Late-Life Depression: A Role for Accelerometer Technology in Diagnosis and Management
“Mr. G,” an 81-year-old Caucasian man with a history of mild neurocognitive disorder and unspecified bipolar or related disorder with atypical features (mood reactivity), was referred by his neurologist for a geriatric psychiatry assessment at the Senior Behavioral Health outpatient program at the University of California, San Diego. At the time of his initial presentation, the patient’s primary complaint was “my head feels as if it’s in a fog.” It was noteworthy that this patient was both affluent and very high functioning at baseline and ran a large business. He had concerns about his ability to function at work. The patient presented with a complicated medical history that included carotid stenosis with transient ischemic attacks (treated with stenting) and hypopharyngeal carcinoma, which required radiation treatment, which in turn caused mandibular osteonecrosis. This subsequently led to mandibular reconstruction surgery 2 years before his initial presentation. The patient also had right preauricular basal cell carcinoma (resected), low testosterone requiring supplementation, hypertension, and hypercholesterolemia. He had been diagnosed with severe carotid stenosis 1 year before presentation. He reported that he had experienced an episode of dizziness and transient unconsciousness (secondary to cerebral hypoxia), which led to the diagnosis of stenosis and emergent stenting of his carotid arteries. At initial presentation, Mr. G was taking aripiprazole at an atypical dosage of 2 mg once a week and fluoxetine at 20 mg/day for depression. He was also taking four additional medications at bedtime for insomnia: melatonin, 2.5 mg; temazepam, 15 mg; mirtazapine, 15 mg; and zaleplon, 10 mg. His nonpsychotropic medications included amlodipine, 5 mg/day; aspirin, 81 mg/day; levothyroxine, 150 mcg/day; lisinopril, 10 mg/day; lovastatin, 40 mg/day; tamsulosin, 0.4 mg at bedtime; and testosterone supplementation. The patient scored 20 out of a possible score of 30 on the Montreal Cognitive Assessment, which is consistent with mild neurocognitive disorder. A review of his performance on a previous administration of the assessment indicated that his score had dropped by 6 points over the past 14 months.
Mr. G’s prior psychiatric history was significant for a major depressive episode that began after he underwent the carotid stenting procedure 1 year before presentation. At the time, he was treated with fluoxetine at 40 mg/day and aripiprazole at 5 mg/day. However, this treatment precipitated what was described as a hypomanic episode, the defining features of which included impulsive business decisions, pressured speech, and insomnia. He was taken off fluoxetine and aripiprazole, and his hypomania resolved. Approximately 2 months before presentation, the patient demonstrated some amotivation and low energy, and he was restarted on fluoxetine at 20 mg/day by his primary care physician, but with minimal effect.
The patient’s physical examination revealed orthostatic hypotension, but his vital signs were otherwise within normal range. A comprehensive laboratory investigation, which included a complete blood count with differential analysis, a comprehensive metabolic panel, liver function tests, a cardiac workup, and assessment for hypothyroidism, hypogonadism, and low vitamin levels, was found to be noncontributory. A volumetric MRI of the brain demonstrated stable age-related volume loss and superimposed white matter changes suggesting chronic microvascular ischemia, but was otherwise found to be unremarkable. His hippocampal volume was found to be higher than the 5th percentile, and his lateral ventricular volume was at the 79th percentile, both within normal limits.
Mr. G was relatively nonspecific when describing his clinical complaints, using terms like “my brain is in a fog,” and he reported both hypersomnia and insomnia at different points in the initial assessment. His wife, who was present during the initial session as well as subsequent sessions, provided reliable collateral information, but stated that she was not always around him and not always observing him. He denied depressed mood, episodes of tearfulness, anhedonia, feelings of worthlessness, changes in appetite, thoughts of death, or fatigue. There was no initial reported history of psychomotor retardation.
We discussed referral for a formal sleep study (including polysomnography and multiple sleep latency tests), but polypharmacy was identified as a primary clinical issue initially, and we decided to defer this referral until after his sedative medications had been reduced. Initially, zaleplon and melatonin were discontinued. His mirtazapine dosage was then reduced to 7.5 mg at bedtime, but he reported recurrence of insomnia, and his nightly dose was increased back to 15 mg. Mr. G noted that he had been on temazepam for over 30 years and had relied on it for sleep. He became anxious at the notion of tapering off this medication, so this was deferred to a later time. Despite long-term use of temazepam, the reported confusion and “brain fog” were relatively recent complaints. In consultation with the patient’s primary care provider, Mr. G’s amlodipine was incrementally discontinued to address his hypotension, but this resulted in an exacerbation of hypertension, with his blood pressure reaching a high of 214/112 mmHg. Subsequently, amlodipine was restarted at 2.5 mg/day, which resulted in successful management of his hypertension without hypotension.
Assessment of Mr. G’s level of functioning suggested ongoing confusion and poor decision making over the course of the first month after resuming treatment with fluoxetine. This was the primary complaint of the patient and his wife, both of whom repeatedly said they were concerned about his ability to manage his business in his current mental state.
Because of unclear objective information about the patient’s sleep pattern, at this point, we recommended that the patient use a wrist-worn actigraph to record his daily movement as well as his sleep. Actigraphs are wearable devices designed to measure, map, and record the wearer’s motion. The primary sensor in an actigraph is an accelerometer, which measures changes in its own acceleration and thus can detect and map its own motion in three dimensions. Accelerometer technology is utilized in a broad range of industries (e.g., automobile, structural engineering, machinery development). Actigraphy, which relies on this technology, has become increasingly popular and accessible over the past 5 years, as wrist-worn devices have become more accurate and compact. The patient was receptive to the idea of actigraphy and purchased a device called “UP” (Jawbone, San Francisco). The patient received education on how to use the device to keep track of his daily step count and to record his movements during sleep, as well as to synchronize it with an app on his smartphone for visualization of data.
After monitoring for 1 week, it was noted at the next clinic visit that the patient had slept for an average of 8–9 hours of good quality sleep, ruling out insomnia. However, a review of his daytime motion data indicated that over the preceding 5 days, the patient had taken an average of only 836 steps per day (Figure 1). Moreover, this number was driven by a single day, when he had taken a long walk, at the insistence of his wife. With that day excluded, his average number of steps was 440 per day.
Further inquiry revealed that the patient lived in a very large house, and this step count was achieved simply by going from his bedroom to the bathroom or the dining room and back, and that the patient was spending the majority of his day lying in bed.
This newly identified severe psychomotor retardation, in combination with previously reported evidence of poor energy, low motivation, indecisiveness and confusion, and objective evidence of adequate sleep, led us to reconsider the treatment plan. We increased Mr. G’s dosage of fluoxetine to 40 mg/day and augmented this with aripiprazole at 2 mg/day. Over the course of the next 3 weeks, Mr. G demonstrated improvement in mood as well as energy, and his actigraphy data revealed that his average step count increased to approximately 4,000 steps per day. Moreover, when asked to provide information on his subjective experience using the device, Mr. G noted that he had found it helpful to keep track of his own movements and sleep and that being able to visualize the increase in his step count as his depression improved provided encouragement as well as positive reinforcement for remaining engaged in exercise. His wife supported this, reporting that the patient was meticulous about recording his step counts and that he had mentioned looking forward to sharing the data with his psychiatrist at his weekly follow-up visits.
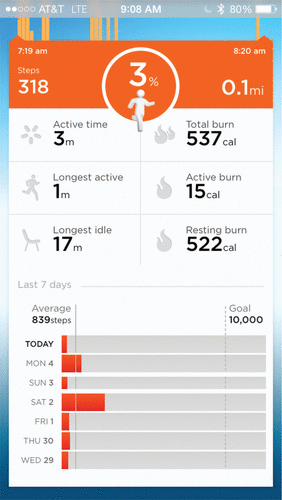
FIGURE 1. Image of Patient’s Cellphone, Indicating Psychomotor Retardation
It is well established that depressive symptoms in late life can be challenging to diagnose because of heterogeneity of clinical presentation (1, 2). Moreover, older adults with depressive symptoms frequently fail to meet DSM criteria for depressive episodes. Older adults also frequently present with subsyndromal depressive symptoms, and the prevalence of subsyndromal depression may be up to three times higher than that for major depression in this population (3, 4). In older adults like Mr. G in the vignette, with comorbid mood symptoms and cognitive impairment, clinical information as well as collateral information may be unreliable or biased. This case highlights how accelerometer-based technology can have an impact on several dimensions of diagnosis and management of depressive symptoms in late life by providing a more accurate clinical understanding of specific factors that can be key determinants of care. Currently available devices are able, at a minimum, to provide quantified information on sleep and physical activity, both of which are diagnostically pertinent factors for clinicians. Here, we discuss key clinical issues related to depressive symptoms in late life where use of accelerometer-based technology may have a role.
Sleep Disturbances and Depression
Sleep disruption in older adults may include insomnia (taking more than 30 minutes to fall asleep or waking up earlier than desired) as well as short duration of sleep. Insomnia is frequently comorbid with depression and can also represent a risk factor for subsequent depression (5). Prevalence of insomnia symptoms may be as high as 75% among older adults (6). Across studies, both subjectively reported insomnia and objectively measured sleep disruption (using polysomnography or wrist actigraphy) have been shown to predict worse depressive symptom ratings, higher rates of attrition in studies, fluctuations in treatment response, and suicidal ideation. Studies of sleep architecture indicate that major depressive disorder in late life is associated with changes (7), including less time in slow wave sleep and a greater percentage of time in REM sleep compared with nondepressed individuals. Poor sleep can also affect functional ability. In one study, insomnia symptoms were found to be a risk factor for increased disability in tasks that may be related to higher levels of functioning and quality of life. The combination of insomnia and short sleep duration has also been associated with adverse physical health outcomes, including poor neurocognitive functioning, hypertension, diabetes, and mortality (6, 8, 9). Given the association between cardiovascular health and depression, sleep disruption may increase the likelihood of negative physical outcomes associated with depression.
There is also important evidence demonstrating the impact of sleep symptoms on depression treatment response. A study by Troxel and colleagues (6) noted that sleep latency of 30 minutes or greater was associated with a higher risk of nonremission of depression in response to both pharmacologic and nonpharmacologic approaches. Moreover, the presence of three or more sleep disturbances (e.g., insomnia, prolonged sleep latency, short duration of sleep, frequent awakenings) may triple the likelihood of nonremission.
The case of Mr. G highlights how clinicians may be able to incorporate objective sleep measurement using wrist-worn devices into regular clinical care in an efficient, easy-to-use manner. The use of actigraphy has been recommended as a tool for identifying patients who may be at risk for poor depression outcomes because of poor sleep (6). While wrist-worn actigraphy may not be as accurate as polysomnography, several studies now utilize it as the primary objective measure of sleep. Currently available wrist-worn devices offer easily accessible and interpretable data that may simplify the process of regular clinical use of objective sleep measurement. Self-report has been noted to vary considerably from objectively measured sleep. In over 30% of the population, there may be variations of 1 hour or greater. Factors associated with higher variance include male gender, functional impairment, and cognitive dysfunction (10). The clinical impact of this variance may be disproportionately higher among older adults, especially those with cognitive impairment, in comparison with younger, relatively healthy populations.
Psychomotor Retardation and Physical Activity In Depression
Depression in older adults, especially those with cognitive impairment, can present primarily as amotivation and neurovegetative symptoms (11). Psychomotor retardation has been reported as more common among older adults (12–14), and a syndrome of depression accompanied by executive dysfunction and characterized by such psychomotor retardation has been described (15). Apathy, which is a common syndrome in late-life depression, can also include psychomotor retardation as a component (16).
Psychomotor retardation can create a diagnostic challenge for clinicians, since self-report of movement ability has been demonstrated as unreliable, especially if it is compounded by other conditions that impede movement, such as arthritis (17). Availability of objective tools to document and measure motion in real time may substantially improve clinicians’ ability to establish the nature and extent of psychomotor retardation in older adults and can have impact on clinical decision making, as demonstrated in the case of Mr. G. With sensor technology proliferating and becoming ubiquitous, especially in smartphones, preliminary studies have suggested a role for such technology in monitoring mental health status (18). One study demonstrated that motion-related data collected by cellphone sensors (including geospatial and kinesthetic activity) can correlate with changes in depression symptoms. Activity monitoring using wrist-worn sensors has been shown in several studies (19, 20) to be a reliable way of measuring movement changes in depression, and the data collected are comparable to data collected from accelerometers worn on the trunk. Measuring movement of the trunk has historically been considered the preferable way of capturing subtle changes in movement associated with mood (20).
However, implementation of research-based actigraphy in regular clinical practice may not be feasible because of the additional time and effort that may be required to download and interpret the data, thus presenting a significant barrier to adoption. The availability of wrist-worn actigraphs may represent a feasible way of overcoming this barrier (21).
Movement, Exercise, Depression, and Cognition
There is an extensive literature on the impact of physical activity on the brain. Exercise can affect both cognition and depression. Although Mr. G was physically fit, his lack of motion may have played a role in the worsening of his physical health, and it has implications for his cognitive health as well. There have been extensive studies—both in animal models and in humans—documenting the impact of activity, especially aerobic exercise, on the brain (22). Exercise has been demonstrated to have a molecular effect on the brain (especially the hippocampus and the frontal cortex), including in gene expression, neuroregeneration, synaptogenesis, and vascularization, that can improve and preserve cognition as well as enhance mood. This is noteworthy because these regions of the brain are susceptible to the fastest rates of atrophy in late life (1%−2% per year), and cognitive dysfunction may accelerate the rate of degeneration (23).
The evidence for beneficial effects of physical exercise, especially aerobic exercise, on depression is also clear. A recent meta-analysis noted that pooled data from 37 clinical trials indicated a modest beneficial effect of exercise over placebo in reducing depressive symptoms (24). Studies indicated that the efficacy rates for exercise were comparable to pharmacotherapy and psychotherapy, possibly with higher rates of adherence. Wearable motion-monitoring devices may provide clinicians with the ability to monitor adherence to and efficacy of exercise as an adjunct to pharmacotherapy for late-life depression with the added potential for a positive impact on cognition.
As noted above, subsyndromal depression may be three times more prevalent than major depression and may not respond to medications (4), and physical activity has been demonstrated to be efficacious in managing these symptoms (3). For clinicians, these data provide direction on how to discuss physical activity with their patients. Technology can also play a key role in facilitating more physical activity. For example, use of commercially available “exergames” or video games that involve physical activity has been demonstrated to significantly improve subsyndromal depression in older adults (25). Using such technology can help improve intrinsic motivation to engage in physical activity, and wrist-worn monitors can help physicians track adherence.
Using motion-sensing technology with Mr. G allowed for the improvement in his degree of activity to be visualized. This had a dual benefit: it gave him and his clinicians the ability to track improvement in psychomotor retardation as the medication changes took effect, and it provided a rationale for him to increase the length and duration of his daily walks once he felt less depressed. This added a therapeutic factor that may have augmented the antidepressant effect of exercise.
Technology Adoption By Older Adults
The easier technology is to use, and the easier it is to visualize data, the more likely are physicians and patients to adopt it. Older adults are likely to adopt technology only if it performs a function that they cannot otherwise perform and if use of the technology is likely to lead to tangible benefit, while being easy to use (26).
The current generation of commercially available wrist-worn fitness monitors (including recently launched “smart watches”) include built-in sophisticated three-dimensional accelerometers and software that can seamlessly process and present easily visualized data. Moreover, about 25% of adults over age 55 already own a fitness monitor. Using such familiar technology for clinical purposes is likely to increase the likelihood of adherence (27). While much of the effort of the health technology industry has been on developing devices targeting specific health problems and specific demographic segments, our experience with Mr. G suggests that an alternative approach by clinicians may have a greater payoff. Widely available popular technologies may be more acceptable to patients, may be cheaper, and, with clear direction and application, can help clinicians add a dimension to care that was previously not possible.
Summary and Conclusions
Use of accelerometer technology in the case of Mr. G contributed to diagnosis, treatment decision making, and prognostic monitoring. Accelerometer data were a primary tool in establishing that the patient’s symptoms were attributable to depressive disorder. This in turn steered our decision to focus on antidepressant dosage as a treatment priority. In addition, there was a clinical dilemma posed by the discrepancy between his self-report and the collateral information provided by his wife. Mr. G fits the demographic profile most associated with unreliable self-reports on sleep (male, with mild neurocognitive impairment), and because of his polypharmacy, the clinical implications of this discrepancy were amplified. Use of motion-detecting technology facilitated the patient’s acceptance of the fact that eliminating two of his insomnia medications did not result in a significant change to his sleep patterns. This demonstrates how such technology may provide clinicians with data that foster reduction of polypharmacy in older adults. Furthermore, once treatment was initiated, being able to visualize his own improvement served as positive reinforcement.
The case of Mr. G highlights several important points. It demonstrates the feasibility of utilizing motion-sensing technology in a clinical environment with older adults who have comorbid mood and cognitive symptoms. It also demonstrates that the use of such technology can add a dimension to clinical diagnostics that was not previously readily available. While there are limitations to using commercially available wrist-worn devices for monitoring motion and sleep in older adults, including the fact that these devices may not be as accurate as polysomnography (28, 29), and that information on the steps taken still needs validation and context based on observational data, this case report provides a template for how these devices may add a dimension to clinical care as a result of maturation of the technology involved and availability of immediately viewable processed data. With further development, such data may even serve as biomarkers of response to antidepressant treatment and guide personalized care (30). As technology grows in scope and application in psychiatric care, it is important to bear in mind that sensor-based devices and other tools may be most effective when the data they generate serve as an adjunct to rather than a substitute for careful clinical inquiry. Future work should assess clinician attitudes and concerns about using commercially available devices for psychiatric care. Future engineering projects should focus on how more nuanced information on movement and behavior may be captured in a noninvasive manner from persons with psychiatric diagnoses.
1 : Depression and cognition in the elderly. Annu Rev Clin Psychol 2015; 11:331–360Crossref, Medline, Google Scholar
2 : Depression in late life: review and commentary. J Gerontol A Biol Sci Med Sci 2003; 58:249–265Crossref, Medline, Google Scholar
3 : A tune in “a minor” can “b major”: a review of epidemiology, illness course, and public health implications of subthreshold depression in older adults. J Affect Disord 2011; 129:126–142Crossref, Medline, Google Scholar
4 : Subthreshold depression and successful aging in older women. Am J Geriatr Psychiatry 2010; 18:212–220Crossref, Medline, Google Scholar
5 : Association between insomnia symptoms and functional status in US older adults. J Gerontol B Psychol Sci Soc Sci 2014; 69(suppl 1):S35–S41Crossref, Medline, Google Scholar
6 : Insomnia and objectively measured sleep disturbances predict treatment outcome in depressed patients treated with psychotherapy or psychotherapy-pharmacotherapy combinations. J Clin Psychiatry 2012; 73:478–485Crossref, Medline, Google Scholar
7 : Sleep architecture and mental health among community-dwelling older men. J Gerontol B Psychol Sci Soc Sci 2015; 70:673–681Crossref, Medline, Google Scholar
8 : Insomnia with objective short sleep duration is associated with type 2 diabetes: a population-based study. Diabetes Care 2009; 32:1980–1985Crossref, Medline, Google Scholar
9 : Insomnia with objective short sleep duration is associated with deficits in neuropsychological performance: a general population study. Sleep 2010; 33:459–465Crossref, Medline, Google Scholar
10 : Disagreement between subjective and actigraphic measures of sleep duration in a population-based study of elderly persons. J Sleep Res 2008; 17:295–302Crossref, Medline, Google Scholar
11 : Depressive symptoms among cognitively normal versus cognitively impaired elderly subjects. Int J Geriatr Psychiatry 2001; 16:455–461Crossref, Medline, Google Scholar
12 : Psychomotor retardation in elderly untreated depressed patients. Front Psychiatry 2014; 5:196Medline, Google Scholar
13 : Stroop performance in major depression: selective attention impairment or psychomotor slowness? J Affect Disord 2010; 122:167–173Crossref, Medline, Google Scholar
14 : Self-reported lack of energy (anergia) among elders in a multiethnic community. J Gerontol A Biol Sci Med Sci 2008; 63:707–714Crossref, Medline, Google Scholar
15 : Assessment of late life depression. Biol Psychiatry 2002; 52:164–174Crossref, Medline, Google Scholar
16 : Apathy in late-life depression: common, persistent, and disabling. Am J Geriatr Psychiatry 2015; 23:488–494Crossref, Medline, Google Scholar
17 : Do disease specific characteristics add to the explanation of mobility limitations in patients with different chronic diseases? A study in the Netherlands. J Epidemiol Community Health 1997; 51:676–685Crossref, Medline, Google Scholar
18 : Next-generation psychiatric assessment: using smartphone sensors to monitor behavior and mental health. Psychiatr Rehabil J 2015; 38:218–226Crossref, Medline, Google Scholar
19 : Activity monitoring in patients with depression: a systematic review. J Affect Disord 2013; 145:21–28Crossref, Medline, Google Scholar
20 : Improving motor activity assessment in depression: which sensor placement, analytic strategy, and diurnal time frame are most powerful in distinguishing patients from controls and monitoring treatment effects. PLoS One 2015; 10:e0124231Crossref, Medline, Google Scholar
21 : Physicians’ perceptions of mobile technology for enhancing asthma care for youth. J Child Health Care [Epub ahead of print, Nov 26, 2014]Google Scholar
22 : Physical activity and brain plasticity in late adulthood. Dialogues Clin Neurosci 2013; 15:99–108Medline, Google Scholar
23 : Differential brain shrinkage over 6 months shows limited association with cognitive practice. Brain Cogn 2013; 82:171–180Crossref, Medline, Google Scholar
24 : Exercise for depression. Cochrane Database Syst Rev 2013; 9:CD004366Medline, Google Scholar
25 : Exergames for subsyndromal depression in older adults: a pilot study of a novel intervention. Am J Geriatr Psychiatry 2010; 18:221–226Crossref, Medline, Google Scholar
26 : How older adults make decisions regarding smart technology: an ethnographic approach. Assist Technol 2012; 24:168–181Crossref, Medline, Google Scholar
27 NPD Group: The demographic divide: fitness trackers and smartwatches attracting very different segments of the market, according to the NPD Group. Press release, Jan 6, 2015. https://www.npd.com/wps/portal/npd/us/news/press-releases/2015/the-demographic-divide-fitness-trackers-and-smartwatches-attracting-very-different-segments-of-the-market-according-to-the-npd-group/Google Scholar
28 : The role of actigraphy in the study of sleep and circadian rhythms. Sleep 2003; 26:342–392Crossref, Medline, Google Scholar
29 : Examination of wrist and hip actigraphy using a novel sleep estimation procedure. Sleep Sci 2014; 7:74–81Crossref, Medline, Google Scholar
30 : Personalizing the care of geriatric depression. Am J Psychiatry 2008; 165:790–792Link, Google Scholar



