In Vivo Evidence for Low Striatal Vesicular Monoamine Transporter 2 (VMAT2) Availability in Cocaine Abusers
Abstract
Objective:
Positron emission tomography (PET) imaging studies in cocaine abusers have shown that low dopamine release in the striatum following an amphetamine challenge is associated with higher relapse rates. One possible mechanism that might lead to lower amphetamine-induced dopamine release is low availability of dopamine storage vesicles in the presynaptic terminals for release. Consistent with this hypothesis, postmortem studies have shown low levels of vesicular monoamine transporter, type 2 (VMAT2), the membrane protein that regulates the size of the vesicular dopamine pool, in cocaine abusers relative to healthy subjects. To confirm the postmortem findings, the authors used PET and the VMAT2 radioligand [11C]-(+)-dihydrotetrabenazine (DTBZ) to assess the in vivo VMAT2 availability in a group of 12 recently abstinent cocaine-dependent subjects and matched healthy comparison subjects.
Method:
[11C]DTBZ nondisplaceable binding potential (BPND) was measured by kinetic analysis using the arterial input function or, if arterial input was unavailable, by the simplified reference tissue method.
Results:
[11C]DTBZ BPND was significantly lower in the cocaine abusers than in the comparison subjects in the limbic striatum (10.0% lower), associative striatum (–13.4%), and sensorimotor striatum (–11.5%).
Conclusions:
The results of this in vivo PET study confirm previous in vitro reports of low VMAT2 availability in the striatum of cocaine abusers. It also suggests a compensatory down-regulation of the dopamine storage vesicles in response to chronic cocaine abuse and/or a loss of dopaminergic terminals. Further research is necessary to understand the clinical relevance of this observation to relapse and outcome in abstinent cocaine abusers.
Positron emission tomography (PET) imaging studies have demonstrated lower baseline dopamine levels and less induction of dopamine release by stimulants (methylphenidate or d-amphetamine) in the striatum of chronic cocaine abusers relative to matched healthy comparison subjects (1–4). These studies also suggest that lower dopamine release in the ventral striatum in abstinent cocaine abusers is associated with relapse (2, 4). A possible mechanism leading to lower stimulant-induced dopamine release in cocaine abusers may be related to the loss of dopaminergic terminals from chronic cocaine abuse. As the dopamine transporter is located exclusively in dopaminergic terminals (5), previous studies focused on the differences in dopamine transporter availability to measure dopaminergic terminals in cocaine abusers and comparison subjects. Unfortunately, the results of these postmortem and imaging studies evaluating the dopamine transporter in chronic cocaine abusers were mixed and inconclusive (6). It is likely that the variation in the time since last use of cocaine (i.e., abstinence period) in different patient cohorts led to these discrepant findings (6). This issue has posed a major obstacle to reliable assessment of the available dopamine transporter in cocaine abusers because the dopamine transporter is highly vulnerable to regulation by drugs such as cocaine that alter synaptic dopamine concentrations (7, 8). This led to the investigation of other markers of dopaminergic terminals, such as the vesicular monoamine transporter, type 2 (VMAT2), in cocaine abusers.
VMAT2 is active in presynaptic vesicular membranes (9) and is involved in the transport of the various monoamines, such as serotonin, norepinephrine, and dopamine, from the cytoplasm to their storage vesicles. While VMAT2 is not specific to the vesicle of one particular monoaminergic terminal, VMAT2 in the striatum has been reported to largely (>95%) represent storage vesicles in the dopaminergic terminals (10). Four postmortem studies have contrasted the VMAT2 density in cocaine abusers and matched healthy comparison subjects with radiolabeled derivatives of dihydrotetrabenazine (DTBZ), as they are selective and specific for VMAT2 (11). Three out of these four studies showed significantly less [3H]DTBZ binding (range: –12% to –22%) in the striatum of cocaine abusers relative to comparison subjects (12–15). As lower VMAT2 binding in cocaine abusers was associated with higher dopamine transporter binding in the study by Little and colleagues (13), the authors hypothesized that individuals who had experienced greater exposure to cocaine (as evidenced by a compensatory up-regulation of dopamine transporter) lost more dopamine terminals (as evidenced by lower VMAT2) because of toxicity. Lower VMAT2 binding in cocaine abusers, which results from a reduction in the number of dopamine storage vesicles and/or loss of presynaptic dopamine terminals, is important because it informs the mechanisms that lead to the blunting of stimulant-induced dopamine release in cocaine dependence. As we know of no in vivo studies that have confirmed the lower VMAT2 binding reported in the in vitro literature, we studied the impact of chronic cocaine exposure on striatal VMAT2 nondisplaceable binding potential (BPND) using PET and [11C]DTBZ in a group of 12 chronic cocaine abusers and 12 healthy comparison subjects matched for age, gender, race, and nicotine smoking status. Table 1 contains a glossary of terms relevant to this study.
| Term | Definition |
|---|---|
| Arterial input function | Concentration of radiotracer in blood as a function of time |
| Dopamine transporter availability | Dopamine transporter that is available for binding to its radiotracer |
| Dopaminergic terminal | Presynaptic axon that contains dopaminergic vesicles |
| Kinetic analysis | Analysis that derives information about neuroreceptor PET outcome measures by estimating the rate constants governing transfer between the plasma and brain compartments (requires arterial blood sampling) |
| Plasma free fraction | Fraction of the radioligand that is not bound to plasma proteins at equilibrium |
| Reference tissue method | Analysis that derives information about neuroreceptor PET outcome measures when blood sampling is not possible |
| Vesicular dopamine | Dopamine that is sequestered in a membrane-enclosed sac |
| Vesicular membrane | Membrane surrounding a sac that encapsulates and stores neurotransmitters within the cytoplasm of a presynaptic axon |
| VMAT2 availability | VMAT2 that is available for binding to [11C]DTBZ |
TABLE 1. Glossary of Terms Used in PET Imaging of Vesicular Monoamine Transporter, Type 2 (VMAT2)
Method
Subjects
The 24 participants enrolled in this study were either cocaine-dependent (N=12) or healthy comparison (N=12) subjects. The study was conducted following approvals by the University of Pittsburgh Institutional Review Board and Radioactive Drug Research Committee. All subjects provided written informed consent. Cocaine abusers were recruited through flyers displayed in local community centers, buses, and addiction medicine clinics. The study criteria for the cocaine abusers were 1) age between 18 and 60 years, in persons of either sex and with any ethnic or racial origin; 2) fulfillment of the DSM-IV criteria for cocaine dependence as assessed by the Structured Clinical Interview for DSM-IV; 3) a positive urine screen for cocaine; 4) no DSM-IV axis I disorder other than cocaine abuse or dependence, including abuse of or dependence on alcohol or other drugs (nicotine dependence was allowed); 5) no current use (as confirmed by urine drug screen) of opiates, cannabis, sedative-hypnotics, amphetamines, 3,4-methylenedioxymethamphetamine, or phencyclidine; 6) no current use of any prescription or over-the-counter medication; 7) no current or past severe medical or neurological illnesses (including glaucoma, seizure disorder, or focal finding on magnetic resonance imaging [MRI], such as stroke or tumor) as assessed by a complete medical assessment; 8) no current pregnancy; 9) no history of significant radioactivity exposure (nuclear medicine studies or occupational exposure); and 10) no metallic objects in the body (contraindicated for MRI). All eligible cocaine-dependent subjects completed a minimum of 12 days of outpatient abstinence monitored with witnessed urine sampling (all subjects underwent urine drug screens for cocaine and other recreational drugs 3 times a week for 2 consecutive weeks). Following the 12-day outpatient abstinence period, the subjects were admitted to an inpatient research unit for 2 days before the PET scan. This abstinence monitoring protocol ensured that all subjects were abstinent for a minimum of 2 weeks before the PET scan.
The healthy comparison subjects had no past or present neurological or psychiatric illnesses, including substance abuse (confirmed by urine drug screen both at screening and on the day of the PET scan). They underwent the PET scan as outpatients.
PET Imaging and Data Analysis
Prior to PET imaging, a spoiled gradient recalled sequence (SPGR) MRI scan was obtained by using a GE Signa 1.5-T scanner for determination of regions of interest.
The method reported previously by Kilbourn et al. (11) was used to synthesize [11C]-(+)-α-DTBZ, referred to hereafter as [11C]DTBZ. The PET imaging sessions were conducted with the ECAT EXACT HR+ camera (Siemens Medical Solutions, Knoxville, Tenn.). Following a 10-minute transmission scan, [11C]DTBZ was injected intravenously over 45 seconds, and emission data were collected in the three-dimensional mode for 60 minutes. The scanning duration was based on previous [11C]DTBZ studies in humans that showed time-stable outcome measures (<5% difference in tissue distribution volume, Vt, in both the regions of interest and reference region) at 60 minutes with kinetic analysis (16).
Arterial blood samples were collected for derivation of a metabolite-corrected arterial input function (30 total samples with 20 during the first 2 minutes; see Figure 1). After centrifugation of the samples (2 minutes at 12,500 g), plasma was collected and activity was measured in 200-μl aliquots on a gamma counter. A subset of samples at 2, 15, 30, 45, and 60 minutes were further processed for determination of the fraction of radioactivity corresponding to the parent compound. Metabolite analysis to determine the percentage of unchanged [11C]DTBZ in the plasma was performed by using a solid-phase extraction procedure described previously (16, 17).
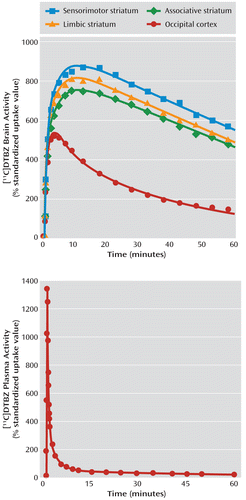
FIGURE 1. [11C]DTBZ Time-Activity Curves in PET Scans of 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjectsa
a [11C]DTBZ is a radioligand selective for vesicular monoamine transporter, type 2 (VMAT2), the membrane protein that regulates the size of the vesicular dopamine pool. The top graph displays [11C]DTBZ time-activity curves in functional subdivisions of the striatum and in the occipital cortex, which was used as a reference region. The lines fitted to the data points are from a three-compartment model. The bottom graph displays a [11C]DTBZ plasma time-activity curve in which the data points are measured activity levels corrected for metabolites and the line is a three-exponential fit.
All region drawing and image analysis was performed by investigators blind to the subjects' diagnoses, using MEDx (Sensor Systems, Sterling, Va.) and SPM2 (Wellcome Trust Centre for Neuroimaging, London). Regions of interest were drawn on the MRI scan and transferred to the coregistered PET scan. The primary region of interest, the striatum, was divided into five anatomical and three functional subdivisions by using published criteria (18). The three functional subdivisions of the striatum were the limbic striatum (which included the ventral striatum), the associative striatum (which included the precommissural dorsal caudate, precommissural anterior putamen, and postcommissural caudate), and sensorimotor striatum (which included the postcommissural putamen). The occipital cortex was used as a reference region (16, 17). Correction for head movement and coregistration of the PET data to the MRI scan were done by using methods described earlier (19).
In this section we specify outcome variables using the consensus nomenclature for in vivo imaging of reversibly binding radioligands (20). Baseline VMAT2 availability was estimated by using the PET outcome measure BPND, i.e., binding potential relative to nondisplaceable uptake. The lack of arterial input function and plasma free fraction measurements in all subjects precluded us from evaluating two other related PET outcome measurements: binding potential relative to total plasma (BPP) and free plasma concentration (BPF) in this data set. As the concentration of VMAT2 is negligible in the occipital cortex (16, 17), such that only free and nonspecifically bound radiotracer is considered to contribute to Vt in the occipital cortex (VtOCC), VtOCC was assumed to be equal to the nondisplaceable distribution volume (VND).
Arterial input function was available for 10 subjects in each group. For them, [11C]DTBZ regional distribution volumes (Vt, in milliliters of plasma per gram of tissue) were derived with kinetic analysis. For this kinetic analysis, a three-compartment model (20) (Figure 1) was used to define both the regions of interest and the reference region, as previously described (21). Following this analysis, [11C]DTBZ BPND was derived as

where ROI is the region of interest, fND is the free fraction of radiotracer in the brain expressed relative to the nondisplaceable concentration, Bavail is the density of VMAT2 available to bind to [11C]DTBZ in vivo, and KD is the equilibrium disassociation constant of [11C]DTBZ. If the arterial input function was unavailable because of inability to place an arterial line (two subjects in each group), [11C]DTBZ BPND was derived by using the simplified reference tissue method (22). Finally, to ensure there was no bias in combining the data from subjects for whom an arterial input function was available and those for whom the input function was not obtained, all of the available data (N=12 in each group) were also analyzed by means of the simplified reference tissue method, without the arterial input function.
Statistical Analysis
Comparisons of group demographic characteristics and baseline scan variables (such as injected dose, mass, plasma clearance, and VND) were performed with unpaired t tests. Group differences in [11C]DTBZ BPND were analyzed with a multivariate analysis of variance (MANOVA) with [11C]DTBZ BPND in the regions of interest (i.e., five anatomical subdivisions) as the dependent measure and diagnostic group as the fixed factor. This primary analysis was followed by contrasts in the individual striatal subdivisions with two-tailed unpaired t tests. A correction for the false discovery rate, with alpha=0.05, was applied to correct for multiple comparisons in the five anatomical subdivisions of the striatum (23). Correlations of the cocaine abusers' VMAT2 availability to the duration of cocaine use and to the amount of money they spent on it were determined by using Pearson product-moment correlation coefficients. A probability value of 0.05 was selected as the significance level for all analyses.
Results
Twelve cocaine-dependent subjects (mean age=43 years, SD=8) and 12 healthy comparison subjects (mean age=41 years, SD=8) were enrolled in this study; each group contained four women and eight men. The groups were matched as well as possible on both ethnicity (cocaine abusers: eight African Americans and four Caucasians; comparison subjects: five African Americans and seven Caucasians) and smoking status (seven smokers in each group). The cocaine abusers reported smoking crack cocaine for a mean of 18 years (SD=7) and were spending an average of $560 (SD=$480) weekly.
Scan Variables
Critical PET scan variables are listed in Table 2. The [11C]DTBZ injected dose, specific activity at the time of injection, and injected mass did not differ between the groups. No significant between-group differences were observed in the rate of [11C]DTBZ clearance from the plasma compartment or in the [11C]DTBZ occipital cortex distribution volume, VND (data available from the 10 subjects in each group for whom arterial line placement was successful).
| Healthy Comparison Subjects | Cocaine-Dependent Subjects | |||
|---|---|---|---|---|
| Variable | Mean | SD | Mean | SD |
| Injected dose of [11C]DTBZ radioligand (mCi) | 10.1 | 1.0 | 10.3 | 0.8 |
| Specific activity at time of injection (Ci/mmol) | 2,660 | 1,340 | 3,270 | 1,672 |
| Injected mass (μg) | 1.5 | 1.0 | 1.2 | 0.5 |
| Clearance (liter/hour)a | 136.9 | 34.9 | 147.6 | 29.9 |
| Occipital distribution volume (ml cm–3)a | 5.30 | 0.44 | 5.56 | 0.66 |
TABLE 2. Variables in PET Scans of 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjects
Regional Volumes
No significant between-group differences were found in the volumes of the regions of interest or reference region (Table 3), suggesting a lack of measurable volumetric changes in the human striatum after chronic cocaine abuse.
| Volume (mm3) | ||||
|---|---|---|---|---|
| Healthy Comparison Subjects | Cocaine-Dependent Subjects | |||
| Region | Mean | SD | Mean | SD |
| Occipital cortex (reference region) | 51,835 | 10,165 | 52,377 | 7,615 |
| Ventral striatum | 1,826 | 336 | 1,708 | 302 |
| Associative striatum | 7,723 | 815 | 7,606 | 828 |
| Precommissural dorsal caudate | 3,436 | 515 | 3,577 | 542 |
| Postcommissural caudate | 2,024 | 292 | 1,886 | 498 |
| Precommissural anterior putamen | 2,262 | 407 | 2,143 | 441 |
| Postcommissural putamen | 4,848 | 1,015 | 4,844 | 518 |
| Whole striatum | 14,396 | 1,699 | 14,158 | 851 |
TABLE 3. Striatal Regional Volumes of 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjects
Measurement of VMAT2 Availability
The MANOVA performed on BPND data derived with kinetic analysis for 10 subjects in each group and with the simplified reference tissue method for two subjects per group demonstrated that the cocaine abusers had significantly lower [11C]DTBZ BPND in the striatal subdivisions relative to the healthy comparison subjects (effect of diagnosis: F=4.98, df=5, 18, p=0.005).
The MANOVA performed on BPND data derived with the simplified reference tissue method for all of the subjects in both groups also demonstrated that the cocaine abusers had significantly lower [11C]DTBZ BPND in the striatal subdivisions than the comparison subjects (effect of diagnosis: F=4.52, df=5, 18, p=0.008).
Values for [11C]DTBZ BPND in the striatum and its subdivisions derived by using a combination of kinetic analysis and the simplified reference tissue method are shown in Table 4; those derived by using only the simplified reference tissue method are shown in Table 5. Also included in these tables are the results of the unpaired t tests that were used to contrast the individual striatal subdivisions. The p values of the five anatomical subdivisions of the striatum in Tables 4 and 5 remained significant (<0.05) after we applied the false discovery rate correction for multiple comparisons. Individual values for the three functional striatal subdivisions appear in Figure 2.
| [11C]DTBZ Nondisplaceable Binding Potential (BPND) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Healthy Comparison Subjects | Cocaine-Dependent Subjects | Two-Tailed, Unpaired t Test | |||||||
| Functional Subdivision | Anatomical Subdivision | Mean | SD | Mean | SD | Group Difference (%) | t | df | p |
| Limbic striatum | Ventral striatum | 2.16 | 0.30 | 1.94 | 0.15 | –10.0 | –2.23 | 16.23 | 0.04 |
| Associative striatumb | 2.34 | 0.20 | 2.03 | 0.16 | –13.4 | –4.22 | 21.24 | 0.0004 | |
| Precommissural dorsal caudate | 2.48 | 0.26 | 2.08 | 0.25 | –16.3 | –3.92 | 21.95 | 0.0007 | |
| Postcommissural caudate | 1.84 | 0.22 | 1.55 | 0.30 | –15.8 | –2.71 | 19.94 | 0.02 | |
| Precommissural anterior putamen | 2.61 | 0.18 | 2.34 | 0.16 | –10.2 | –3.77 | 21.75 | 0.002 | |
| Sensorimotor striatum | Postcommissural putamen | 3.03 | 0.24 | 2.68 | 0.22 | –11.5 | –3.66 | 21.89 | 0.002 |
| Whole striatumc | 2.55 | 0.20 | 2.25 | 0.16 | –11.7 | –4.06 | 20.85 | 0.0006 | |
TABLE 4. [11C]DTBZ Binding Potential in PET Scans of Striatal Regions in 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjects, Derived by Either the Arterial Input Function or Simplified Reference Tissue Methoda
| [11C]DTBZ Nondisplaceable Binding Potential (BPND) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Healthy Comparison Subjects | Cocaine-Dependent Subjects | Two-Tailed, Unpaired t Test | |||||||
| Functional Subdivision | Anatomical Subdivision | Mean | SD | Mean | SD | Group Difference (%) | t | df | p |
| Limbic striatum | Ventral striatum | 2.00 | 0.27 | 1.80 | 0.15 | –10.0 | –2.22 | 17.01 | <0.05 |
| Associative striatumb | 2.14 | 0.20 | 1.84 | 0.20 | –13.9 | –3.73 | 22.00 | 0.002 | |
| Precommissural dorsal caudate | 2.25 | 0.24 | 1.87 | 0.26 | –16.6 | –3.60 | 21.85 | 0.002 | |
| Postcommissural caudate | 1.66 | 0.20 | 1.39 | 0.29 | –16.1 | –2.65 | 19.37 | 0.02 | |
| Precommissural anterior putamen | 2.42 | 0.18 | 2.15 | 0.16 | –11.1 | –3.94 | 21.68 | 0.0007 | |
| Sensorimotor striatum | Postcommissural putamen | 2.74 | 0.21 | 2.44 | 0.20 | –11.3 | –3.68 | 21.99 | 0.002 |
| Whole striatumc | 2.32 | 0.19 | 2.04 | 0.17 | –12.2 | –3.80 | 21.81 | 0.001 | |
TABLE 5. [11C]DTBZ Binding Potential in PET Scans of Striatal Regions of 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjects, Derived by the Simplified Reference Tissue Methoda
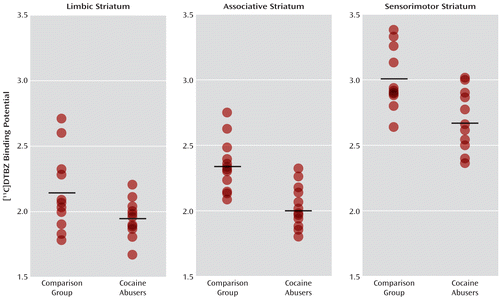
FIGURE 2. Individual Values for [11C]DTBZ Binding Potential in Functional Striatal Subdivisions Shown by PET Scans of 12 Healthy Comparison Subjects and 12 Recently Abstinent Cocaine-Dependent Subjectsa
a [11C]DTBZ is a radioligand selective for vesicular monoamine transporter, type 2 (VMAT2), the membrane protein that regulates the size of the vesicular dopamine pool. The horizontal lines in each graph represent the mean values. The cocaine abusers displayed lower VMAT2 availability than the comparison subjects in all three subdivisions of the striatum (see Table 4).
Age-corrected correlation analyses revealed no significant associations between VMAT2 availability in the striatum and the number of years of cocaine use (r=–0.42, N=12, p=0.20) or the weekly expenditure (r=0.26, df=12, p=0.43). No significant associations were noted when the same correlations were performed for VMAT2 availability in the functional or anatomical subdivisions of the striatum.
Discussion
In this human imaging study, we investigated VMAT2 availability in a group of subjects who regularly abused cocaine for nearly two decades, and we confirmed lower VMAT2 availability (10.0% to 16.3%) in the cocaine-dependent subjects than in matched healthy comparison subjects. The results of this study are in agreement with those from three of four studies that previously evaluated this issue in postmortem brain tissue and showed comparably lower VMAT2 (12% to 22%) in chronic cocaine abusers than in comparison subjects. They are also in agreement with the results of one (24), but not another (25), [11C]DTBZ PET study that evaluated this issue in methamphetamine abusers. The finding in the study by Boileau et al. (25), which demonstrated 10%–22% greater VMAT2 binding in the subdivisions of the striatum in methamphetamine abusers than in healthy comparison subjects, is inconsistent with both the results of a previous study by Johanson et al. (24) of methamphetamine abusers and the results of this study of cocaine abusers, showing lower VMAT2. A possible reason for the paradoxically greater VMAT2 binding in methamphetamine dependence that was reported by Boileau et al. is related to the scanning of a relatively large proportion of the methamphetamine abusers shortly after the cessation of drug use (eight of the 14 methamphetamine-dependent subjects tested positive for methamphetamine and/or cocaine on the day of the PET scan), as opposed to a longer period of abstinence. Thus, it is likely that the evaluation of VMAT2 binding in their study was influenced by methamphetamine-induced transient alterations in dopamine concentration, as demonstrated by these authors in follow-up investigations (26, 27). In our study and the study by Johanson et al. (24), the interaction between cocaine- or methamphetamine-induced transient alterations in dopamine and VMAT2 was less of an issue, as all subjects had been abstinent from drugs for a minimum of 14 days before the PET scan. Nevertheless, as shown in Figure 2, data from this study indicate an overlap in [11C]DTBZ BPND between the cocaine abusers and comparison subjects, which may be due to various demographic and clinical factors, such as social status, the duration and amount of cocaine abuse, or genetic polymorphisms—all of which need to be investigated in a larger cohort.
It is not possible to ascertain from this study whether the lower VMAT2 in cocaine addicts reflects a compensatory down-regulation of presynaptic dopamine storage vesicles, a loss of dopaminergic terminals, or a combination of both. A down-regulation of vesicular dopamine stores may suggest a compensatory mechanism that counteracts the repeated release of dopamine into the synapse that is caused by chronic cocaine abuse. Alternatively, the loss of dopamine terminals would suggest a more permanent cocaine-induced neurotoxicity that leads to reduced dopamine release. Future studies in cocaine addicts are necessary to determine whether there is recovery of [11C]DTBZ binding potential to the levels observed in healthy subjects with a more prolonged duration of abstinence, such as 6 to 12 months. Such a recovery would indicate a compensatory mechanism, as opposed to neurotoxicity of chronic cocaine abuse in humans.
The blunting of stimulant-induced dopamine release in cocaine abusers (Figure 3) is one of the most robust and replicated findings in the addiction imaging literature (1, 2, 4, 28). As more recent studies in laboratory and clinical settings also suggest that this phenomenon is associated with relapse in cocaine addicts, it is critical to understand the mechanisms contributing to lower dopamine release in order to advance therapeutics for addictive disorders (2, 4). Two possibilities have been identified and discussed in the literature with respect to the mechanism of action of amphetamines (and to a lesser extent other stimulants, such as methylphenidate): vesicular depletion, the process by which amphetamine displaces dopamine from secretory vesicles into the neuronal cytoplasm (29), and reverse transport, the process by which cytoplasmic dopamine is released into the extracellular space through outward transport by the dopamine transporter (30). The relative importance of these two processes, vesicular depletion and reverse transport, with respect to the amount of dopamine released following an acute amphetamine challenge has been debated extensively in the literature (31–33). Nevertheless, preclinical studies clearly show that the number (Bmax) of dopamine storage vesicles and dopamine transporters affects the amount of dopamine released after an acute amphetamine challenge (34, 35). On the basis of these data we hypothesized that a decrease in available dopamine storage vesicles and/or dopamine transporters following chronic cocaine use leads to blunting of amphetamine-induced dopamine release in cocaine dependence. In a review of the literature on this topic, we found little evidence to support lower than normal dopamine transporter availability in cocaine abusers (6). Seven of the 10 postmortem studies that contrasted dopamine transporter binding in cocaine abusers and comparison subjects supported greater binding in the cocaine abusers, two studies showed no difference, and one showed lower binding (reviewed in Table 4 of reference 6). The in vivo PET studies that had evaluated this issue were also split—with one suggesting a modest up-regulation of dopamine transporter during early withdrawal (<96 hours) (36) and the other suggesting no change after 3 to 12 weeks of abstinence (37). In contrast to the data on the dopamine transporter, the in vitro literature and this PET study support lower VMAT2 availability in cocaine abusers. As VMAT2 both directly regulates the size of the vesicular dopamine pool and indirectly influences the amount of dopamine that is available in the cytosol for carrier-mediated dopamine release (reverse transport of dopamine transporter), it is likely that lower VMAT2 leads to reduced stimulant-induced dopamine release in cocaine addicts. Investigators in future studies should attempt to understand the clinical relevance of lower VMAT2 to relapse and outcome in abstinent cocaine abusers.
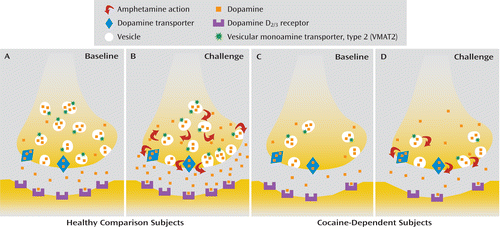
FIGURE 3. Mechanisms Contributing to Blunted Amphetamine-Induced Dopamine Release in Cocaine Abusers, Relative to Healthy Comparison Subjectsa
a At baseline, cocaine abusers (panel C) have 1) fewer vesicular monoamine transporters, 2) a comparable number of dopamine transporters, and 3) fewer dopamine D2/3 receptors than healthy comparison subjects (panel A). After an acute amphetamine challenge (the red arrows show the mechanisms by which amphetamine releases dopamine: reverse transport at the dopamine transporter and depletion of the vesicular dopamine stores), less dopamine is released in the cocaine abusers (panel D) than in the healthy subjects (panel B), which may be due to a reduction in the number of presynaptic dopamine storage vesicles (chronic cocaine-induced adaptation) and/or a loss of terminals (chronic cocaine-induced toxicity). The lower VMAT2 binding in the cocaine abusers in this study, relative to healthy comparison subjects, could be due to either or both of these possibilities.
Finally, in contrast to the postmortem and imaging data from cocaine abusers, no such evidence of lower [3H]DTBZ binding has been found in chronically cocaine-treated rodents (38, 39). This discrepancy in VMAT2 binding between the human and rodent data on chronic cocaine use may explain the paradoxical findings observed with regard to acute amphetamine challenge across species. In human cocaine abusers, low dopamine release is observed, whereas studies of rodents repeatedly exposed to cocaine demonstrate greater dopamine release following an acute amphetamine challenge (termed “sensitization,” reviewed in reference 6). These data suggest that perhaps chronic and repeated exposure to cocaine leads to loss or death of dopaminergic nerve terminals in humans, but not in rodents, which are typically exposed to cocaine for a shorter time (weeks, compared to decades in humans) in the laboratory. Future investigations need to investigate whether relatively modest reductions in presynaptic dopamine nerve terminals (measured as 10% to 20% reductions in VMAT2 binding) lead to profound reductions (60% to 90%) in stimulant-induced dopamine release, as observed in cocaine-abusing humans relative to healthy comparison subjects (2).
In conclusion, we found that repeated exposure to cocaine is associated with lower VMAT2 availability in the striatum of cocaine abusers. This lower striatal VMAT2 availability, which leads to lower vesicular dopamine in the presynaptic terminals, may be one of the several mechanisms that contribute to the lower dopamine release in the brain's reward circuit and thereby drive relapse in cocaine addicts. Further research is necessary to understand the clinical significance of this finding in a much larger group of cocaine-dependent subjects.
1. : Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 1997; 386:830–833Crossref, Medline, Google Scholar
2. : Amphetamine-induced dopamine release: markedly blunted in cocaine dependence and predictive of the choice to self-administer cocaine. Am J Psychiatry 2007; 164:622–629Link, Google Scholar
3. : Lower level of endogenous dopamine in patients with cocaine dependence: findings from PET imaging of D2/D3 receptors following acute dopamine depletion. Am J Psychiatry 2009; 166:1170–1177Link, Google Scholar
4. : Imaging dopamine transmission in cocaine dependence: link between neurochemistry and response to treatment. Am J Psychiatry 2011; 168:634–641Link, Google Scholar
5. : The dopamine transporter is localized to dendritic and axonal plasma membranes of nigrostriatal dopaminergic neurons. J Neurosci 1996; 16:436–447Crossref, Medline, Google Scholar
6. : Cocaine abuse and sensitization of striatal dopamine transmission: a critical review of the preclinical and clinical imaging literature. Synapse 2008; 62:851–869Crossref, Medline, Google Scholar
7. : The vesicular monoamine transporter is not regulated by dopaminergic drug treatments. Eur J Pharmacol 1995; 294:577–583Crossref, Medline, Google Scholar
8. : Effects of dopaminergic drug treatments on in vivo radioligand binding to brain vesicular monoamine transporters. Nucl Med Biol 1996; 23:467–471Crossref, Medline, Google Scholar
9. : Radioligands of the vesicular monoamine transporter and their use as markers of monoamine storage vesicles. Biochem Pharmacol 1989; 38:2395–2404Crossref, Medline, Google Scholar
10. : Striatal monoamine neurotransmitters and metabolites in dominantly inherited olivopontocerebellar atrophy. Neurology 1992; 42:1573–1577Crossref, Medline, Google Scholar
11. : Binding of alpha-dihydrotetrabenazine to the vesicular monoamine transporter is stereospecific. Eur J Pharmacol 1995; 278:249–252Crossref, Medline, Google Scholar
12. : Striatal dopaminergic abnormalities in human cocaine users. Am J Psychiatry 1999; 156:238–245Abstract, Google Scholar
13. : Loss of striatal vesicular monoamine transporter protein (VMAT2) in human cocaine users. Am J Psychiatry 2003; 160:47–55Link, Google Scholar
14. : Striatal dopamine, dopamine transporter, and vesicular monoamine transporter in chronic cocaine users. Ann Neurol 1996; 40:428–439Crossref, Medline, Google Scholar
15. : Radioligand binding and immunoautoradiographic evidence for a lack of toxicity to dopaminergic nerve terminals in human cocaine overdose victims. Brain Res 1997; 747:219–229Crossref, Medline, Google Scholar
16. : Equilibrium versus compartmental analysis for assessment of the vesicular monoamine transporter using (+)-alpha-[C-11]dihydrotetrabenazine (DTBZ) and positron emission tomography. J Cereb Blood Flow Metab 1997; 17:919–931Crossref, Medline, Google Scholar
17. : Reproducibility studies with 11C-DTBZ, a monoamine vesicular transporter inhibitor in healthy human subjects. J Nucl Med 1999; 40:283–289Medline, Google Scholar
18. : Imaging human mesolimbic dopamine transmission with positron emission tomography, part II: amphetamine-induced dopamine release in the functional subdivisions of the striatum. J Cereb Blood Flow Metab 2003; 23:285–300Crossref, Medline, Google Scholar
19. : A comparative evaluation of the dopamine D2/3 agonist radiotracer [11C]NPA and antagonist [11C]raclopride to measure amphetamine-induced dopamine release in the human striatum. J Pharmacol Exp Ther 2010; 63:574–584Google Scholar
20. : Consensus nomenclature for in vivo imaging of reversibly binding radioligands. J Cereb Blood Flow Metab 2007; 27:1533–1539Crossref, Medline, Google Scholar
21. : Kinetic evaluation of [11C]dihydrotetrabenazine by dynamic PET: measurement of vesicular monoamine transporter. J Cereb Blood Flow Metab 1996; 16:1288–1299Crossref, Medline, Google Scholar
22. : Simplified reference tissue model for PET receptor studies. Neuroimage 1996; 4(3, part 1):153–158Crossref, Medline, Google Scholar
23. : Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 1995; 57:289–300Google Scholar
24. : Cognitive function and nigrostriatal markers in abstinent methamphetamine abusers. Psychopharmacology 2006; 185:327–338Crossref, Medline, Google Scholar
25. : Increased vesicular monoamine transporter binding during early abstinence in human methamphetamine users: is VMAT2 a stable dopamine neuron biomarker? J Neurosci 2008; 28:9850–9856Crossref, Medline, Google Scholar
26. : Dopamine modulating drugs influence striatal (+)-[11C]DTBZ binding in rats: VMAT2 binding is sensitive to changes in vesicular dopamine concentration. Synapse 2008; 62:873–876Crossref, Medline, Google Scholar
27. : Influence of a low dose of amphetamine on vesicular monoamine transporter binding: a PET (+)[11C]DTBZ study in humans. Synapse 2010; 64:417–420Crossref, Medline, Google Scholar
28. : Reduced amphetamine-stimulated dopamine release in cocaine addicts as measured by [123I]IBZM SPECT. J Nucl Med 1999; 40(5 suppl):110PGoogle Scholar
29. : Amphetamine and other psychostimulants reduce pH gradients in midbrain dopaminergic neurons and chromaffin granules: a mechanism of action. Neuron 1990; 5:797–808Crossref, Medline, Google Scholar
30. : Evidence for carrier-mediated efflux of dopamine from corpus striatum. Biochem Pharmacol 1982; 31:2479–2484Crossref, Medline, Google Scholar
31. : Weak base model of amphetamine action. Ann N Y Acad Sci 1992; 654:525–528Crossref, Medline, Google Scholar
32. : Amphetamine and other weak bases act to promote reverse transport of dopamine in ventral midbrain neurons. J Neurochem 1993; 60:527–535Crossref, Medline, Google Scholar
33. : Amphetamine: effects on catecholamine systems and behavior. Annu Rev Pharmacol Toxicol 1993; 33:639–677Crossref, Medline, Google Scholar
34. : Presynaptic control of striatal dopamine neurotransmission in adult vesicular monoamine transporter 2 (VMAT2) mutant mice. J Neurochem 2003; 85:898–910Crossref, Medline, Google Scholar
35. : Mechanisms of amphetamine action revealed in mice lacking the dopamine transporter. J Neurosci 1998; 18:1979–1986Crossref, Medline, Google Scholar
36. : Elevated striatal dopamine transporters during acute cocaine abstinence as measured by [123I]β-CIT SPECT. Am J Psychiatry 1998; 155:832–834Abstract, Google Scholar
37. : Cocaine uptake is decreased in the brain of detoxified cocaine abusers. Neuropsychopharmacology 1996; 14:159–168Crossref, Medline, Google Scholar
38. : The vesicular monoamine transporter, in contrast to the dopamine transporter, is not altered by chronic cocaine self-administration in the rat. J Neurosci 1996; 16:3507–3510Crossref, Medline, Google Scholar
39. : Locomotor sensitization and decrease in [3H]mazindol binding to the dopamine transporter in the nucleus accumbens are delayed after chronic treatments by GBR12783 or cocaine. J Pharmacol Exp Ther 1996; 278:330–337Medline, Google Scholar



