Influences of Maternal and Paternal PTSD on Epigenetic Regulation of the Glucocorticoid Receptor Gene in Holocaust Survivor Offspring
Abstract
Objective
Differential effects of maternal and paternal posttraumatic stress disorder (PTSD) have been observed in adult offspring of Holocaust survivors in both glucocorticoid receptor sensitivity and vulnerability to psychiatric disorder. The authors examined the relative influences of maternal and paternal PTSD on DNA methylation of the exon 1F promoter of the glucocorticoid receptor (GR-1F) gene (NR3C1) in peripheral blood mononuclear cells and its relationship to glucocorticoid receptor sensitivity in Holocaust offspring.
Method
Adult offspring with at least one Holocaust survivor parent (N=80) and demographically similar participants without parental Holocaust exposure or parental PTSD (N=15) completed clinical interviews, self-report measures, and biological procedures. Blood samples were collected for analysis of GR-1F promoter methylation and of cortisol levels in response to low-dose dexamethasone, and two-way analysis of covariance was performed using maternal and paternal PTSD as main effects. Hierarchical clustering analysis was used to permit visualization of maternal compared with paternal PTSD effects on clinical variables and GR-1F promoter methylation.
Results
A significant interaction demonstrated that in the absence of maternal PTSD, offspring with paternal PTSD showed higher GR-1F promoter methylation, whereas offspring with both maternal and paternal PTSD showed lower methylation. Lower GR-1F promoter methylation was significantly associated with greater postdexamethasone cortisol suppression. The clustering analysis revealed that maternal and paternal PTSD effects were differentially associated with clinical indicators and GR-1F promoter methylation.
Conclusions
This is the first study to demonstrate alterations of GR-1F promoter methylation in relation to parental PTSD and neuroendocrine outcomes. The moderation of paternal PTSD effects by maternal PTSD suggests different mechanisms for the intergenerational transmission of trauma-related vulnerabilities.
Offspring of trauma survivors are at increased risk for mental and physical illness (1–3). Although studies of the intergenerational transmission of trauma traditionally emphasize the influence of exposure (1, 2), there is evidence for the effects of parental trauma-related psychopathology. Parental posttraumatic stress disorder (PTSD), but not Holocaust exposure, is associated with alterations in hypothalamic-pituitary-adrenal (HPA) axis function, including enhanced cortisol suppression following dexamethasone administration (4) and lower baseline cortisol levels (5), in offspring. Cortisol levels in infants of mothers who developed PTSD following exposure to the September 11th, 2001, World Trade Center attacks were lower than those of mothers who did not develop PTSD (6). These neuroendocrine findings in offspring of trauma survivors are similar to observations in Holocaust survivors and other trauma-exposed persons with PTSD (7).
Transmission of effects from parents to children is thought to be mediated by developmental programming of glucocorticoid signaling (8). Variations in maternal care in rats predict hippocampal glucocorticoid receptor gene expression and levels of cytosine methylation of the exon 17 promoter of the rat glucocorticoid receptor gene (9). Studies of postmortem human brain tissue have demonstrated an association between methylation status of the orthologous exon 1F promoter region in the human glucocorticoid receptor gene (NR3C1) in the hippocampus and history of childhood abuse (10). This same association between childhood adversity and GR-1F methylation has been observed in whole blood (leukocytes) (11, 12).
Although the majority of studies of transgenerational transmission of trauma effects highlight maternal influences, few studies have directly compared maternal and paternal effects on the offspring (1, 2). Among offspring of Holocaust survivors, maternal PTSD was associated with increased risk for developing PTSD, whereas paternal PTSD was associated with greater risk for major depressive disorder, an effect that emerged after controlling for the effect of maternal PTSD (13). Maternal PTSD has been more strongly associated with lower cortisol levels in Holocaust offspring compared with paternal PTSD (14), and we recently demonstrated that maternal PTSD is related to increased glucocorticoid receptor sensitivity (15). In a longitudinal study of adopted children, differential and interacting maternal and paternal effects on children’s cortisol variability was reported (16). Inconsistent, over-reactive parenting by mothers predicted lower cortisol variability, whereas a similar parenting style among fathers generally predicted higher cortisol variability in offspring but only in the absence of low maternal cortisol variability. Similarly, we recently observed that both 24-hour urinary cortisol excretion and cortisol nonsuppression following dexamethasone administration were higher in offspring with paternal PTSD in the absence of maternal PTSD but lower in those with maternal PTSD (15). In the present study, we examined the distinct influences of maternal and paternal PTSD on DNA methylation of the exon 1F promoter of the glucocorticoid receptor (GR-1F) gene in Holocaust survivor offspring. We hypothesized that maternal PTSD would be associated with lower offspring GR-1F promoter methylation and that paternal PTSD would be associated with higher GR-1F promoter methylation. We further hypothesized that GR-1F promoter methylation would inversely correlate with glucocorticoid receptor sensitivity.
Method
Participants
A total of 120 participants were recruited over a 2-year period (2010–2012), as previously described (15), and 95 participants completed study procedures. The study was approved by the institutional review board at the Icahn School of Medicine at Mount Sinai, and written informed consent was obtained. Parental Holocaust exposure was defined as 1) being interned in a Nazi concentration camp, 2) having witnessed and/or experienced torture, or 3) having to flee for one’s life or hide during the Nazi era. Although the main questions concerned the effect of maternal and/or paternal PTSD, a small group of demographically similar Jewish participants without parental PTSD, whose parents were not in Nazi-occupied Europe before and during World War II, were recruited in order to control for potential effects of Holocaust exposure.
Offspring of Holocaust survivors had to have been born after World War II or after their parents had escaped to safety and have at least one Holocaust survivor parent. Exclusion criteria were any history of psychotic disorder or bipolar illness, significant current alcohol or drug use, and the presence of current PTSD (to distinguish the effects of parental PTSD from those associated with expressed PTSD). Individuals were also excluded if they had a major medical condition or were taking systemic steroids.
Clinical Evaluation
Axis I diagnoses were determined by clinical psychologists using the Structured Clinical Interview for DSM-IV (17). Parental PTSD was determined by consensus of at least three clinicians based on the Parental PTSD Questionnaire, completed by the offspring, and a semistructured interview. The Parental PTSD Questionnaire was previously validated against direct clinician assessment of the parent (18) and includes offspring perceptions of the impact of the Holocaust on the offspring. To assess relevant psychiatric symptoms and early-life experiences, participants also completed measures such as the Beck Depression Inventory (19), the Spielberger State-Trait Anxiety Inventory (20), the Dissociative Experiences Scale (21), the Relationship Scales Questionnaire (22), and the Childhood Trauma Questionnaire (23), as well as a measure for perceived emotional health (24).
Cytosine Methylation Assessment
Basal morning blood samples were collected for assessment of GR-1F promoter methylation and GR-1F expression and of plasma cortisol levels. Peripheral blood mononuclear cells were purified from EDTA-pretreated blood, and DNA was extracted as previously described (25). Cytosine methylation was estimated across 39 C—phosphate—G (CpG) sites in the GR-1F promoter, using 30 clones per sample, in four batches (10, 25). Variability in the DNA bisulfate treatment between batches did not exceed 2%. The number of methylated clones at each of the 39 CpG sites was converted to a percentage and summed across the GR-1F promoter sequence to create a total methylation percentage. As expected, when methylation in CpG islands is low, the distribution of this variable is positively skewed, and it was transformed (natural logarithm) for analytic purposes. The number of CpG sites (out of a possible 39) showing methylation in any clone was also determined for each participant. The summed percent methylation and number of methylated sites were highly correlated (r=0.820, p<0.0005, N=95).
Leukocyte Type Determination
Since peripheral blood mononuclear cell-type composition may affect estimates of DNA methylation (26), the ratio of lymphocytes to monocytes was calculated (peripheral blood mononuclear cell ratio) as a proxy of peripheral blood mononuclear cell type and used as a covariate in methylation analyses.
GR-1F Expression
GR-1F transcript expression was run as a validation of methylation estimates of the corresponding promoter. RNA, from TRIzol-dissolved peripheral blood mononuclear cells (Life Technologies, Grand Island, N.Y.), was extracted and used for determination of the GR-1F expression by quantitative polymerase chain reaction as previously described (25). For details of the primers and probes used, see Table S1 in the data supplement accompanying the online edition of this article. Data analysis was performed using the qBase v2.5 software (Biogazelle NV, Zwijnaarde, Belgium).
Hormone Determination
Day-1 cortisol levels and day-2 cortisol and dexamethasone levels obtained in association with the low-dose (0.5 mg) dexamethasone suppression test were determined by radioimmunoassay as previously described (25).
Statistical Analyses
A two-way analysis of covariance (ANCOVA) was used to examine the effects of the presence or absence of maternal and paternal PTSD on offspring GR-1F promoter methylation. Because a significant percentage of Holocaust offspring indicated no lifetime parental PTSD, non-Holocaust offspring were compared with the Holocaust offspring with no parental PTSD on key demographic and clinical variables (see Table S2 in the online data supplement). Because no relevant differences were observed, participants from both groups were coded as having no maternal or paternal PTSD. To identify effects of parental PTSD, rather than Holocaust exposure, the presence or absence of maternal and paternal Holocaust exposure was included as a covariate. Other covariates were age (27), lifetime smoking (pack-years, associated with percent methylation: N=95, r=−0.315, p=0.002; number of methylated sites: N=95, r=−0.301, p=0.003), and peripheral blood mononuclear cell type (26). There were no associations of GR-1F promoter methylation with gender, and including gender as a covariate did not affect any of the analyses. Bonferroni post hoc tests were conducted to investigate significant effects. Pearson’s correlational analysis was used to investigate the association of GR-1F promoter methylation with gene expression. A correlation between GR-1F promoter methylation and cortisol decline following dexamethasone administration was performed, partialling out the effects of dexamethasone levels, body mass index, age, smoking history, and peripheral blood mononuclear cell type.
Phenotypic Clustering
Psychological and clinical data were subject to an unsupervised hierarchical clustering analysis, which allowed phenotypic clustering in relation to maternal and paternal PTSD. Data derived from the clinical rating scales were log2 transformed, and z-scores were calculated. Contrast was enhanced by ensuring that high numbers reflected more deleterious outcomes. An initial clustering analysis was performed using an average linkage-clustering algorithm with the output expressed using Euclidean distance (CIMminer tool; Genomics and Bioinformatics Group, Laboratory of Molecular Pharmacology, Center for Cancer Research, National Cancer Institute, Bethesda, Md.). This was followed by a clustering analysis adding GR-1F promoter percent methylation to determine whether this measure would cluster with any of the identified phenotype-by-group-clusters. The data are presented in a heat map that includes dendrograms of the phenotype and group clusters. Two-way analyses of variance (ANOVAs) were conducted to test the statistical significance of the relationships of the phenotype measures included in the clustering analysis with maternal and paternal PTSD.
Results
Demographic and Clinical Characteristics
Demographic and clinical data based on the presence or absence of maternal and paternal PTSD are summarized in Table 1. Seventy-five percent of Holocaust offspring had two Holocaust-exposed parents. PTSD was reported for 55.8% (N=53) of mothers and 44.2% (N=42) of fathers; only 15 Holocaust offspring reported no parental PTSD. Participants with maternal PTSD were slightly older than those with paternal PTSD, and those with paternal PTSD only were the youngest. There were no differences in gender, body mass index, education, or current axis I psychopathology. There were differences in lifetime rates of depression and anxiety disorders, such that those without parental PTSD had lower rates of these disorders than those with maternal or paternal PTSD.
| Characteristic | Parental PTSD | 2×2 Analysis of Variance | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No Parental PTSD (N=31) | Paternal PTSD (N=11) | Maternal PTSD (N=22) | Both Parents With PTSD (N=31) | Maternal PTSD | Paternal PTSD | Maternal-by-Paternal PTSD | |||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | F | df | p | F | df | p | F | df | p | |
| Age (years) | 57.13 | 7.78 | 47.64 | 7.93 | 57.36 | 8.10 | 58.68 | 7.91 | 10.10 | 1, 95 | 0.002 | 5.31 | 1, 95 | 0.02 | 9.28 | 1, 95 | 0.003 |
| Body mass index | 27.32 | 4.87 | 24.37 | 4.97 | 26.84 | 5.04 | 25.87 | 4.96 | 0.21 | 1, 95 | n.s. | 3.15 | 1, 95 | n.s. | 0.80 | 1, 95 | n.s. |
| Education (years) | 18.00 | 2.46 | 17.73 | 2.49 | 17.18 | 2.54 | 17.45 | 2.51 | 0.97 | 1, 95 | n.s. | 0.00 | 1, 95 | n.s. | 0.24 | 1, 95 | n.s. |
| Chi-Square Analysis | |||||||||||||||||
| N | % | N | % | N | % | N | % | χ2 | df | p | |||||||
| Female | 16 | 51.6 | 7 | 63.6 | 18 | 81.8 | 25 | 80.6 | 6.52 | 3 | n.s. | ||||||
| Major depressive disordera | |||||||||||||||||
| Current | 1 | 3.2 | 1 | 9.1 | 3 | 13.6 | 0 | 0.0 | 5.06 | 3 | n.s. | ||||||
| Lifetime | 11 | 35.5 | 8 | 72.7 | 16 | 72.7 | 26 | 83.9 | 15.75 | 3 | 0.001 | ||||||
| Anxiety disorderb | |||||||||||||||||
| Current | 9 | 29.0 | 6 | 54.5 | 13 | 59.1 | 15 | 48.4 | 4.50 | 3 | n.s. | ||||||
| Lifetime | 9 | 29.0 | 7 | 63.6 | 16 | 72.7 | 18 | 58.1 | 9.71 | 3 | 0.02 | ||||||
Maternal PTSD, Paternal PTSD, and GR-1F Promoter Methylation
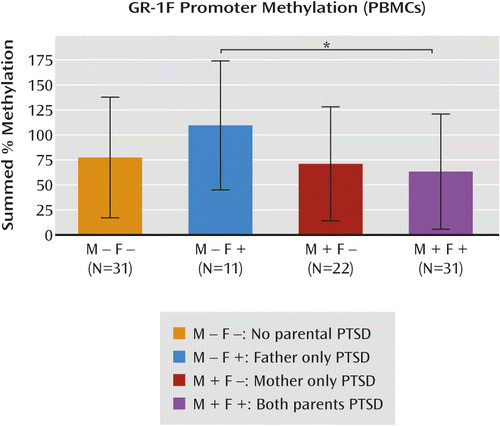
a Results are based on the presence or absence of maternal and paternal posttraumatic stress disorder, controlling for parental Holocaust exposure, age, smoking history, and PBMC type. The represented data (mean, standard deviation) are based on an analysis of covariance using raw data. The post hoc statistic (*) is from the analysis using transformed (natural logarithm) data.
Correlations Between GR-1F Promoter Methylation and GR-1F Expression and Functional Outcomes
GR-1F promoter methylation was also associated with the cortisol response to the low-dose dexamethasone suppression test. Partial correlation demonstrated a negative association between GR-1F promoter percent methylation and cortisol decline following dexamethasone administration, such that greater cortisol suppression was associated with lower methylation (r=−0.249, df=82, p=0.03).
Phenotypic Clustering
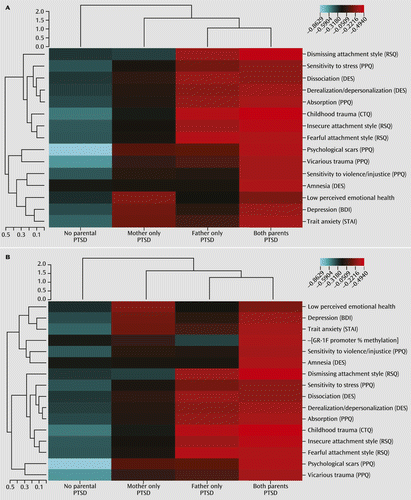
a Rows represent different phenotypes that are also presented in Table 2. Columns represent experimental groups (no parental PTSD, mother only PTSD, father only PTSD, both parents PTSD). The unsupervised hierarchical clustering is presented in dendrograms, with vertical dendrograms representing phenotypic clustering, horizontal dendrograms representing group clustering, and the dendrogram scales represent Euclidean distances. The heat map depicts phenotype-by-group clusters with the color scale based on a phenotype z-score, where blue represents a low score and red represents a high score. Panel A and panel B contain the same phenotypic variables with the exception of percent methylation in the exon 1F promoter of the glucocorticoid receptor gene, which is presented only in panel B. BDI=Beck Depression Inventory; CTQ=Childhood Trauma Questionnaire; DES=Dissociative Experiences Scale; PPQ=Parental PTSD Questionnaire; RSQ=Relationship Scales Questionnaire; STAI=Spielberger State-Trait Anxiety Inventory.
To provide further interpretability, a series of separate two-way ANOVAs were conducted to determine the relative influence of maternal and paternal PTSD on the clinical indicators included in the clustering analysis (Table 2). Maternal and paternal PTSD were associated with different clinical and perceived childhood characteristics. Maternal PTSD was associated with higher self-reported depressive symptoms and trait anxiety and lower perceived emotional health. Paternal PTSD was associated with higher reports of childhood trauma and less adaptive attachment styles. There were no significant interactions between maternal and paternal PTSD on any of the clinical measures.
| Characteristic | Parental PTSD | Analysis | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No Parental PTSD (N=31) | Paternal PTSD (N=11) | Maternal PTSD (N=22) | Both Parents With PTSD (N=31) | Maternal PTSD | Paternal PTSD | Maternal PTSD-by-Paternal PTSD | |||||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | F | df | p | F | df | p | F | df | p | |
| Beck Depression Inventory score | 7.9 | 7.67 | 10.6 | 7.63 | 12.7 | 7.67 | 13.4 | 7.79 | 5.00a | 1, 91 | 0.03 | 0.92 | 1, 91 | n.s. | 0.35 | 1, 91 | n.s. |
| Childhood Trauma Questionnaire total score | 6.9 | 2.19 | 9.1 | 2.32 | 7.6 | 2.40 | 9.3 | 2.23 | 0.71 | 1, 90 | n.s. | 13.87a | 1, 90 | 0.00 | 0.256 | 1, 90 | n.s. |
| Dissociative Experiences Scale | |||||||||||||||||
| Absorption | 5.6 | 7.12 | 8.6 | 7.30 | 6.2 | 7.67 | 11.4 | 7.24 | 1.05 | 1, 89 | n.s. | 2.86 | 1, 89 | n.s. | 0.40 | 1, 89 | n.s. |
| Amnesia | 1.4 | 3.29 | 1.8 | 3.65 | 1.5 | 3.84 | 3.8 | 3.34 | 1.63 | 1, 89 | n.s. | 6.10a | 1, 89 | 0.02 | 1.31 | 1, 89 | n.s. |
| Derealization/Depersonalization | 0.9 | 4.93 | 2.3 | 5.31 | 1.7 | 5.28 | 4.0 | 5.01 | 1.18 | 1, 89 | n.s. | 2.44 | 1, 89 | n.s. | 0.15 | 1, 89 | n.s. |
| Dissociation | 1.6 | 5.48 | 3.6 | 5.31 | 2.5 | 5.75 | 5.2 | 5.57 | 1.07 | 1, 89 | n.s. | 3.80 | 1, 89 | 0.06 | 0.08 | 1, 89 | n.s. |
| Perceived emotional health | 3.7 | 1.10 | 3.5 | 0.99 | 3.1 | 0.96 | 3.1 | 1.11 | 4.71a | 1, 91 | 0.03 | 0.19 | 1, 91 | n.s. | 0.07 | 1, 91 | n.s. |
| Parental PTSD Questionnaire (PPQ) score | |||||||||||||||||
| Psychological scarsb | 2.4 | 1.64 | 3.7 | 1.33 | 3.8 | 1.44 | 4.2 | 1.11 | 7.94a | 1, 75 | 0.006 | 7.15a | 1, 75 | 0.009 | 2.0, | 1, 75 | n.s. |
| Sensitivity to violence/injusticec | 3.4 | 1.64 | 3.7 | 0.99 | 3.8 | 0.96 | 4.3 | 1.11 | 3.57 | 1, 75 | 0.06 | 2.38 | 1, 75 | n.s. | 0.19 | 1, 75 | n.s. |
| Sensitivity to stressd | 2.4 | 2.19 | 3.3 | 1.66 | 2.8 | 1.44 | 3.3 | 1.67 | 0.39 | 1, 75 | n.s. | 3.98a | 1, 75 | 0.05 | 0.21 | 1, 75 | n.s. |
| Vicarious traumae | 2.9 | 1.64 | 3.7 | 1.33 | 3.6 | 1.44 | 4.2 | 1.11 | 3.78 | 1, 75 | 0.06 | 5.01a | 1, 75 | 0.03 | 0.27 | 1, 75 | n.s. |
| Relationship Scales Questionnaire | |||||||||||||||||
| Dismissing attachment | 3.1 | 0.55 | 3.6 | 0.66 | 3.1 | 0.48 | 3.7 | 0.56 | 0.068 | 1, 89 | n.s. | 11.27a | 1, 89 | 0.001 | 0.08 | 1, 89 | n.s. |
| Fearful attachment | 2.6 | 1.10 | 3.5 | 0.99 | 2.9 | 0.96 | 3.5 | 1.11 | 0.347 | 1, 89 | n.s. | 10.29a | 1, 89 | 0.002 | 0.62 | 1, 89 | n.s. |
| Secure attachment | 3.4 | 0.55 | 2.8 | 0.66 | 3.1 | 0.96 | 2.8 | 0.56 | 0.80 | 1, 89 | n.s. | 8.17a | 1, 89 | 0.005 | 0.19 | 1, 89 | n.s. |
| Preoccupied attachment | 2.8 | 0.55 | 2.7 | 0.66 | 2.9 | 0.96 | 3.0 | 0.56 | 1.56 | 1, 89 | n.s. | 0.10 | 1, 89 | n.s. | 0.66 | 1, 89 | n.s. |
| Spielberger State-Trait Anxiety Inventory | |||||||||||||||||
| State | 14.0 | 11.50 | 21.3 | 1.13 | 21.2 | 11.99 | 18.4 | 11.14 | 0.73 | 1, 89 | n.s. | 0.74 | 1, 89 | n.s. | 3.88 | 1, 89 | 0.06 |
| Trait | 17.0 | 11.50 | 21.7 | 1.16 | 23.6 | 11.99 | 26.9 | 11.69 | 5.13a | 1, 89 | 0.03 | 2.38 | 1, 89 | n.s. | 0.07 | 1, 89 | n.s. |
Discussion
This is the first study, to our knowledge, to demonstrate alterations of GR-1F promoter methylation in relation to maternal and paternal PTSD. Maternal PTSD moderated the effect of paternal PTSD on GR-1F promoter methylation. Paternal PTSD, only in the absence of maternal PTSD, was associated with higher levels of GR-1F promoter methylation, while offspring with both maternal and paternal PTSD displayed the lowest level of methylation. Although the sample size was limited, findings are consistent with those of previous reports on the effects of parental PTSD on offspring phenotype. In a different cohort of Holocaust offspring, we observed that maternal PTSD significantly enhanced the risk for PTSD, while paternal PTSD significantly elevated the risk for depression when controlling for maternal PTSD (13). The hierarchical clustering analysis similarly revealed that maternal and paternal PTSD were associated with different offspring psychological characteristics and that lower GR-1F promoter methylation was specifically associated with maternal PTSD.
In this study, phenotypic clustering analysis also demonstrated an association of paternal PTSD, but not maternal PTSD, with childhood abuse and neglect, which is consistent with findings in offspring of male war veterans with PTSD (28). It is notable that the Childhood Trauma Questionnaire assesses a range of potentially traumatic experiences but does not identify the perpetrator. Thus, it cannot be concluded that fathers with PTSD are more likely than mothers to abuse their children. The finding of relatively higher GR-1F promoter methylation with paternal PTSD (in the absence of maternal PTSD) is consistent with a previous finding in postmortem human hippocampus of an association between higher GR-1F promoter methylation and childhood abuse (10), as well as with more recent findings of an association of higher methylation of specific CpG sites within the GR-1F promoter in whole blood (leukocytes) with lower parental care, higher childhood maltreatment, and parental loss in healthy adults (11) and with childhood sexual abuse and extent of childhood maltreatment in individuals with borderline personality disorder (12). The type, severity, chronicity, and developmental stage of adversity, as well as sex of the parent and offspring, may be critical for these associations. Unfortunately, our sample size was too small to investigate the influence of these factors.
The hypothesis that GR-1F methylation would be associated with early adversity was based on an interpretation of differences in offspring methylation status on the orthologous promoter region in the rat, based on naturally occurring variations of maternal care (9). The implication of rodent studies of the effect of maternal care on offspring is that early-life challenges, including variations in parental care, are capable of producing enduring epigenetic changes resulting in sustained phenotypic outcomes in the offspring, including altered stress reactivity. The impact of these effects will depend on context and the degree to which the resulting phenotypic variation meets the particular demands of the prevailing environment. Chronic stress produces variations in the maternal care of the rat (i.e., reduced pup licking/grooming) that are associated with methylation-mediated decreased hippocampal glucocorticoid receptor expression and increased HPA responses to stress (29, 30). This increased stress reactivity may be considered adaptive in highly adverse conditions (31). In Holocaust offspring, a parent may have similarly primed his or her offspring for a highly threatening environment that in a post-Holocaust context results in an overgeneralized and exacerbated fear response.
Although offspring with paternal PTSD reported more childhood trauma, the effect of paternal PTSD differed depending on the presence of maternal PTSD. This raises the possibility of indirect effects, such that PTSD in one parent may affect the parenting of the other or create a qualitative shift in the family environment. For example, paternal PTSD has been associated with higher levels of family conflict in this sample (15). It seems that paternal PTSD alone produces effects such as higher cortisol excretion, reduced glucocorticoid receptor sensitivity, and higher GR-1F promoter methylation, similar to those seen in other samples with major depressive disorder and history of childhood abuse (10, 32, 33). When both parents have PTSD, offspring phenotype, including lower GR-1F promoter methylation, appears instead to resemble the biology of PTSD risk or expression (7, 34). It is possible that when both parents are traumatized or express symptoms, there is no buffer for the child, resulting in an unmediated experience of constant or repeated threat imposed by the unpredictability of parental behavior and resulting in the expression of hypervigilance in the offspring. Because most of the offspring in the present study were raised in the social context of the late 1940s–1960s, with families structured around traditional gender roles, mothers were more likely to be the primary caregiver, whereas many offspring described fathers who worked long hours and were somewhat distant from the family unit. Thus, social context may moderate the processes underlying the intergenerational transmission of trauma (35). While we were able to show different offspring phenotypes based on maternal compared with paternal PTSD, delineations of early parent-child and interparental interactions that may mediate these relationships require further empirical investigation.
The functional relevance of the differences in methylation is apparent in the negative correlation between GR-1F promoter methylation and expression of the respective transcript (i.e., GR-1F transcript). Furthermore, lower levels of GR-1F promoter methylation were associated with enhanced glucocorticoid negative feedback inhibition assessed by the dexamethasone suppression test, suggesting that the epigenetic status in blood of offspring resulting from parental PTSD reflects neuroendocrine function. We previously suggested that glucocorticoid receptor sensitivity as assessed by the cortisol response to dexamethasone may be a relatively stable marker of risk in Holocaust offspring (4, 15). Furthermore, it has previously been suggested that glucocorticoid receptor binding in peripheral tissue (peripheral blood mononuclear cells) is sufficiently stable to associate with PTSD risk as a trait (36). To our knowledge, there has only been one other study demonstrating an association between GR-1F promoter methylation and glucocorticoid receptor sensitivity (25); however, associations with reduced negative feedback inhibition have been observed in association with higher methylation of specific CpG sites on the GR-1F promoter (11). A similar moderating effect of maternal PTSD on the effects of paternal PTSD was observed in the same participants examined in the present study with respect to both the cortisol response to dexamethasone and 24-hour urinary cortisol excretion (15). A maternal PTSD effect associated with increased glucocorticoid receptor sensitivity in peripheral tissue was also observed (15). Since DNA methylation is a chemically stable epigenetic mark, these findings suggest that the differences in GR-1F promoter methylation might mediate the variation in glucocorticoid receptor function and HPA-axis feedback sensitivity that associate with the transgenerational transmitted effects of Holocaust-related psychopathology trauma on the risk for PTSD in the offspring.
The exact mechanisms underlying this transgenerational transmission are unknown. The offspring in this sample were conceived after, and in some cases decades after, parental Holocaust exposure. However, studies with rodents demonstrate that preconception paternal stress can influence behavior and biology in progeny through epigenetic changes in sperm (37), such that germ-line transmission of epigenetic marks associated with PTSD risk is possible. Additionally, variations in the methylation status of the GR-1F promoter can occur in association with pre- as well as postnatal exposures (10, 11, 38, 39). Moreover, stress in one parent may also exert indirect effects on offspring through its influence on the other parent (e.g., paternally driven maternal effects) (40). Finally, there may be direct effects on parent-offspring interactions that are ultimately reflected in the epigenome.
Holocaust offspring who met criteria for PTSD were excluded from this study in order to better focus on effects associated with transmission of vulnerability and not expressed PTSD. While the presence of psychiatric disorder represents a clear expression of illness vulnerability, it is also useful to identify more subtle or cumulative effects resulting from maternal and paternal PTSD and their interaction. The psychological constructs in the clustering analysis demonstrate aggregate phenotypic differences in both the early environment of the offspring and more distal indicators of offspring distress and functioning. In addition to higher reported levels of childhood trauma, paternal PTSD was associated with a dismissing, fearful, or insecure attachment style, as well as more dissociative experiences and greater sensitivity to violence. Maternal PTSD was associated with more self-reported symptoms of depression and higher trait anxiety, which is consistent with findings of an association between maternal Holocaust exposure and higher levels of psychological distress in offspring (24). It is possible that the higher self-reported depression symptoms associated with maternal PTSD are best understood as an index of distress or negative affectivity, particularly in a sample in which PTSD is excluded and with low rates of current major depressive disorder.
Such fine-grained phenotypic distinctions suggest important differences in the expression and effect of maternal and paternal PTSD on offspring phenotype that contribute to psychiatric vulnerability. Research on the intergenerational transmission of trauma requires nuanced assessment of both potential mechanisms of transmission (e.g., parenting and attachment) and indicators of vulnerability and phenotypic differences that represent cumulative effects of being raised from birth by traumatized, symptomatic parents.
1 : Intergenerational effects of war trauma among Palestinian families mediated via psychological maltreatment. Child Abuse Negl 2013; 37:955–968Crossref, Medline, Google Scholar
2 : Parental styles in second generation effects of genocide stemming from the Khmer Rouge regime in Cambodia. Attach Hum Dev 2011; 13:611–628Crossref, Medline, Google Scholar
3 : Vulnerability to posttraumatic stress disorder in adult offspring of Holocaust survivors. Am J Psychiatry 1998; 155:1163–1171Link, Google Scholar
4 : Effects of parental PTSD on the cortisol response to dexamethasone administration in their adult offspring. Am J Psychiatry 2007; 164:163–166Link, Google Scholar
5 : Parental posttraumatic stress disorder as a vulnerability factor for low cortisol trait in offspring of Holocaust survivors. Arch Gen Psychiatry 2007; 64:1040–1048Crossref, Medline, Google Scholar
6 : Transgenerational effects of posttraumatic stress disorder in babies of mothers exposed to the World Trade Center attacks during pregnancy. J Clin Endocrinol Metab 2005; 90:4115–4118Crossref, Medline, Google Scholar
7 : Post-traumatic stress disorder. N Engl J Med 2002; 346:108–114Crossref, Medline, Google Scholar
8 : Stress-related psychiatric disorders with low cortisol levels: a metabolic hypothesis. Endocrinology 2011; 152:4496–4503Crossref, Medline, Google Scholar
9 : Epigenetic programming by maternal behavior. Nat Neurosci 2004; 7:847–854Crossref, Medline, Google Scholar
10 : Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat Neurosci 2009; 12:342–348Crossref, Medline, Google Scholar
11 : Childhood adversity and epigenetic modulation of the leukocyte glucocorticoid receptor: preliminary findings in healthy adults. PLoS ONE 2012; 7:e30148Crossref, Medline, Google Scholar
12 : Increased methylation of glucocorticoid receptor gene (NR3C1) in adults with a history of childhood maltreatment: a link with the severity and type of trauma. Transcult Psychiatry 2011; 1:e59Crossref, Google Scholar
13 : Maternal, not paternal, PTSD is related to increased risk for PTSD in offspring of Holocaust survivors. J Psychiatr Res 2008; 42:1104–1111Crossref, Medline, Google Scholar
14 : Transgenerational transmission of cortisol and PTSD risk. Prog Brain Res 2008; 167:121–135Crossref, Medline, Google Scholar
15 : Maternal PTSD associates with greater glucocorticoid sensitivity in offspring of Holocaust survivors. Psychoneuroendocrinology 2014; 40:213–220Crossref, Medline, Google Scholar
16 : Disentangling the effects of genetic, prenatal and parenting influences on children’s cortisol variability. Stress 2013; 16:607–615Crossref, Medline, Google Scholar
17 : Structured Clinical Interview for DSM-IV (SCID). New York, New York State Psychiatric Institute, Biometrics Research, 1995Google Scholar
18 : Are adult offspring reliable informants about parental PTSD? a validation study. Ann N Y Acad Sci 2006; 1071:484–487Crossref, Medline, Google Scholar
19 : A systematic investigation of depression. Compr Psychiatry 1961; 2:163–170Crossref, Medline, Google Scholar
20 : The State-Trait Anxiety Inventory (S T A I): Test Manual for Form X. Palo Alto, Calif, Consulting Psychologists Press, 1968Google Scholar
21 : Development, reliability, and validity of a dissociation scale. J Nerv Ment Dis 1986; 174:727–735Crossref, Medline, Google Scholar
22 : Models of the self and other: fundamental dimensions underlying measures of adult attachment. J Pers Soc Psychol 1994; 67:430Crossref, Google Scholar
23 : Development and validation of a brief screening version of the Childhood Trauma Questionnaire. Child Abuse Negl 2003; 27:169–190Crossref, Medline, Google Scholar
24 : Maternal exposure to the Holocaust and health complaints in offspring. Dis Markers 2011; 30:133–139Crossref, Medline, Google Scholar
25 : Epigenetic biomarkers as predictors and correlates of symptom improvement following psychotherapy in combat veterans with PTSD. Front Psychiatry 2013; 4:118Crossref, Medline, Google Scholar
26 : Factors underlying variable DNA methylation in a human community cohort. Proc Natl Acad Sci USA 2012; 109(suppl 2):17253–17260Crossref, Medline, Google Scholar
27 : Epigenetic differences arise during the lifetime of monozygotic twins. Proc Natl Acad Sci USA 2005; 102:10604–10609Crossref, Medline, Google Scholar
28 : Childhood maltreatment in adult offspring of Portuguese war veterans with and without PTSD. Eur J Psychotraumatol (EPub Feb 4, 2014)Google Scholar
29 : Dysfunctional nurturing behavior in rat dams with limited access to nesting material: a clinically relevant model for early-life stress. Neuroscience 2008; 154:1132–1142Crossref, Medline, Google Scholar
30 : Stress during gestation alters postpartum maternal care and the development of the offspring in a rodent model. Biol Psychiatry 2006; 59:1227–1235Crossref, Medline, Google Scholar
31 : Maternal care and hippocampal plasticity: evidence for experience-dependent structural plasticity, altered synaptic functioning, and differential responsiveness to glucocorticoids and stress. J Neurosci 2008; 28:6037–6045Crossref, Medline, Google Scholar
32 : Glucocorticoid receptors in major depression: relevance to pathophysiology and treatment. Biol Psychiatry 2001; 49:391–404Crossref, Medline, Google Scholar
33 : Pituitary-adrenal and autonomic responses to stress in women after sexual and physical abuse in childhood. JAMA 2000; 284:592–597Crossref, Medline, Google Scholar
34 : Lower methylation of glucocorticoid receptor gene promoter 1F in peripheral blood of veterans with post-traumatic stress disorder. Biol Psychiatry (Epub ahead of print, Feb 17, 2014)Google Scholar
35 : How does the social environment ‘get into the mind’? epigenetics at the intersection of social and psychiatric epidemiology. Soc Sci Med 2012; 74:67–74Crossref, Medline, Google Scholar
36 : Pre-existing high glucocorticoid receptor number predicting development of posttraumatic stress symptoms after military deployment. Am J Psychiatry 2011; 168:89–96Link, Google Scholar
37 : Epigenetic transmission of the impact of early stress across generations. Biol Psychiatry 2010; 68:408–415Crossref, Medline, Google Scholar
38 : Transgenerational impact of intimate partner violence on methylation in the promoter of the glucocorticoid receptor. Transcult Psychiatry 2011; 1:e21Crossref, Google Scholar
39 : Prenatal exposure to maternal depression, neonatal methylation of human glucocorticoid receptor gene (NR3C1) and infant cortisol stress responses. Epigenetics 2008; 3:97–106Crossref, Medline, Google Scholar
40 : Epigenetics and the origins of paternal effects. Horm Behav 2011; 59:306–314Crossref, Medline, Google Scholar



