Neurofunctional Domains Derived From Deep Behavioral Phenotyping in Alcohol Use Disorder
Abstract
Objective:
The authors evaluated whether three neurofunctional domains proposed to be critical in the addiction cycle, namely, incentive salience, negative emotionality, and executive function, could be identified through factor analysis of a deeply phenotyped clinical sample.
Methods:
Clinical, behavioral, and self-report measures of addiction, personality, cognition, behavior, and exposure to early-life stress were collected as part of a screening and natural history study of alcohol use disorders in 454 individuals representing the spectrum of alcohol use and use disorders. The multiple indicators, multiple causes (MIMIC) approach was used to identify significant predictors of the latent factors identified by the analysis.
Results:
The results showed significant support for both three- and four-factor models to explain biobehavioral variation in this sample of participants with alcohol use disorder and control subjects, but the three-factor model had the best fit indices. With some nuances, including cross-correlation (lack of independence) between the three factors, the factors corresponded to incentive salience, negative emotionality, and executive function (executive control). The MIMIC model revealed that both exposure to early-life stress and sociodemographic variables predicted these factors.
Conclusions:
These findings suggest that three correlated neurofunctional domains are relevant for alcohol use disorder. More work is required to validate and standardize measures of neurofunctional domains in alcohol use disorder, to extend these findings to other addictive disorders, and to relate variations in them to predisposition, clinical course, treatment response, neuroimaging data, and other psychophysical indicators.
Addiction to diverse substances, including alcohol, tobacco, and illicit drugs, is a leading preventable contributor to global disease burden (1). In 2016, substance use and use disorders accounted for two-thirds of the estimated 64,026 drug overdose fatalities in the United States (2), and 88,000 fatalities were associated with alcohol-related adverse effects (3). Although several pharmacological and behavioral treatments for substance use disorders have shown efficacy in controlled clinical trials, there is a need for more effective treatments. Better knowledge, and better measures, of the functional domains underlying substance use disorders could help in assessing severity, modeling heterogeneity, predicting course, targeting treatments, and detecting efficacy and treatment mechanisms.
Building on conceptual frameworks derived from neurobiology, clinical studies, and social psychology (4, 5), we recently proposed a neuroscience-based framework (the Addictions Neuroclinical Assessment [ANA]) to better understand the heterogeneity of addiction (6). This framework postulates that three domains are implicated in the development and maintenance of substance use disorders: incentive salience, negative emotionality, and executive function (7). As reviewed in detail elsewhere (6), previous studies have shown group differences between addicted and nonaddicted individuals in various assessments, including neuropsychological and neuroimaging, purporting to measure those domains. Additional disruptions in function in those domains have been demonstrated in some individuals at risk for developing addictive disorders, suggesting that compromised function may be a risk factor for, or a consequence of, addiction. One constraint of these previous studies is that they typically evaluated a single domain of function, such as cognitive control or emotion regulation, rather than using a comprehensive framework. An exception to this pattern is the National Consortium on Alcohol and Neurodevelopment in Adolescence study, which evaluated multiple functional domains in adolescents across the spectrum of alcohol use (37). That study found differences in cognitive, affective, and motor domains related to levels of alcohol consumption. These initial findings suggest that further testing of a comprehensive model in clinical samples is warranted. The present study attempts to begin such a line of inquiry.
We sought to test the ANA model by applying factor-analytic techniques to a deeply phenotyped sample across the spectrum of alcohol use and alcohol use disorders, ranging from healthy individuals to patients with DSM-IV alcohol abuse or dependence or DSM-5 alcohol use disorder. We hypothesized, on the basis of developmental models of substance use disorders (8–10), that family history, adverse childhood experiences, and age at first drink would predict the scores of individuals on factors emergent from this analysis.
Methods
Participants
Participants (N=454) included individuals seeking treatment for alcohol use disorder at the National Institutes of Health (NIH) Clinical Center and non-treatment-seeking individuals (ranging from healthy volunteers to individuals with alcohol use disorder not seeking treatment) screened in the National Institute on Alcohol Abuse and Alcoholism (NIAAA) outpatient clinic for participation in research studies between January 2015 and February 2017. All participants completed the NIAAA Screening and Natural History Protocol, which provides a platform for common assessments and uses minimal eligibility criteria, to maximize generalizability. Individuals were excluded only if they were pregnant, breastfeeding, or under legal confinement. Treatment-seeking patients were diagnosed with alcohol dependence or abuse using DSM-IV terminology; we refer to their diagnosis as alcohol use disorder, using the DSM-5 term, to include those with dependence and abuse in the same group, as is consistent with the current nosology. Most patients had multiple relapses despite prior treatment. All study participants provided written informed consent under an NIH institutional review board–approved protocol.
Assessments and Indicator Variables
Study participants underwent a deep phenotyping with measures relevant for addiction and related constructs, including psychiatric diagnoses, attention, impulsivity, aggression, and personality. Specific instruments are listed in the footnotes of Table 1.
| Measure | Total Sample (N=454) | Participants With Alcohol Use Disordera (N=186) | Participants Without Alcohol Use Disorder (N=252) | p | |||
|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | ||
| Female | 185 | 40.8 | 53 | 28.5 | 124 | 49.2 | <0.001 |
| Caucasian | 185 | 40.8 | 74 | 39.8 | 107 | 42.5 | 0.02 |
| Hispanic | 26 | 5.7 | 4 | 2.2 | 21 | 8.3 | 0.006 |
| Current alcohol use disorder | 186 | 43.3 | 186 | 100.0 | 0 | 0.0 | |
| Lifetime alcohol use disorder | 200 | 46.5 | 186 | 100.0 | 14 | 5.6 | <0.001 |
| Mean | SD | Mean | SD | Mean | SD | ||
| Age (years) | 40.3 | 13.5 | 45.2 | 11.9 | 36.4 | 13.4 | <0.001 |
| Years of education | 14.7 | 3.6 | 13.7 | 3.2 | 15.6 | 3.6 | <0.001 |
| Family history of alcohol use problemsb | 0.1 | 0.3 | 0.2 | 0.3 | 0.1 | 0.2 | <0.001 |
| Age at first drink (years) | 16.5 | 4.3 | 15.2 | 5.3 | 17.6 | 3.1 | <0.001 |
| Alcohol dependence severityc | 8.5 | 10.2 | 17.6 | 9.1 | 1.3 | 2.3 | <0.001 |
| Obsessive drinking subscaled | 3.2 | 4.7 | 7.0 | 5.1 | 0.3 | 1.1 | <0.001 |
| Compulsive drinking subscaled | 5.6 | 5.6 | 10.6 | 4.8 | 1.8 | 2.3 | <0.001 |
| N | % | N | % | N | % | ||
| Current substance use disorder (other than alcohol) | 28 | 6.5 | 28 | 15.1 | 0 | 0.0 | <0.001 |
| Lifetime substance use disorder (other than alcohol) | 129 | 30.0 | 111 | 59.7 | 18 | 7.1 | <0.001 |
| Current mood disorder | 32 | 7.4 | 29 | 15.6 | 3 | 1.2 | <0.001 |
| Lifetime mood disorder | 80 | 18.6 | 51 | 27.4 | 29 | 11.5 | <0.001 |
| Current anxiety disorder | 55 | 12.8 | 43 | 23.1 | 12 | 4.8 | <0.001 |
| Lifetime anxiety disorder | 68 | 15.8 | 56 | 30.1 | 12 | 4.8 | <0.001 |
| Current PTSD | 24 | 5.6 | 19 | 10.2 | 5 | 2.0 | <0.001 |
| Lifetime PTSD | 34 | 7.9 | 29 | 15.6 | 5 | 2.0 | <0.001 |
| Mean | SD | Mean | SD | Mean | SD | ||
| Emotional abusee | 8.3 | 4.6 | 9.4 | 5.2 | 7.3 | 3.8 | <0.001 |
| Physical abusee | 7.3 | 3.5 | 8.3 | 4.4 | 6.5 | 2.3 | <0.001 |
| Sexual abusee | 6.2 | 3.9 | 6.7 | 4.4 | 5.7 | 2.9 | 0.007 |
| Emotional neglecte | 9.5 | 4.5 | 10.2 | 4.9 | 8.7 | 4.0 | 0.001 |
| Physical neglecte | 6.9 | 3.1 | 7.5 | 3.7 | 6.4 | 2.3 | <0.001 |
| Indicator variables | |||||||
| Continuous measures | |||||||
| Attentional impulsivityf | 14.1 | 4.2 | 16.3 | 4.6 | 12.3 | 3.0 | <0.001 |
| Motor impulsivityf | 22.3 | 4.4 | 24.5 | 5.0 | 20.6 | 3.2 | <0.001 |
| Nonplanning impulsivityf | 22.6 | 6.2 | 26.3 | 6.1 | 19.6 | 4.4 | <0.001 |
| Negative urgencyg | 2.0 | 0.7 | 2.4 | 0.7 | 1.6 | 0.5 | <0.001 |
| Premeditationg | 1.8 | 0.5 | 2.0 | 0.6 | 1.7 | 0.4 | <0.001 |
| Perseveranceg | 1.8 | 0.5 | 2.0 | 0.5 | 1.6 | 0.4 | <0.001 |
| Positive urgencyg | 1.7 | 0.7 | 2.1 | 0.8 | 1.4 | 0.5 | <0.001 |
| Neuroticismh | 49.0 | 11.6 | 54.9 | 11.4 | 45.0 | 10.0 | <0.001 |
| Extraversionh | 51.0 | 10.1 | 52.0 | 10.2 | 50.3 | 10.0 | 0.11 |
| Opennessh | 54.1 | 10.5 | 51.7 | 10.0 | 55.8 | 10.4 | <0.001 |
| Agreeablenessh | 48.7 | 11.0 | 46.6 | 11.6 | 50.1 | 10.3 | 0.002 |
| Conscientiousnessh | 50.2 | 11.0 | 45.3 | 11.4 | 53.7 | 9.2 | <0.001 |
| Delay discounting k function | 0.1 | 0.2 | 0.1 | 0.2 | 0.1 | 0.2 | 0.23 |
| Aggressioni | 84.6 | 18.8 | 92.4 | 19.5 | 77.9 | 14.8 | <0.001 |
| MADRS depression scorej | 5.9 | 8.8 | 12.1 | 10.1 | 1.2 | 3.0 | <0.001 |
| Spielberger trait anxietyk | 35.6 | 12.8 | 44.6 | 13.0 | 28.6 | 7.5 | <0.001 |
| N | % | N | % | N | % | ||
| Categorical measures | |||||||
| Symptoms consistent with ADHDl | |||||||
| 0 | 403 | 89.2 | 143 | 77.7 | 245 | 97.2 | <0.001 |
| 1 | 49 | 10.8 | 41 | 22.3 | 7 | 2.8 | |
| ADS item 18: Do you almost constantly think about drinking alcohol?m | |||||||
| 0 | 351 | 77.7 | 90 | 48.9 | 251 | 99.6 | <0.001 |
| 1 | 101 | 22.3 | 94 | 51.1 | 1 | 0.4 | |
| N | % | N | % | N | % | ||
| OCDS item 1: How much of your time when you’re not drinking is occupied by ideas, thoughts, impulses, or images related to drinking?n | |||||||
| 0 | 238 | 52.5 | 23 | 12.4 | 211 | 83.7 | <0.001 |
| 1 | 98 | 21.6 | 55 | 29.7 | 37 | 14.7 | |
| 2 | 58 | 12.8 | 53 | 28.7 | 3 | 1.2 | |
| 3 | 28 | 6.2 | 27 | 14.6 | 0 | 0.0 | |
| 4 | 31 | 6.8 | 27 | 14.6 | 1 | 0.4 | |
| OCDS item 11: If you were prevented from drinking alcohol when you desired a drink, how anxious or upset would you become?o | |||||||
| 0 | 263 | 58.1 | 29 | 15.7 | 232 | 92.1 | <0.001 |
| 1 | 77 | 17.0 | 53 | 28.7 | 18 | 7.1 | |
| 2 | 54 | 11.9 | 48 | 26.0 | 2 | 0.8 | |
| 3 | 44 | 9.7 | 41 | 22.2 | 0 | 0.0 | |
| 4 | 15 | 3.3 | 14 | 7.6 | 0 | 0.0 | |
| OCDS item 13: How strong is the drive to consume alcoholic beverages?p | |||||||
| 0 | 238 | 52.5 | 23 | 12.4 | 210 | 83.3 | <0.001 |
| 1 | 89 | 19.7 | 50 | 27.0 | 37 | 14.7 | |
| 2 | 42 | 9.3 | 32 | 17.3 | 3 | 1.2 | |
| 3 | 55 | 12.1 | 53 | 28.7 | 2 | 0.8 | |
| 4 | 29 | 6.4 | 27 | 14.6 | 0 | 0.0 | |
TABLE 1. Demographic and clinical characteristics and indicator measures used in the analysis in a study of alcohol use disorder
Statistical Analyses
Exploratory and confirmatory factor analysis.
The data set was randomly split into two halves (each with N=227), one for discovery and the other for replication. We used exploratory factor analysis in the discovery half of the sample to identify latent factors underlying the indicator variables included in the assessments. Analyses were conducted in Mplus, version 7.4 (http://www.statmodel.com/). A robust weighted least squares estimator, which does not assume normally distributed variables (11), was used along with the geomin oblique rotation. The geomin rotation allows correlation between factors and is recommended when indicators are predicted to load onto more than one factor (12). The exploratory factor analysis models were estimated using full information maximum likelihood. Factor selection was guided by examination of fit indices and overall interpretability. The fit indices examined were the root mean square error of approximation (RMSEA), the comparative fit index (CFI), and the Tucker-Lewis index (TLI). We followed the recommendations of Hu et al. (13), who suggest CFI and TLI values above 0.95 and RMSEA values below 0.06 to represent good model fit. Variables with a loading ≥0.35 were considered to load onto a particular factor. Confirmatory factor analysis was performed in the replication half of the data set. Variables with loadings ≤0.35 in the exploratory factor analysis were fixed at 0, and modification indices were examined and applied if they improved model fit and were conceptually meaningful.
MIMIC analysis.
To assess the influence of several predictor variables on the latent factors and individual indicators, we conducted a multiple indicator, multiple cause (MIMIC) analysis (14) using the final solution of the confirmatory factor analysis, with the resulting factor scores as the outcome variables. For this analysis, we recombined the data to include the full sample. Specifically, we evaluated the association of both the latent factors and the indicators with the following: three demographic variables (sex, age, and race/ethnicity), current alcohol use disorder diagnosis, family history of alcohol problems, age at first drink, and exposure to childhood adversity as assessed by the five subscales of the Childhood Trauma Questionnaire. Direct effects (i.e., effects that were not mediated by the latent factors) of the predictors on indicator measures were identified using modification indices with a cutoff of 10 and a p threshold of 0.001 to identify predictors with significant direct effects (15).
Receiver operating characteristic curve analysis.
To assess the ability of each factor/domain to predict alcohol use disorder (determined via Structured Clinical Interview for DSM-IV Disorders [SCID-IV] interview), we plotted receiver operating characteristic (ROC) curves using the factor scores from each of the three factors and calculated the area under the curve as a measure of how well the scores distinguished between individuals with and without alcohol use disorder. This analysis was performed in SAS, version 9.4 (SAS Institute, Cary, N.C.).
Results
Sample
The sample included a total of 454 individuals. Approximately 40% of the sample were female, and 40% were Caucasian, with most of the rest being African-American. The mean age was just over 40 years, and the mean educational attainment was 14 years. A substantial percentage (43.5%) of the participants had current alcohol use disorder. Participants had high rates of other lifetime psychiatric disorders, including substance use disorders (30%), mood disorders (18.6%), anxiety disorders (15.8%), and posttraumatic stress disorder (7.9%). The participants’ demographic and clinical characteristics are summarized in Table 1.
Factor Structure
In the exploratory factor analysis in the discovery data set, good model fit was found for both a three-factor model (RMSEA=0.04, CFI=0.97, TLI=0.96) and a four-factor model (RMSEA=0.04, CFI=0.98, TLI=0.97). However, the four-factor model, while yielding roughly the same first three factors as the three-factor solution, included a fourth factor with only a single indicator (extraversion). Hence, we chose the three-factor model as the model for subsequent analyses because of its greater parsimony and better correspondence with the three hypothesized ANA domains. Factor loadings for the three-factor solution appear in Table 2. Overall, the factors aligned well with the hypothesized ANA domains, with a few nuances. Factor 1 defined a negative emotionality domain, with positive loadings for neuroticism, aggression, and trait anxiety and negative loadings for extraversion and agreeableness; however, positive urgency also loaded onto factor 1. Factor 2 defined an (impaired) executive function (executive control) domain, with positive loadings for ADHD, the three impulsivity measures from the Barratt Impulsiveness Scale, and the four impulsivity measures from the UPPS-P Impulsive Behavior Scale and negative loading for conscientiousness. Factor 3 defined an incentive salience (and mood) domain, with positive loadings for the three items from the Obsessive-Compulsive Drinking Scale and the Alcohol Dependence Scale that assess thinking about drinking and drive to consume alcohol. The Montgomery-Åsberg Depression Rating Scale depression score and the Obsessive-Compulsive Drinking Scale item assessing anxiety when not able to drink also loaded onto factor 3. Two variables, delay discounting and openness, did not load onto any factor. In addition to Table 2, a correlation matrix of continuous indicators appears in the online supplement in Table S1, and the four-factor solution appears in Table S2.
| Proposed Grouping and Indicator or Measure | Factor 1 | Factor 2 | Factor 3 |
|---|---|---|---|
| Executive function | |||
| ADHD | 0.166 | 0.443 | 0.193 |
| Attentional impulsivity | 0.259 | 0.675 | –0.024 |
| Motor impulsivity | 0.068 | 0.662 | –0.042 |
| Nonplanning impulsivity | 0.093 | 0.730 | 0.074 |
| Delay discounting (k) | 0.174 | –0.184 | 0.027 |
| Conscientiousness | 0.036 | –0.723 | –0.014 |
| Negative urgency | 0.329 | 0.536 | 0.143 |
| Premeditation | –0.008 | 1.022 | –0.310 |
| Perseverance | 0.157 | 0.640 | –0.002 |
| Positive urgency | 0.374 | 0.504 | 0.042 |
| Negative emotionality | |||
| Depression | 0.151 | –0.081 | 0.689 |
| Neuroticism | 0.534 | 0.007 | 0.294 |
| Extraversion | –0.394 | 0.307 | 0.008 |
| Openness | –0.039 | 0.009 | –0.117 |
| Agreeableness | –0.598 | –0.042 | 0.174 |
| Trait anxiety | 0.350 | 0.112 | 0.524 |
| Aggression | 0.931 | –0.053 | –0.001 |
| OCDS item 11 | 0.005 | –0.062 | 0.976 |
| Incentive salience | |||
| OCDS item 1 | –0.123 | 0.032 | 0.903 |
| OCDS item 13 | 0.003 | –0.191 | 0.997 |
| ADS item 18 | –0.119 | 0.017 | 0.986 |
TABLE 2. Three-factor solution from exploratory factor analysis in a study of alcohol use disordera
Confirmatory factor analysis of the three-factor model in the replication half of the data set also resulted in good model fit (RMSEA=0.06, CFI=0.94, TLI=0.93). We subsequently ran a confirmatory factor analysis in the full recombined data set, which also fit well (RMSEA=0.05, CFI=0.95, TLI=0.94). All three factors were correlated with each other (factor 1/factor 2 correlation coefficient=0.90, factor 1/factor 3 correlation coefficient=0.76, factor 2/factor 3 correlation coefficient=0.76). A visual representation of the model is presented in Figure 1.
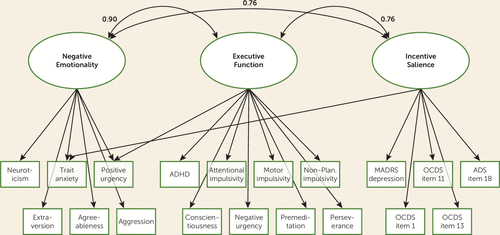
FIGURE 1. Visual representation of the three latent factors and the specific indicators in a study of alcohol use disordera
a The numbers indicate the correlations between the three factors. ADHD=attention deficit hyperactivity disorder; ADS=Alcohol Dependence Scale; MADRS=Montgomery-Åsberg Depression Rating Scale; OCDS=Obsessive-Compulsive Drinking Scale.
MIMIC Analysis
The results of the MIMIC analysis are presented in Table 3 and Figure 2. Predictors of higher scores for negative emotionality included alcohol use disorder, being male, emotional abuse, sexual abuse, and the emotional neglect subscales of the Childhood Trauma Questionnaire. Predictors of higher scores for executive function included alcohol use disorder, emotional and sexual abuse, family history, and age at first drink. Non-Caucasian race was associated with a lower score (indicating better executive function), as was higher age at first drink. Conversely, emotional abuse, sexual abuse, alcohol use disorder, and family history were associated with a higher score (poorer executive function). Higher scores in the incentive salience factor included alcohol use disorder, emotional abuse, and family history (although the latter association was marginally significant). Non-Caucasian race was associated with lower scores. There were only four direct effects of predictors on indicator measures: sex on agreeableness and neuroticism, age on agreeableness, and alcohol use disorder on extraversion.
| Predictor | Estimate | SE | p | Estimate | SE | p | Estimate | SE | p |
|---|---|---|---|---|---|---|---|---|---|
| Effects of predictors on latent factors | |||||||||
| Negative emotionality | Executive function | Incentive salience | |||||||
| Alcohol use disorderb | 0.641 | 0.105 | <0.001 | 0.923 | 0.083 | <0.001 | 1.507 | 0.073 | <0.001 |
| Sexc | 0.329 | 0.104 | 0.001 | 0.093 | 0.089 | 0.295 | –0.069 | 0.079 | 0.381 |
| Raced | –0.056 | 0.097 | 0.564 | –0.377 | 0.085 | <0.001 | –0.332 | 0.074 | <0.001 |
| Age | –0.027 | 0.048 | 0.574 | –0.055 | 0.045 | 0.22 | 0.002 | 0.042 | 0.966 |
| Emotional abuse | 0.202 | 0.068 | 0.003 | 0.133 | 0.06 | 0.025 | 0.187 | 0.054 | 0.001 |
| Physical abuse | 0.07 | 0.065 | 0.281 | 0.006 | 0.058 | 0.925 | –0.067 | 0.054 | 0.219 |
| Sexual abuse | 0.107 | 0.047 | 0.023 | 0.116 | 0.045 | 0.01 | 0.007 | 0.044 | 0.881 |
| Emotional neglect | 0.19 | 0.072 | 0.008 | 0.014 | 0.063 | 0.821 | –0.031 | 0.053 | 0.56 |
| Physical neglect | –0.072 | 0.068 | 0.293 | 0.066 | 0.062 | 0.28 | 0.067 | 0.048 | 0.159 |
| Family history | 0.043 | 0.049 | 0.382 | 0.088 | 0.044 | 0.044 | 0.07 | 0.035 | 0.049 |
| Age at first drink | –0.075 | 0.053 | 0.161 | –0.109 | 0.044 | 0.014 | –0.055 | 0.035 | 0.114 |
| Significant direct effects of predictors on indicators | |||||||||
| Extraversion | Neuroticism | Agreeableness | |||||||
| Alcohol use disorderb | 0.447 | 0.134 | 0.001 | – | – | – | – | – | – |
| Sexc | – | – | – | –0.401 | 0.092 | <0.001 | –0.337 | 0.105 | 0.001 |
| Age | – | – | – | – | – | – | 0.212 | 0.054 | <0.001 |
TABLE 3. MIMIC model results in a study of alcohol use disordera
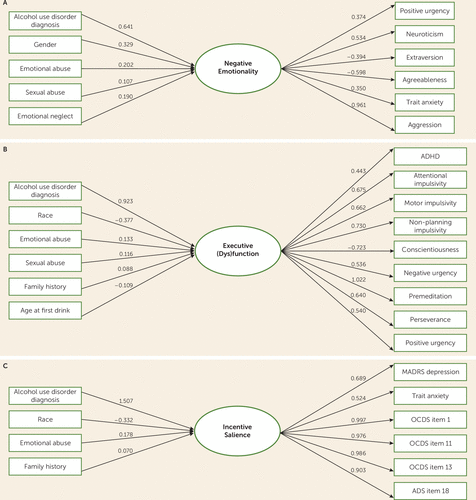
FIGURE 2. Predictors and indicators of the three latent factors, including variable loadings, in a study of alcohol use disordera
a Only significant predictors are included. ADHD=attention deficit hyperactivity disorder; ADS=Alcohol Dependence Scale; MADRS=Montgomery-Åsberg Depression Rating Scale; OCDS=Obsessive-Compulsive Drinking Scale.
ROC Analysis
All three factors distinguished well between individuals with and without alcohol use disorder, with incentive salience having the highest area under the curve (AUC) at 0.96. The AUCs for negative emotionality and executive function were 0.86 and 0.85, respectively. The ROC curves based on the scores output for each factor appear in Figure S2 in the online supplement.
Discussion
In a large and diverse clinical sample enriched for alcohol use disorder, factors representing three neurofunctional domains, incentive salience, negative emotionality, and executive function, underpinned and incorporated results from a broad range of scales and neuropsychological tests. Our findings support the existence of neurofunctional domains relevant to substance use disorders that have been previously theorized (6, 16) and for which previous evidence in model organisms and humans has focused attention on particular aspects of cognition, behavior, emotion, and circuit function (e.g., 7, 17, 18).
Factor analyses of clinical data from substance use disorders are limited by the lack of measures that may directly access the underlying domains, thereby increasing the likelihood, if not ensuring, that such studies return class structures based on severity, onset, exposure, and use. Several clinical trials, including Project MATCH for the treatment of alcohol dependence, have targeted treatments to clinical subgroups or patients with particular clinical features. For example, long-term, reward-blunted patients with alcohol dependence may be less responsive to naltrexone, explaining the heterogeneous efficacy of this drug in clinical trials (19, 20).
We hypothesized that noninvasive deep phenotyping of individuals with and without alcohol use disorder across multiple cognitive, psychological, and behavioral domains might detect latent domains mediating vulnerability and progression of addiction. We asked whether the latent factors discovered from this analysis would align with three neurofunctional domains identified in studies of patients with substance use disorders that have used assessments such as positron emission tomography and functional MRI in humans and neuropharmacological, neurocircuit, and genetic manipulations in animal models. Notably, these methods are more invasive and costly and are not suitable for clinical practice.
We found that a three-factor model generally demonstrated a good fit with our assessment measures, providing strong support for our first hypothesis. Furthermore, the factors aligned closely with the ANA domains of incentive salience, negative emotionality, and executive function. Relatively inexpensive and noninvasive measures recovered a framework predicted by a large body of preclinical data, clinical psychology, and brain imaging studies (4, 5). Preclinical data demonstrate decreases in dopaminergic and serotonergic transmission in the nucleus accumbens during withdrawal, paralleled by up-regulation of brain stress systems (e.g., corticotropin-releasing factor, dynorphin) as addiction progresses (4, 21). This increase in stress during withdrawal is also related to increased vulnerability to relapse and failure to regulate negative affective states in humans. Clinical studies have found that substance use disorders, whether because of innate predisposition or the effect of exposure to various environmental stimuli, such as stress or alcohol, is marked by disruptions of function in prefrontal brain regions implicated in executive function (22–24), in striatocortical reward regions implicated in incentive salience (25), and in the extended amygdala, as implicated in negative emotionality (26, 27). Neurobehavioral and cognitive assessment could represent a bridge between clinical assessment of symptoms, use, and course and deeper levels of neurofunctional assessment, recovering indices of the same process.
The three principal factors in our sample were intercorrelated, consistent with models of psychopathology based on clinical rather than neuroscience-based assessments (28). These intercorrelations are likely explained by shared underlying neural circuitry, within and between-system adaptations occurring as part of the addiction process (29, 30), and shared genetic and environmental risk factors. Increasingly, different substance use disorders demonstrate common disruptions and vulnerability processes. An important goal of deep phenotyping is to define how far sharing of vulnerability and consequences extends. Sharing of vulnerability is observed via family studies and several large, epidemiologically sound twin studies demonstrating cross-inheritance of addictions, as well as agent-specific genetic factors (31). Clinical research in general population samples has found that remission of one disorder is associated with remission of other disorders and lower probability of new onset of another disorder (32, 33). Clinical research has also found common outcomes and, for example, higher levels of dysphoria and anxiety in substance use disorder populations, regardless of other subtypology. For example, in one study of patients with substance use disorders from different populations, dysphoria and anxiety were elevated regardless of whether or not patients had antisocial personality disorder features (34). An important future direction is to examine whether change in any one of the ANA domains is accompanied by change in other domains, in accordance with the relatively strong correlation between domains observed in our sample. If that is the case, treatments that target one domain may have positive spillover effects on other domains. Conversely, it may be essential to simultaneously address several deficits to avoid the possibility that unaddressed deficits may interfere with remission or predispose to relapse.
In the ROC analyses, each of the three neurofunctional domains was moderately to highly accurate in predicting alcohol use disorder, with areas under the curve ranging from 0.84 to 0.96. An important caveat is that a variety of other clinical instruments of different lengths and complexity, but of much greater brevity than the SCID, can also be used to diagnose alcohol use disorder. In one study, the four-item CAGE Questionnaire had an AUC of 0.81 for alcohol dependence in men and 0.75 in women (35). Our ROC analysis shows that the three neurofunctional domains are germane to alcohol use disorder, but it does not demonstrate that measuring these latent factors can improve clinical assessment of alcohol use disorder or substance use disorders. Nevertheless, the demonstration that the three proposed domains differentiate between participants with and without alcohol use disorder underscores the relevance of these domains for addiction and is a critical finding of this data analysis.
The replication of the three-factor structure in the two split samples provides further support for the robustness of the three-factor solution. Because the ANA model is based on a combination of preclinical and clinical data and had not previously been tested in its entirety, these findings suggest that the development of a battery of assessments designed specifically to measure the ANA domains among individuals with a broader range of substance use disorders is a critical next step for evaluating the validity and clinical utility of this model.
We hypothesized that sociodemographic characteristics would predict severity of the neurofunctional factors. Our MIMIC model indicated that the three domains were associated with genetic (e.g., sex, family history), environmental (childhood adversity), and developmental (age at first drink) correlates. These findings, consistent with biopsychosocial models of substance use disorders (8–10), underscore the widespread effects of these risk factors on the neurobiological underpinnings of alcohol use disorder and offer potential targets for prevention. Gene-by-environment interactions may play a role in explaining these findings. For example, individuals with impaired executive function due to genetic predisposition may also be early users of alcohol, which itself is an environmental variable shaping alcohol use disorder onset and progression (36, 37). In this scenario, an individual may have both a genetic predisposition for impaired executive function and exposure early in life to alcohol as a result of a family environment that may also be influenced by the same genetic liability.
For the negative emotionality domain, it is critical to consider the role of exposure to adverse childhood experiences. Exposure to adverse childhood experiences is one of the strongest risk factors for later development of addictive disorders, likely through genetic and environmental mechanisms (38). Childhood trauma has an enduring effect on propensity for negative affect (39) and may lead individuals to consume alcohol and other substances to relieve dysphoria. The MIMIC analysis found that all five subscales of the Childhood Trauma Questionnaire predicted the negative emotionality factor, whereas fewer subscales predicted the other two factors. The finding that Childhood Trauma Questionnaire subscales predicted all three factors constitutes an example of a single predictor leading to disruptions in multiple domains of function, i.e., multifinality (40).
Finally, although our sample included healthy volunteers as well as individuals with other substance use disorders, we note that differences in factor scores may be secondary to alcohol use disorder—that is, occurring as a consequence of the disease—while others may be preexisting. Many individuals become dysphoric as a function of the development of alcohol use disorder or the use of other substances—the “dark side of addiction” described by Koob and colleagues (41, 42)—whereas in others, preexisting dysphoria makes them vulnerable to alcohol use and alcohol use disorder (43). Individuals with alcohol use disorder, regardless of subtype, appear to be more anxious and dysphoric (44), and in the present study we found strong correlations between the negative emotionality factor and other variables. Increased sensitivity to incentive salience is another correlate of alcohol use disorder. It is also possible that some individuals are predisposed to be less motivated for natural rewards and thus more likely to pursue the exaggerated and immediate reward response to cues and context associated with drugs of abuse. Here, incentive salience is defined as motivation for rewards derived from both one’s physiological state and previously learned associations about a reward cue and is often linked to activation of the mesocorticolimbic dopamine system (45). Indeed, the fact that depression and trait anxiety scores loaded more heavily onto the incentive salience factor than the (predicted) negative emotionality factor may be considered evidence of a coupling between negative affect and craving for alcohol as addiction becomes severe (e.g., 7). In this model, craving becomes associated with negative emotions, that is, shifting from “reward” craving to “relief” craving. Finally, the impact of chronic heavy alcohol consumption on executive function may also be profound, while preexisting executive dysfunction is also a known correlate of alcohol use disorder (46). Thus, across all three domains, chronic alcohol consumption leading to alcohol use disorder has the potential to induce negative emotional states, exaggerated salience for alcohol and related cues, and executive dysfunction.
These results must be interpreted in the context of the study’s strengths and limitations. Strengths include a well-characterized sample of individuals across the spectrum of alcohol use, including individuals seeking inpatient treatment for alcohol use disorder, the use of measures with good psychometric properties, and the fact that the factor structure was replicated across both halves of the randomly split sample. Limitations include that measures were not collected prospectively and may be biased by recollection. Furthermore, although study measures allowed us to recover the three factors hypothesized by the ANA, it is possible that additional factors would emerge if other, more diverse measures were included. We note that measures hypothesized to represent the ANA executive function domain comprise a subset of measures related to cognitive control rather than the broad capacities included in executive function (e.g., working memory, attention, response inhibition, planning), and delay discounting did not load onto this factor.
In summary, the neuroclinical assessment of addictions can capture important dimensions of neuropsychological functioning in individuals with varying levels of alcohol use and use disorders, and these domains are highly relevant to substance use disorders. Future studies combining brain imaging and standardized measures for ANA will help refine our understanding of the relationship of these measures to neural circuits implicated in executive function, negative emotionality, and incentive salience in substance use disorders and other addictive disorders. Further studies will be needed to learn whether assessment of ANA domains can improve prevention or treatment, as already hinted at by treatment studies of substance use disorders.
1 : Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009; 373:2223–2233Crossref, Medline, Google Scholar
2 : Provisional Drug Overdose Death Counts. Hyattsville, Md, National Center for Health Statistics, 2018Google Scholar
3 Centers for Disease Control and Prevention: Alcohol and public health: Alcohol-related disease impact (https://nccd.cdc.gov/DPH_ARDI/Default/Report.aspx?T=AAM&P=f6d7eda7-036e-4553-9968-9b17ffad620e&R=d7a9b303-48e9-4440-bf47-070a4827e1fd&M=8E1C5233-5640-4EE8-9247-1ECA7DA325B9&F=&D=)Google Scholar
4 : Drug abuse: hedonic homeostatic dysregulation. Science 1997; 278:52–58Crossref, Medline, Google Scholar
5 : Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry 2002; 159:1642–1652Link, Google Scholar
6 : Addictions Neuroclinical Assessment: a neuroscience-based framework for addictive disorders. Biol Psychiatry 2016; 80:179–189Crossref, Medline, Google Scholar
7 : Neurocircuitry of addiction. Neuropsychopharmacology 2010; 35:217–238Crossref, Medline, Google Scholar
8 : Towards a comprehensive developmental model of cannabis use disorders. Addiction 2014; 109:284–294Crossref, Medline, Google Scholar
9 : Toward a comprehensive developmental model of smoking initiation and nicotine dependence. Drug Alcohol Depend 2014; 144:160–169Crossref, Medline, Google Scholar
10 : Towards a comprehensive developmental model of pathological gambling. Addiction 2015; 110:1340–1351Crossref, Medline, Google Scholar
11 : Mplus User’s Guide, 7th ed. Los Angeles, Muthén & Muthén, 2015Google Scholar
12 : An overview of analytic rotation in exploratory factor analysis. Multivariate Behav Res 2001; 36:111–150Crossref, Google Scholar
13 : Can test statistics in covariance structure analysis be trusted? Psychol Bull 1992; 112:351–362Crossref, Medline, Google Scholar
14 : Estimation of a model with multiple indicators and multiple causes of a single latent variable. J Am Stat Assoc 1975; 70:631–639Google Scholar
15 : Detecting differential item functioning with confirmatory factor analysis and item response theory: toward a unified strategy. J Appl Psychol 2006; 91:1292–1306Crossref, Medline, Google Scholar
16 : Heterogeneity of alcohol use disorder: understanding mechanisms to advance personalized treatment. Alcohol Clin Exp Res 2015; 39:579–584Crossref, Medline, Google Scholar
17 : Neuropsychological consequences of alcohol and drug abuse on different components of executive functions. J Psychopharmacol 2010; 24:1317–1332Crossref, Medline, Google Scholar
18 : Effects of adrenal sensitivity, stress- and cue-induced craving, and anxiety on subsequent alcohol relapse and treatment outcomes. Arch Gen Psychiatry 2011; 68:942–952Crossref, Medline, Google Scholar
19 : Predicting naltrexone response in alcohol-dependent patients: the contribution of functional magnetic resonance imaging. Alcohol Clin Exp Res 2014; 38:2754–2762Crossref, Medline, Google Scholar
20 : Precision medicine in alcohol dependence: a controlled trial testing pharmacotherapy response among reward and relief drinking phenotypes. Neuropsychopharmacology 2018; 43:891–899Crossref, Medline, Google Scholar
21 : A role for brain stress systems in addiction. Neuron 2008; 59:11–34Crossref, Medline, Google Scholar
22 : Disrupted control network connectivity in abstinent patients with alcohol dependence. Psychiatry Investig 2017; 14:325–332Crossref, Medline, Google Scholar
23 : Addiction: beyond dopamine reward circuitry. Proc Natl Acad Sci USA 2011; 108:15037–15042Crossref, Medline, Google Scholar
24 : Severity of neuropsychological impairment in cocaine and alcohol addiction: association with metabolism in the prefrontal cortex. Neuropsychologia 2004; 42:1447–1458Crossref, Medline, Google Scholar
25 : Profound decreases in dopamine release in striatum in detoxified alcoholics: possible orbitofrontal involvement. J Neurosci 2007; 27:12700–12706Crossref, Medline, Google Scholar
26 : High levels of dopamine D2 receptors in unaffected members of alcoholic families: possible protective factors. Arch Gen Psychiatry 2006; 63:999–1008Crossref, Medline, Google Scholar
27 : Indirect effect of corticotropin-releasing hormone receptor 1 gene variation on negative emotionality and alcohol use via right ventrolateral prefrontal cortex. J Neurosci 2014; 34:4099–4107Crossref, Medline, Google Scholar
28 : Mapping common psychiatric disorders: structure and predictive validity in the national epidemiologic survey on alcohol and related conditions. JAMA Psychiatry 2013; 70:199–208Crossref, Medline, Google Scholar
29 : Cellular and molecular mechanisms of drug dependence. Science 1988; 242:715–723Crossref, Medline, Google Scholar
30 : Addiction, a disease of compulsion and drive: involvement of the orbitofrontal cortex. Cereb Cortex 2000; 10:318–325Crossref, Medline, Google Scholar
31 : General and specific inheritance of substance abuse and alcoholism. Arch Gen Psychiatry 1998; 55:964–965Crossref, Medline, Google Scholar
32 : Testing the drug substitution switching-addictions hypothesis: a prospective study in a nationally representative sample. JAMA Psychiatry 2014; 71:1246–1253Crossref, Medline, Google Scholar
33 : Examining heterotypic continuity of psychopathology: a prospective national study. Psychol Med 2017; 47:2097–2106Crossref, Medline, Google Scholar
34 : Increased anxiety and other similarities in temperament of alcoholics with and without antisocial personality disorder across three diverse populations. Alcohol 2007; 41:3–12Crossref, Medline, Google Scholar
35 : Validity of the CAGE questionnaire in an American Indian population. J Stud Alcohol 2001; 62:294–300Crossref, Medline, Google Scholar
36 : The development of alcohol use disorders. Annu Rev Clin Psychol 2005; 1:493–523Crossref, Medline, Google Scholar
37 : Cognitive, emotion control, and motor performance of adolescents in the NCANDA study: contributions from alcohol consumption, age, sex, ethnicity, and family history of addiction. Neuropsychology 2016; 30:449–473Crossref, Medline, Google Scholar
38 : The influence of GABRA2, childhood trauma, and their interaction on alcohol, heroin, and cocaine dependence. Biol Psychiatry 2010; 67:20–27Crossref, Medline, Google Scholar
39 : Adverse childhood experiences and the risk of depressive disorders in adulthood. J Affect Disord 2004; 82:217–225Crossref, Medline, Google Scholar
40 : Equifinality and multifinality in developmental psychopathology. Dev Psychopathol 1996; 8:597–600Crossref, Google Scholar
41 : Dark side of drug dependence. Nature 1994; 371:108–109Crossref, Medline, Google Scholar
42 : Plasticity of reward neurocircuitry and the “dark side” of drug addiction. Nat Neurosci 2005; 8:1442–1444Crossref, Medline, Google Scholar
43 : Drinking to cope with depressive symptoms and ruminative thinking: a multiple mediation model among college students. Subst Use Misuse 2017; 52:52–62Crossref, Medline, Google Scholar
44 : Deconstruction of vulnerability to complex diseases: enhanced effect sizes and power of intermediate phenotypes. Sci World J 2007; 7:124–130Crossref, Google Scholar
45 : From prediction error to incentive salience: mesolimbic computation of reward motivation. Eur J Neurosci 2012; 35:1124–1143Crossref, Medline, Google Scholar
46 : Executive functions in adult offspring of alcohol-dependent probands: toward a cognitive endophenotype? Alcohol Clin Exp Res 2013; 37(suppl 1):E356–E363Crossref, Medline, Google Scholar



