consisted of three blocks with four trials of each type of CS (reinforced conditioned stimulus, CS+; nonreinforced conditioned stimulus, CS–; noise probe alone, NA) for 12 trials per block and a total of 36 trials. CSs were shapes of different colors presented on a computer monitor for 6 seconds each. The unconditioned stimulus (aversive stimulus) was a 250-ms air blast of 140-psi intensity to the larynx that, as we have shown previously, produces a robust fear-potentiated startle response (22). The air blast was delivered from a compressed air tank via a polyethylene tube and controlled by a switch. The intertrial intervals throughout the acquisition phase were randomized to be between 9 and 22 seconds.
Startle response data were acquired using the electromyography (EMG) module of the BIOPAC MP150 for Windows (Biopac Systems, Goleta, Calif.). The acquired data were filtered, rectified, and smoothed using the MindWare software suite (MindWare Technologies, Gahanna, Ohio) and exported for statistical analysis. The EMG signal was sampled at a frequency of 1 kHz and filtered with low- and high-frequency cutoffs at 28 Hz and 500 Hz, respectively. The maximum amplitude of the eye-blink muscle contraction 20–200 ms after presentation of the startle probe was used as a measure of the acoustic startle response. The eye-blink component of the acoustic startle response was measured by EMG recordings of the right orbicularis oculi muscle with two 5-mm Ag/AgCl electrodes filled with electrolyte gel, as previously described (22). One electrode was positioned 1 cm below the pupil of the right eye, and the other was placed 1 cm below the lateral canthus. Impedance levels were less than 6 kOhms for each participant. The startle probe was a 40-ms burst of 108-dB(A) SPL broadband noise with near instantaneous rise time, delivered binaurally through headphones.
Fear-potentiated startle was assessed by comparing the average startle magnitude on the CS trials to the average startle magnitude on the NA trials using a mixed-model analysis of variance (ANOVA) with trial type and block as within-subject factors. Fear acquisition was measured using a difference score by subtracting startle magnitude in the NA trials from startle magnitude in the presence of a CS in each conditioning block. Analyses assessing the effects of CRP levels on fear-potentiated startle were conducted in 135 individuals from the larger sample for whom both CRP and fear-potentiated startle data were available. Analyses assessing the effects of rs1130864 on fear-potentiated startle were conducted in 188 individuals for whom genetic data and data on fear-potentiated startle were available.
Statistical Analysis
Bivariate correlation and linear regression were used to assess the association between CRP levels and total current PTSD symptoms and fear-potentiated startle. Chi-square analysis and the calculation of odds ratios and 95% confidence intervals were used to assess differences between low and high CRP levels on categorical variables (PTSD diagnosis), and ANOVAs were used for continuous variables (PTSD symptoms, fear-potentiated startle). Analyses controlled for potential confounders that have previously been associated with CRP levels, such as age and sex (32). The data were analyzed using SPSS, version 20 (IBM, Armonk, N.Y.), and summarized as means and standard deviations. The significance threshold was set at 0.05, and the Bonferroni method was used to correct for multiple comparisons when necessary.
Results
Demographic Characteristics
The participants' demographic characteristics, for the CRP genotype, CRP level, and fear-potentiated startle subgroups, are summarized in Table 1. Women (70.3%) outnumbered men (29.7%), and self-identified race in our sample was predominantly African American (92.9%) in the larger CRP genotype cohort. The majority of the participants (69.1%) were unemployed and had a high school education or less (58.2%). The socioeconomic status of the majority of participants was very low, with 86.5% of the sample having a mean monthly household income below $2,000 and 24.8% having a mean monthly household income below $250. The mean total number of traumas experienced and witnessed in the overall sample was 4.72.
| Characteristic | Genetic Sample (N=2,692) | CRP Sample (N=187) | Startle Sample (N=135) | |||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Female | 1,892 | 70.3 | 107 | 57.4 | 63 | 47.0 |
| Race | ||||||
| African American/black | 2,500 | 92.9 | 168 | 89.9 | 124 | 91.8 |
| Hispanic or Latino | 21 | 0.80 | 1 | 0.80 | 0 | 0 |
| Asian | 5 | 0.20 | 1 | 0.80 | 0 | 0 |
| Caucasian or white | 99 | 3.70 | 11 | 5.40 | 7 | 5.2 |
| Mixed | 35 | 1.30 | 3 | 1.60 | 3 | 2.2 |
| Other | 32 | 1.20 | 3 | 1.60 | 1 | 0.7 |
| Unemployed | 1,860 | 69.1 | 149 | 79.8 | 106 | 79.1 |
| Education | ||||||
| <12th grade | 608 | 22.6 | 52 | 27.9 | 29 | 21.6 |
| High school graduate | 958 | 35.6 | 64 | 34.1 | 51 | 37.3 |
| General Equivalency Diploma | 129 | 4.80 | 18 | 9.30 | 11 | 8.2 |
| Some college or technical school | 641 | 23.8 | 36 | 19.4 | 32 | 23.9 |
| Technical school graduate | 113 | 4.20 | 10 | 5.40 | 4 | 3.0 |
| College graduate | 205 | 7.60 | 6 | 3.10 | 7 | 5.2 |
| Graduate school | 38 | 1.40 | 2 | 0.80 | 1 | 0.7 |
| Monthly household income ($) | ||||||
| 0–249 | 669 | 24.8 | 54 | 28.5 | 43 | 32.1 |
| 250–499 | 262 | 9.70 | 25 | 13.8 | 21 | 15.3 |
| 500–999 | 719 | 26.7 | 53 | 28.5 | 36 | 26.7 |
| 1000–1999 | 678 | 25.2 | 44 | 23.6 | 25 | 18.3 |
| 2000 or more | 364 | 13.5 | 11 | 5.70 | 10 | 7.6 |
| Mean | SD | Mean | SD | Mean | SD | |
| Age (years) | 39.3 | 12.5 | 39.4 | 14.5 | 41.7 | 11.0 |
| PTSD Symptom Scale score | 13.0 | 12.4 | 15.3 | 11.9 | 16.1 | 12.7 |
| Total traumas experienced or witnessed | 4.72 | 3.11 | 5.22 | 4.10 | 5.66 | 3.75 |
TABLE 1. Demographic Characteristics of the Overall Sample and the Subsamples Used for Analyses of CRP Level and Fear-Potentiated Startle Results
CRP Genotype, CRP Level, and PTSD Symptoms
Examination of the six SNPs spanning the CRP gene yielded a single SNP that survived multiple testing when we assessed SNP effects on the number of PTSD symptoms and CRP level. Using a dominant model, rs1130864 was significantly associated with overall PTSD symptoms (N=2,692, t=2.86, p=0.004; permutation p=0.004) and CRP level (N=137, t=2.77, p=0.007; permutation p=0.0006) (Table 2). The main effects of genotype on PTSD symptoms (N=2,692, t=2.65, p=0.008) and CRP level (N=129, t=2.77, p=0.007) were maintained when the analyses controlled for population stratification (using principal components) in the dominant model. Traumatized participants with the CC genotype had an average PTSD Symptom Scale score of 14.1, whereas those with one or two T alleles had an average score of 15.7. Additionally, individuals with the CC genotype had lower serum CRP levels compared with those carrying the T allele (p<0.01) (Figure 1A).
| PTSD Symptoms | CRP Level | |||||
|---|---|---|---|---|---|---|
| SNP | N | t | p | N | t | p |
| rs1205 | 2,692 | –0.11 | 0.91 | 137 | –0.68 | 0.50 |
| rs1130864 | 2,692 | –0.11 | 0.004 | 137 | 2.73 | 0.007 |
| rs3091244 | 2,686 | 0.39 | 0.70 | 137 | –2.76 | 0.007 |
| rs3093066 | 2,691 | 0.66 | 0.51 | 137 | –2.05 | 0.04 |
| rs3093067 | 2,692 | 1.31 | 0.19 | 137 | 0.55 | 0.59 |
| rs3093068 | 2,686 | –2.31 | 0.02 | 137 | 0.55 | 0.58 |
TABLE 2. Main Effects (Dominant Models) of CRP Single-Nucleotide Polymorphisms (SNPs) on Symptoms of Posttraumatic Stress Disorder (PTSD) (N>2,600) and on CRP Levels (N=137)
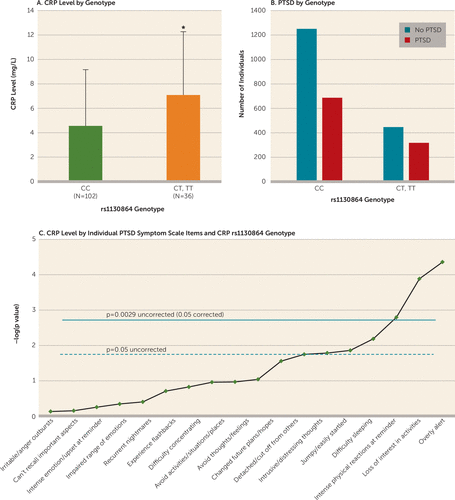
FIGURE 1. Association of CRP Genotype (rs1130864) With CRP Level, Diagnosis of Posttraumatic Stress Disorder (PTSD), and Hypervigilance Symptomsa
a In panel A, which shows the mean serum CRP levels in traumatized individuals of each rs1130864 genotype, individuals with the CC genotype have lower serum CRP levels compared with carriers of the T allele (N=137, t=2.73, p=0.007). Error bars indicate standard deviation. Panel B shows the number of individuals with and without a PTSD diagnosis, among traumatized subjects, of each rs1130864 genotype (dominant model, N=2,547, t=2.89, p=0.004). Panel C presents the Bonferroni-corrected results showing the association between rs1130864 and the 17 different symptom questions on the PTSD Symptom Scale, covarying for the top 10 principal components. The most robust effect was with the hypervigilance symptom of being overly alert (N=2,698, t=4.33, p=1.52×10−5).
* p<0.01.
Using a dominant model, rs1130864 was also associated with a categorical diagnosis of PTSD (N=2,692, t=2.89, p=0.0040) (Figure 1B), and the association remained after covarying for principal components (t=2.80, p=0.005). Thus, individuals carrying the T allele had higher odds for PTSD (odds ratio=1.29, 95% CI=1.09, 1.53) compared with those with the CC genotype, with 41% of traumatized T carriers having a PTSD diagnosis, compared with 35% of those with the CC genotype. Note that, using a dominant model, these effects of rs1130864 genotype on PTSD symptoms (N=2,571, t=2.70, p=0.007), CRP level (N=117, t=2.26, p=0.026), and PTSD diagnosis (N=2,571, t=2.81, p=0.005) survived after covarying for principal components, sex, age, education, current employment, income, and total trauma experienced/witnessed.
After examining the effects of rs1130864 on 17 symptoms measured on the PTSD Symptom Scale and covarying for principal components, three symptoms survived Bonferroni correction (p<0.0029): the hypervigilance symptom of being overly alert, loss of interest in activities, and intense physical reactions at reminders (Figure 1C). The most robust effect was with the hypervigilance symptom of being overly alert (N=2,698, t=4.33, p=1.5×10−5), with a 19% higher score for the hypervigilance symptom in carriers of the T allele compared with individuals with the CC genotype.
CRP Level, CRP Genotype, and Fear Physiology
Figure 2 presents a diagram of the fear conditioning experiment; while the CS+ was reinforced with the air blast (danger signal), the CS– was never paired with the aversive air blast, thus providing a learned safety signal. Results showed that CRP level was positively correlated with fear-potentiated startle to a safety signal (r=0.186, p<0.05). A linear regression analysis controlling for age, race, and PTSD symptoms showed that CRP level still independently predicted fear-potentiated startle (β=0.20, t=2.24, p<0.05). However, adding sex to the model reduced the effect of CRP to a nonsignificant level (p=0.075), with sex also sharing some of the variance in startle response (p=0.068). When sex and CRP level were included in the regression as an interaction term, sex was significantly predictive of increased startle (β=0.24, t=2.74, p=0.007). Follow-up analyses separately within each sex indicated that CRP was associated with startle response in women more than men, although neither was statistically significant (β=0.22, p=0.10, and β=0.02, p=0.88, respectively). Furthermore, individuals with high CRP levels (>3 mg/L) had higher fear-potentiated startle to a safety signal (F=8.60, df=1, 134, p<0.005) (Figure 3), suggesting an impaired ability to control fearful responses, compared with those with lower CRP levels. This main effect of high CRP levels on fear-potentiated startle to a safety signal was still significant after covarying for age, sex, race, and PTSD symptoms (F=8.60, df=1, 124, p=0.006). Given the above interaction with sex, we repeated this analysis separately for males and females and found that high CRP level was associated with higher fear-potentiated startle to the safety signal in women (F=5.86, df=1, 58, p=0.019) but not in men. Lastly, rs1130864 was not associated with fear-potentiated startle to a danger or safety signal in a subsample of 188 individuals (data not shown).

FIGURE 2. The Fear-Potentiated Startle Experimenta
a CS+=reinforced conditioned stimulus (danger signal); CS–=nonreinforced conditioned stimulus (safety signal).
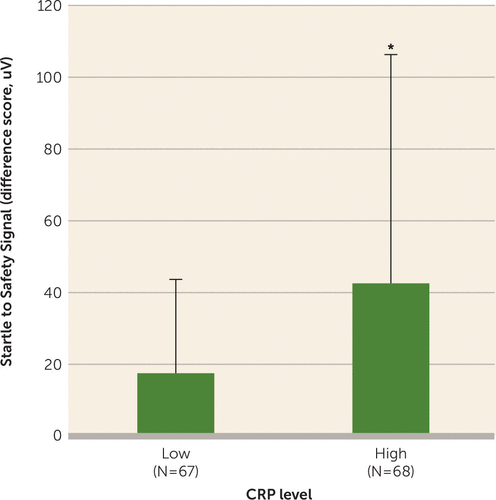
FIGURE 3. Association of CRP Level With Fear-Potentiated Startlea
a The chart shows mean fear-potentiated startle to the safety signal (CS–) in traumatized individuals with low and high CRP levels (N=135). Error bars indicate standard deviation.
*p<0.005.
CRP Level and PTSD
CRP level was positively associated with total frequency of current PTSD symptoms as determined by the CAPS (r=0.179, p=0.018); it was not significantly associated with age or body mass index. High CRP levels were associated with greater PTSD symptoms (F=3.71, df=1, 173, p=0.05) and a diagnosis of PTSD as determined by the CAPS in traumatized participants (χ2=4.58, p=0.032, N=187) (Figure 4). In addition, individuals with high CRP levels had higher odds for PTSD compared with individuals with low CRP levels (odds ratio=2.24, 95% CI=1.06, 4.74; p=0.035).
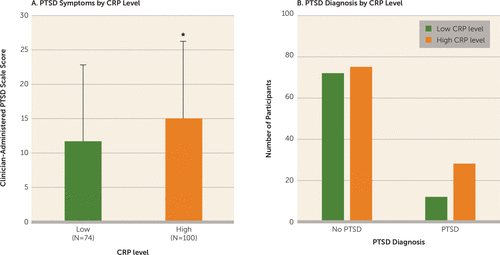
FIGURE 4. Association of CRP Level With Symptoms of Posttraumatic Stress Disorder (PTSD) and Proportion of Participants With a PTSD Diagnosisa
a In panel A, which shows mean PTSD symptom score in traumatized individuals with low (<3 mg/L) and high (>3 mg/L) CRP levels, individuals with high CRP levels had greater PTSD symptom severity (p=0.05; N=174). Error bars indicate standard deviation. Panel B shows the number of individuals with and without a PTSD diagnosis among traumatized subjects with low and high CRP levels (χ2=4.578, p=0.032; N=187).
* p=0.05.
Discussion
Our results indicate that genetic variation in the CRP gene in traumatized individuals augments CRP levels and confers vulnerability to increased hypervigilance. While CRP SNPs are linked to increased susceptibility to cardiovascular and metabolic disease and to increased CRP levels, this is the first demonstration that a SNP within the CRP gene (rs1130864) is associated with PTSD symptoms and CRP levels in traumatized individuals. Furthermore, we demonstrated that elevated CRP levels are associated with increased psychophysiological hyperarousal as determined by increased fear-potentiated startle, exacerbated PTSD symptoms, and greater odds of a PTSD diagnosis. These observations are consistent with previous reports linking a proinflammatory state with psychopathology (12), including PTSD (5, 6, 9).
To our knowledge, this study is the first to describe a genetic locus within the CRP gene—rs1130864—that is associated with increased susceptibility to a PTSD diagnosis and greater PTSD symptom levels. The rs1130864 SNP has previously been associated with increased CRP levels (26), a result replicated here. While both rs1130864 and rs3091244 were the only two loci that were associated with CRP level, only rs1130864 survived multiple-test correction for effects on CRP level and PTSD symptoms. The data indicate that while the rates of PTSD were high in both rs1130864 allelic groups (CC and T) in our sample, paralleling the high overall rates of trauma in our inner-city population (21), the differences between groups were largely based on three symptoms, highlighting the significant effects of the rs1130864 genotype on PTSD symptoms. Specifically, an analysis of the effects of rs1130864 on individual PTSD symptoms in trauma-exposed individuals yielded a strong association with being “overly alert,” suggesting that this genetic locus confers individual vulnerability to hypervigilance by influencing CRP level. Furthermore, a recent study (33) suggested that PTSD in women and increased inflammation are correlated with intrusive re-experiencing, a hallmark symptom of PTSD. Increased alertness and re-experiencing in PTSD may in fact continually activate the stress axis and the immune system and thus dysregulate cytokine-glucocorticoid negative feedback (34) and alter fear physiology (35) in PTSD.
In the present study, higher CRP levels were associated with impaired inhibition of fear-potentiated startle to a safety signal, which is thought to be a biomarker for PTSD (35) and is related to the severity of current PTSD symptoms (36), and more specifically, hyperarousal symptoms (22). This effect of CRP on fear physiology was independent of PTSD symptoms, indicating that inflammation may increase vulnerability to a heightened fear response. The association between CRP level and fear-potentiated startle was stronger in women than in men, supporting recent evidence for heightened fear-potentiated startle after puberty in females compared with males (37) and the role of menstrual cycle phase in modulating fear-potentiated startle in females (38). This sex difference in the CRP/fear-potentiated startle relationship also corroborates previous findings of higher vulnerability to PTSD in females (39, 40) and data indicating sex differences in immune function that leave women more susceptible to increased inflammation (41, 42). Further studies are necessary to elucidate the effects of increased inflammation and female gonadal hormones on fear psychophysiology.
The rs1130864 SNP that was associated with both CRP level and PTSD symptoms was not associated with fear-potentiated startle. The lack of a genotype effect on fear-potentiated startle should be considered preliminary, as it could be due to our small sample of participants who underwent fear-potentiated startle. While self-reported fear has been linked to increased CRP level in healthy individuals (43), there have been no reports in humans or in rodents that describe the effects of inflammation on the psychophysiology of fear responses. It is important to note that the exaggerated neurobiological sensitivity to threat that is characteristic of anxiety disorders, including PTSD, can lead to increased activity of the stress and immune axes, and thus promotes a state of chronic inflammation (44). These data, taken together with our finding that increased fear-potentiated startle is associated with greater serum CRP levels, provide a rationale for assessing the role of inflammation in fear responses and hypervigilance in future research.
Our results corroborate previous reports of heightened CRP levels in individuals with PTSD (13, 14), while other studies have shown decreased CRP levels in individuals with PTSD (15) or even a lack of association between PTSD and CRP level (11, 16). The inconsistencies between these reports may be related to small sample sizes (16), distinct study populations (45), the presence of uncontrolled confounders (13), and the use of control groups with high rates of infection (15). The positive association between CRP level and PTSD symptoms in the present study was described in a highly traumatized inner-city sample consisting primarily of African American participants (21). This population has a higher prevalence of PTSD (21) as well as an elevated risk for cardiovascular and metabolic disease (4, 46), suggesting that increased inflammation in this population may serve as an underlying mechanism by which psychopathology and illness manifest together. Indeed, even though our study could not account for comorbid physical illness in the sample, CRP levels were high (mean=5.14 mg/L, SD=4.77), which suggests that some traumatized individuals recruited to our study from primary care settings also suffered from physical illness. Future studies from clinical populations should analyze both illness comorbidity and the potential confounder of obesity through measures such as body mass index. Additionally, although our study is the largest to date examining CRP genotype in PTSD-related phenotypes, the number of subjects for whom we had data on CRP level was limited, so we lacked adequate statistical power to examine mediation between CRP genotype, CRP level, fear physiology, and PTSD symptoms.
Our findings describe a SNP within the CRP gene that is associated with increased susceptibility to a PTSD diagnosis and greater PTSD symptoms in our inner-city, primarily African American population. This finding joins those of a group of previous reports based on this growing cohort linking distinct candidate genes to increased risk for psychopathology and altered psychophysiology. Genetic loci within genes critical for the neuroendocrine regulation of the stress axis have been associated with increased risk for psychopathology. A SNP within the corticotropin-releasing hormone (CRH) receptor 1 (CRHR1) gene is associated with increased depressive symptoms in individuals who have experienced childhood trauma (47). Polymorphisms within the FKBP5 gene, an important regulator of glucocorticoid negative feedback, increase risk for PTSD symptoms in individuals exposed to childhood trauma (48). Additionally, a polymorphism in the receptor gene (ADCYAP1R1) for pituitary adenylate cyclase-activating polypeptide (PACAP), a peptide implicated in stress-related behavior and physiology (49–51), is associated with PTSD in a sex-dependent manner (52). The ADCYAP1R1 SNP is predictive of PTSD diagnosis and symptoms as well as dysregulated fear discrimination in females (52). In complement to this finding, we also found that a polymorphism within the testosterone metabolic pathway, SRD5A2, was associated with PTSD only in males (53). Results from these retrospective candidate gene studies, together with data from the present study, suggest that SNPs within genes that regulate neuroendoimmunological function confer increased risk for psychopathology, and PTSD in particular, in individuals exposed to psychosocial stressors. In the future, we aim to assess in a prospective manner whether these genetic loci confer increased risk for PTSD and to utilize genome-wide association study approaches.
This study has some limitations that should kept in mind when interpreting the results. Because the study was cross-sectional in nature, it does not allow for the determination of causality. Additionally, the study was not able to account for potential confounders of the association between CRP level and PTSD, including smoking status (54), cardiovascular disease (20), and obesity (55), as these measures were not obtained in the large cohort. However, our genetic data linking the rs1130864 SNP in the CRP gene to increased CRP levels and increased PTSD symptoms suggest that heightened CRP levels due to a genetic trait increase PTSD symptoms in trauma-exposed individuals in a manner independent of other experiential factors. The notion that baseline CRP levels influence PTSD symptoms is corroborated by recent prospective data indicating that baseline CRP level is predictive of PTSD following deployment in a military cohort (17).
While the homogeneity of the study population is a strength of this study, it could also serve as a limitation, as the results may not be generalizable to other samples. Furthermore, our inner-city hospital sample is not a population-based sample, and thus selection biases could be present in the small subset of individuals who participated in CRP level and fear-potentiated startle analyses, which limits the generalizability of our study, as these individuals self-selected to participate further. However, genetic associations with PTSD and depression that were demonstrated in previous initial studies from this cohort (48, 52) have been replicated in a variety of other populations of varied trauma status, race, and socioeconomic status (56, 57). Additionally, high rates of trauma exposure (21) and inflammation (58) are prevalent in urban, low-socioeconomic-status, African American communities and may lead to adverse health outcomes via dysregulation of the stress-immune system, as evidenced by findings also indicating that African Americans (58) and individuals of low socioeconomic status (59) have elevated CRP levels. Our data suggest a possible therapeutic target for alleviating the symptoms associated with PTSD in this population, similar to what has been described in some types of depression (60).
1 : Diseases among men 20 years after exposure to severe stress: implications for clinical research and medical care. Psychosom Med 1997; 59:605–614Crossref, Medline, Google Scholar
2 : Posttraumatic stress disorder and physical illness: results from clinical and epidemiologic studies. Ann N Y Acad Sci 2004; 1032:141–153Crossref, Medline, Google Scholar
3 : Electrocardiogram abnormalities among men with stress-related psychiatric disorders: implications for coronary heart disease and clinical research. Ann Behav Med 1999; 21:227–234Crossref, Medline, Google Scholar
4 : Posttraumatic stress disorder is a risk factor for metabolic syndrome in an impoverished urban population. Gen Hosp Psychiatry 2011; 33:135–142Crossref, Medline, Google Scholar
5 : Elevated serum interleukin-6 (IL-6) and IL-6 receptor concentrations in posttraumatic stress disorder following accidental man-made traumatic events. Biol Psychiatry 1999; 45:833–839Crossref, Medline, Google Scholar
6 : Elevated levels of serum interleukin-1 beta in combat-related posttraumatic stress disorder. Biol Psychiatry 1997; 42:345–348Crossref, Medline, Google Scholar
7 : Differential immune system DNA methylation and cytokine regulation in post-traumatic stress disorder. Am J Med Genet B Neuropsychiatr Genet 2011; 156B:700–708Crossref, Medline, Google Scholar
8 : Transcriptional control of monocyte gene expression in post-traumatic stress disorder. Dis Markers 2011; 30:123–132Crossref, Medline, Google Scholar
9 : Increased peripheral NF-κB pathway activity in women with childhood abuse-related posttraumatic stress disorder. Brain Behav Immun 2012; 26:13–17Crossref, Medline, Google Scholar
10 : Enhanced sensitivity to glucocorticoids in peripheral mononuclear leukocytes in posttraumatic stress disorder. Biol Psychiatry 2004; 55:1110–1116Crossref, Medline, Google Scholar
11 : Evidence for low-grade systemic proinflammatory activity in patients with posttraumatic stress disorder. J Psychiatr Res 2007; 41:744–752Crossref, Medline, Google Scholar
12 : Cytokines sing the blues: inflammation and the pathogenesis of depression. Trends Immunol 2006; 27:24–31Crossref, Medline, Google Scholar
13 : Association of posttraumatic stress disorder with low-grade elevation of C-reactive protein: evidence from the general population. J Psychiatr Res 2010; 44:15–21Crossref, Medline, Google Scholar
14 : C-reactive protein and interleukin 6 receptor in post-traumatic stress disorder: a pilot study. Cytokine 2001; 13:253–255Crossref, Medline, Google Scholar
15 : The inflammatory markers C-reactive protein and serum amyloid A in refugees with and without posttraumatic stress disorder. Clinica Chim Acta 2004; 342:93–98Crossref, Medline, Google Scholar
16 : C-reactive protein, interleukin-6, and posttraumatic stress disorder symptomology in urban police officers. Cytokine 2011; 55:74–78Crossref, Medline, Google Scholar
17 : Assessment of plasma C-reactive protein as a biomarker of posttraumatic stress disorder risk. JAMA Psychiatry 2014; 71:423–431Crossref, Medline, Google Scholar
18 : Familial and genetic determinants of systemic markers of inflammation: the NHLBI family heart study. Atherosclerosis 2001; 154:681–689Crossref, Medline, Google Scholar
19 : Contribution of clinical correlates and 13 C-reactive protein gene polymorphisms to interindividual variability in serum C-reactive protein level. Circulation 2006; 113:1415–1423Crossref, Medline, Google Scholar
20 : Association of polymorphisms in the CRP gene with circulating C-reactive protein levels and cardiovascular events. JAMA 2006; 296:2703–2711Crossref, Medline, Google Scholar
21 : Trauma exposure and stress-related disorders in inner city primary care patients. Gen Hosp Psychiatry 2009; 31:505–514Crossref, Medline, Google Scholar
22 : Impaired fear inhibition is a biomarker of PTSD but not depression. Depress Anxiety 2010; 27:244–251Crossref, Medline, Google Scholar
23 : Reliability and validity of a brief instrument for assessing post-traumatic stress disorder. J Trauma Stress 1993; 6:459–473Crossref, Google Scholar
24 : The development of a Clinician-Administered PTSD Scale. J Trauma Stress 1995; 8:75–90Crossref, Medline, Google Scholar
25 : Polymorphisms of the CRP gene inhibit inflammatory response and increase susceptibility to depression: the Health in Men Study. Int J Epidemiol 2009; 38:1049–1059Crossref, Medline, Google Scholar
26 : C-reactive protein (CRP) gene polymorphisms, CRP levels, and risk of incident essential hypertension: findings from an observational cohort of Han Chinese. Hypertens Res 2012; 35:1019–1023Crossref, Medline, Google Scholar
27 : CRP polymorphisms and progression of chronic kidney disease in African Americans. Clin J Am Soc Nephrol 2010; 5:24–33Crossref, Medline, Google Scholar
28 : Polymorphism in the C-reactive protein (CRP) gene affects CRP levels in plasma and one early marker of atherosclerosis in men: the Health 2000 Survey. Scand J Clin Lab Invest 2011; 71:353–361Crossref, Medline, Google Scholar
29 : PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81:559–575Crossref, Medline, Google Scholar
30 : Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 2006; 38:904–909Crossref, Medline, Google Scholar
31 : Handbook on Analyzing Human Genetic Data: Computational Approaches and Software. New York, Springer, 2009Crossref, Google Scholar
32 : Serum levels of high sensitive C reactive protein in healthy adults from southern Brazil. J Clin Lab Anal 2013; 27:207–210Crossref, Medline, Google Scholar
33 : Interpersonal violence, PTSD, and inflammation: potential psychogenic pathways to higher C-reactive protein levels. Cytokine 2013; 63:172–178Crossref, Medline, Google Scholar
34 : Cytokine-effects on glucocorticoid receptor function: relevance to glucocorticoid resistance and the pathophysiology and treatment of major depression. Brain Behav Immun 2007; 21:9–19Crossref, Medline, Google Scholar
35 : Impaired safety signal learning may be a biomarker of PTSD. Neuropharmacology 2012; 62:695–704Crossref, Medline, Google Scholar
36 : Posttraumatic stress disorder may be associated with impaired fear inhibition: relation to symptom severity. Psychiatry Res 2009; 167:151–160Crossref, Medline, Google Scholar
37 : Developmental investigation of fear-potentiated startle across puberty. Biol Psychol 2014; 97:15–21Crossref, Medline, Google Scholar
38 : The impact of sex and menstrual cycle on the acoustic startle response. Behav Brain Res 2014; 274:326–333Crossref, Medline, Google Scholar
39 : Gender differences in trauma and posttraumatic stress disorder. J Gend Specif Med 2002; 5:34–40Medline, Google Scholar
40 : National estimates of exposure to traumatic events and PTSD prevalence using DSM-IV and DSM-5 criteria. J Trauma Stress 2013; 26:537–547Crossref, Medline, Google Scholar
41 : Systems analysis of sex differences reveals an immunosuppressive role for testosterone in the response to influenza vaccination. Proc Natl Acad Sci USA 2014; 111:869–874Crossref, Medline, Google Scholar
42 : The effects of hormones on sex differences in infection: from genes to behavior. Neurosci Biobehav Rev 2000; 24:627–638Crossref, Medline, Google Scholar
43 : Association of fear of terror with low-grade inflammation among apparently healthy employed adults. Psychosom Med 2004; 66:484–491Crossref, Medline, Google Scholar
44 : Exaggerated neurobiological sensitivity to threat as a mechanism linking anxiety with increased risk for diseases of aging. Neurosci Biobehav Rev 2013; 37:96–108Crossref, Medline, Google Scholar
45 : Relation of genetic variation in the gene coding for C-reactive protein with its plasma protein concentrations: findings from the Women’s Health Initiative Observational Cohort. Clin Chem 2009; 55:351–360Crossref, Medline, Google Scholar
46 : The association between childhood trauma and lipid levels in an adult low-income, minority population. Gen Hosp Psychiatry 2014; 36:150–155 Crossref, Medline, Google Scholar
47 : Influence of child abuse on adult depression: moderation by the corticotropin-releasing hormone receptor gene. Arch Gen Psychiatry 2008; 65:190–200Crossref, Medline, Google Scholar
48 : Association of FKBP5 polymorphisms and childhood abuse with risk of posttraumatic stress disorder symptoms in adults. JAMA 2008; 299:1291–1305Crossref, Medline, Google Scholar
49 : Chronic stress increases pituitary adenylate cyclase-activating peptide (PACAP) and brain-derived neurotrophic factor (BDNF) mRNA expression in the bed nucleus of the stria terminalis (BNST): roles for PACAP in anxiety-like behavior. Psychoneuroendocrinology 2009; 34:833–843Crossref, Medline, Google Scholar
50 : New insights into the central PACAPergic system from the phenotypes in PACAP- and PACAP receptor-knockout mice. Ann N Y Acad Sci 2006; 1070:75–89Crossref, Medline, Google Scholar
51 : Role of PACAP in the physiology and pathology of the sympathoadrenal system. Front Neuroendocrinol 2008; 29:128–141Crossref, Medline, Google Scholar
52 : Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature 2011; 470:492–497Crossref, Medline, Google Scholar
53 : Sex dependent influence of a functional polymorphism in steroid 5-α-reductase type 2 (SRD5A2) on post-traumatic stress symptoms. Am J Med Genet B Neuropsychiatr Genet 2013; 162B:283–292Crossref, Medline, Google Scholar
54 : Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry 1995; 52:1048–1060Crossref, Medline, Google Scholar
55 : Obesity indices and metabolic markers are related to hs-CRP and adiponectin levels in overweight and obese females. Obes Res Clin Pract 2013; 7:e315–e320Crossref, Medline, Google Scholar
56 : Predicting PTSD using the New York Risk Score with genotype data: potential clinical and research opportunities. Neuropsychiatr Dis Treat 2013; 9:517–527Crossref, Medline, Google Scholar
57 : PAC1 receptor (ADCYAP1R1) genotype is associated with PTSD’s emotional numbing symptoms in Chinese earthquake survivors. J Affect Disord 2013; 150:156–159Crossref, Medline, Google Scholar
58 : Psychosocial and behavioral predictors of inflammation in middle-aged and older adults: the Chicago health, aging, and social relations study. Psychosom Med 2006; 68:376–381Crossref, Medline, Google Scholar
59 : Socioeconomic status, C-reactive protein, immune factors, and responses to acute mental stress. Brain Behav Immun 2003; 17:286–295Crossref, Medline, Google Scholar
60 : A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. JAMA Psychiatry 2013; 70:31–41Crossref, Medline, Google Scholar



