Lower Dopamine D2 Receptor Expression Levels in Human Dopaminergic Neurons Derived From Opioid-Dependent iPSCs
To the Editor: We report the decreased expression level of dopamine D2 receptors in human dopaminergic neurons generated from opioid-dependent induced pluripotent stem cells (iPSCs) compared with dopaminergic neurons generated from a comparison group of iPSCs. We also report the expression of kappa and delta but not mu opioid receptor subtypes in these cells.
The dopaminergic system has been implicated to have an important role in addiction. However, human dopaminergic neurons from drug-dependent subjects were not available for study until recent developments in iPSC technology. Our objective was to study human dopaminergic neurons derived from drug-dependent individuals. The iPSCs were derived from four subjects (two opioid-dependent subjects and two comparison subjects) as shown in Figure 1. We successfully generated midbrain dopamine neurons that expressed forkhead box protein A2, LIM homeobox transcription factor 1 alpha, tyrosine hydroxylase, the dopamine D2 receptor, the Nuclear receptor related 1 protein, the vesicular monoamine transporter 2, and the dopamine transporter (Figure 1). All iPSC lines generated dopamine neurons that released dopamine detectable by high-performance liquid chromatography (ranging from 10.83 nm [SD=1.89] to 60.52 nm [SD=4.83]). There was no apparent difference in dopamine differentiation efficiency between cell lines derived from the comparison subjects and the opioid-dependent subjects with regard to the percentage of cells positive for tyrosine hydroxylase. Importantly, there were different expression levels of D2 receptors in the comparison group’s iPSC-derived dopamine neurons compared with the opioid-dependent group’s iPSC-derived dopamine neurons. Specifically, our results showed lower expression levels of D2 receptors in both of the opioid-dependent iPSC lines compared with the dopamine neurons derived from the comparison group’s iPSC lines (relative dopamine D2 mRNA levels were equal to 0.37 [SD=0.04] and to 0.33 [SD=0.09] for the opioid-dependent dopaminergic neurons, and levels were equal to 0.50 [SD=0.11] and to 0.67 [SD=0.17] for the comparison dopaminergic neurons). These results are consistent with previous neuroimaging findings showing that D2 receptor levels are lower in opioid-dependent subjects and in a variety of other addictions (1, 2).
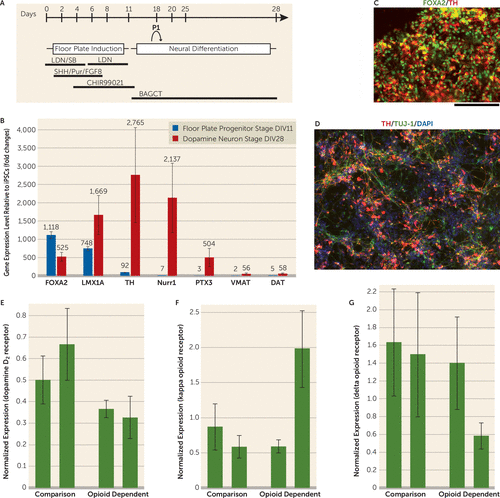
FIGURE 1. Differentiation of Dopamine Neurons From iPSCs Using a Floor Plate–Based Differentiation Protocola
a Panel A shows stages of induction and media components. Panel B shows a comparison of gene-expression levels for various genes at floor plate progenitor stage DIV11 and at dopamine neuron stage DIV28, normalized to expression in undifferentiated induced pluripotent stem cells (iPSCs) and measured by a qualitative real-time polymerase chain reaction. Panel C shows coimmunostaining for forkhead box protein A2 (FOXA2) and tyrosine hydroxylase (TH) in differentiated cells. Panel D shows coexpression of TH/TuJ-1 in differentiated cells at DIV28 (scale bar=100 μm). Expression levels are shown for dopamine D2 receptors (panel E), for kappa opioid receptors (panel F), and for delta opioid receptors (panel G) in opioid-dependent and comparison human dopaminergic neurons. Mu opioid receptor expression was not detected by reverse transcription polymerase chain reaction. P1=passage 1; LDN (LDN-193189) and SB (SB431542) are SMAD inhibitors; SHH=sonic hedgehog; Pur=purmorphamine; FGF8=fibroblast growth factor 8; CHIR99021=inhibitor of glycogen synthase kinase 3; BAGCT comprises brain-derived neurotrophic factor, ascorbic acid, glial cell-derived neurotrophic factor, cAMP, and TGFβ3; LMX1A=LIM homeobox transcription factor 1 alpha; Nurr1=Nuclear receptor related 1 protein; PTX3=pentraxin 3; VMAT=vesicular monoamine transporter; DAT=dopamine transporter; TUJ-1=neuron-specific class III beta-tubulin; DAPI=4',6-diamidino-2-phenylindole. All error bars represent standard deviation.
We also examined the expression of different classes of opioid receptors. Previous reports in animal studies suggest that mu-receptor-specific opioid agonists might excite dopamine neurons only through indirect mechanisms (3). Our data showed that mu opioid receptors were not detected in our differentiated human dopamine neurons derived from iPS cells, supporting this hypothesis in human dopamine neurons. In contrast, kappa and delta opioid receptors were detected in our human dopamine neuronal cultures from all four iPSC lines. This is consistent with reports that kappa opioid agonists can directly inhibit midbrain dopamine neurons and that delta receptor activation in the substantia nigra or ventral tegmental area results in protection in Parkinson’s disease models and protection against alcohol addiction (4, 5). Although iPSCs have been used in multiple disease models, including amyotrophic lateral sclerosis, multiple sclerosis, and Parkinson’s disease (6), the use of iPS cell technology for drug abuse has been very limited. Our data suggest that human iPSC-derived dopamine neurons may serve as a useful model to study human dopamine neurons in vitro and to compare differences between drug-dependent and comparison subjects. In the future, it might also be useful to test toxicity and pharmacological responses to potential treatments on an individual basis.
1 : Dopamine in drug abuse and addiction: results from imaging studies and treatment implications. Mol Psychiatry 2004; 9:557–569Crossref, Medline, Google Scholar
2 : Dopamine D2 receptor availability in opiate-dependent subjects before and after naloxone-precipitated withdrawal. Neuropsychopharmacology 1997; 16:174–182Crossref, Medline, Google Scholar
3 : Opioids excite dopamine neurons by hyperpolarization of local interneurons. J Neurosci 1992; 12:483–488Crossref, Medline, Google Scholar
4 : Kappa-opioid agonists directly inhibit midbrain dopaminergic neurons. J Neurosci 2003; 23:9981–9986Crossref, Medline, Google Scholar
5 : Delta-opioid receptor expression in the ventral tegmental area protects against elevated alcohol consumption. J Neurosci 2008; 28:12672–12681Crossref, Medline, Google Scholar
6 : A fresh look at iPS cells. Cell 2009; 137:13–17Crossref, Medline, Google Scholar



