A 3p26-3p25 Genetic Linkage Finding for DSM-IV Major Depression in Heavy Smoking Families
Abstract
Objective:
The authors tested for genetic linkage of DSM-IV-diagnosed major depressive disorder in families that were ascertained for cigarette smoking.
Method:
Within a study that targeted families characterized by a history of smoking, analyses derived a subset of 91 Australian families with two or more offspring with a history of DSM-IV major depressive disorder (affected sibling pairs, N=187) and 25 Finnish families (affected sibling pairs, N=33). Within this affected sibling pairs design, the authors conducted nonparametric linkage analysis.
Results:
In the Australian heavy smoking families, the authors found a genome-wide significant multipoint LOD score of 4.14 for major depressive disorder on chromosome 3 at 24.9 cM (3p26-3p25).
Conclusions:
Genome-wide significant linkage was detected for major depressive disorder on chromosome 3p in a sample ascertained for smoking. A linkage peak at this location was also observed in an independent study of major depressive disorder.
Genetic linkage studies of major depressive disorder have found suggestive evidence across multiple genomic regions but with little convergence of findings (1). Genetic association findings, including those from genome-wide association studies (GWASs), have been similarly disparate (2–7). These inconsistencies raise the question of whether major depressive disorder encompasses a number of poorly understood subtypes (e.g., depression in smokers). Among cigarette smokers seeking cessation treatment, lifetime rates of major depression have been estimated at over 60% (8). Cigarette smokers with a history of depression tend to report more severe nicotine withdrawal symptoms (9–11), are more likely to relapse to smoking after an attempt to quit (8), and may be at increased risk for a recurrent episode of depression after smoking cessation (12). Models describing the relationship between symptoms of depression and cigarette smoking include examples of shared genetic risk (13). We carried out genetic linkage analyses of DSM-IV-diagnosed major depressive disorder in two samples that are part of the Nicotine Addiction Genetics project (14, 15), an international consortium focused on tobacco dependence. We used an affected sibling-pair design, in which at least two adult offspring per family reported a history of DSM-IV major depressive disorder, and tested for linkage. Results appear to confirm a genome-wide significant linkage signal at chromosome 3p26-3p25, also reported in an independent linkage study of major depressive disorder conducted by Breen et al. (16).
Method
Samples
The Nicotine Addiction Genetics linkage project enrolled participants at the Queensland Institute of Medical Research in Australia and the University of Helsinki in Finland. Both sites utilized twin registries and targeted families of index cases with a previously reported history of cigarette smoking in interview or questionnaire surveys; the Australian site used a higher threshold measure when recruiting index cases with a history of heavy smoking (14). These original linkage samples included microsatellite marker data and telephone diagnostic interviews for 289 families from the Australian site (offspring, N=917; parents, N=392) and 161 families from the Finnish site (offspring, N=522; parents, N=19). More than 90% of the participants from the Australian site were of Anglo-Celtic or Northern European ancestry, and all of the participants from the Finnish site were of Finnish ancestry. The assessment included a diagnostic telephone interview, adapted from the Semi-Structured Assessment for the Genetics of Alcoholism (17, 18), which obtained a comprehensive evaluation of lifetime DSM-IV (19) major depressive disorder as well as a tobacco use and dependence assessment derived from the Composite International Diagnostic Interview (20). Additional details on the original samples and assessments have been described elsewhere (14, 15, 21). For the present study, we conducted genetic linkage analyses using the 91 Australian families who had one or more affected sibling pairs concordant for a history of major depressive disorder (N=187). In the Finnish sample, only 25 families had one or more affected sibling pairs (N=33), and thus the primary analyses focused on the Australian sample. A summary of the two major depressive disorder linkage samples is presented in Table 1 of the data supplement accompanying the online version of this article.
Analyses
For both the Australian and Finnish samples, 381 autosomal microsatellite markers were genotyped and spaced at approximately 10 cM across the genome, positioned using the deCODE Genetics (Reykjavik, Iceland) map (22). Details of genotyping, including quality control procedures, are described elsewhere (14, 15). Single- and multipoint affected sibling pair nonparametric linkage (the latter using a 2-cM grid) was conducted in MERLIN (Multipoint Engine for Rapid Likelihood Inference [23]), which generated LOD scores (24, 25). Upon detection of LOD scores >3, genome-wide corrected p values were calculated from 1,000 replicates simulated in MERLIN (26, 27).
Follow-up fine mapping included the addition of 290 single nucleotide polymorphisms (SNPs) on chromosome 3 that were available for a portion of the Australian major depressive disorder linkage sample (84 out of 91 families), from other Australian projects with overlapping samples that obtained GWAS data (see reference 28). Through the use of Snagger software (29), these SNPs were selected to be in low linkage disequilibrium (maximum pairwise r2=0.2), to have a minor allele frequency >40%, and to have a minimum distance between two tags of 450 kb in order to optimize information content.
Results
For the Australian linkage sample, a multipoint LOD score of 4.14 for major depressive disorder was found on chromosome 3 (at 24.9 cM). The highest single-point linkage signal emerged at microsatellite marker D3S1304, with a LOD score of 3.7. The LOD score of 4.14 at 3p in this sample met genome-wide significance (p=0.004). A complete report of these single- and multipoint findings, along with marker positions by chromosome, is provided in Table 2 of the data supplement. The addition of 290 SNPs on chromosome 3 narrowed the linkage locus and increased the LOD score to 4.55 (at 25.3 cM) (Figure 1). For the Finnish major depressive disorder sample, a multipoint LOD score of 2.10 was found on chromosome 20 (at 90.9 cM).
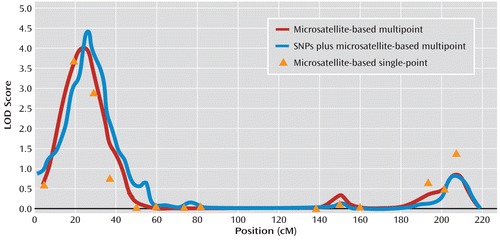
FIGURE 1. Findings Observed on Chromosome 3 in an Australian Major Depressive Disorder Linkage Samplea
a The sample is taken from the Nicotine Addiction Genetics linkage project. The LOD score of 4.14 meets genome-wide significance (p=0.004), with 1,000 simulations in MERLIN.
Given that no linkage signals greater than 1.5 overlapped across both samples, we did not conduct a combined analysis. However, if we were to correct conservatively for testing in both samples by doubling the p value generated from the 1,000 simulations, we would still maintain a genome-wide significant p value of 0.008.
Discussion
Overall, major depressive disorder was associated with a significant genetic linkage peak (a LOD score of 4.14) on chromosome 3 in the Nicotine Addiction Genetics Australian sample, replicating the linkage reported at the same location by Breen et al. (16). These converging findings suggest that the genomic region spanning across 3p26-3p25 is an important area for further investigation in genetic research on major depressive disorder. Given the small number of Finnish affected sibling pairs, the lack of confirmation in this particular sample is not unexpected. The genetic variants accounting for this linkage signal have not yet been convincingly identified. Although our highest single-point microsatellite marker (D3S1304 [LOD score=3.7]) lies within the metabotropic glutamate receptor 7 gene (GRM7), subsidiary association analyses within a one-LOD support interval, using GWAS data available in the Australian sample (28), found only nominal association for two SNPs within GRM7 (p<0.05). Even our strongest association effect, which was a p value of 0.00014 for rs6765537 (a nonsynonymous SNP within C3or20 at 33.87 cM), did not replicate in the Finnish families, nor did it replicate in the Genetic Association Information Network major depressive disorder sample from the Netherlands (2, 30). Thus, while others have found suggestive association between SNPs in GRM7 and major depressive disorder (7, 31), genome-wide significant effects have not been reported. Further, because linkage implicates very broad regions, GRM7 is among many genes that might be hypothesized to explain our signal.
In terms of other linkage findings, one for quantity smoked in samples ascertained for depression has been reported near our chromosome 3 finding for major depressive disorder (32), raising the possibility of common genetic influences across major depressive disorder and smoking-related behavior or of gene-by-environment (i.e., smoking) interaction effects on major depressive disorder.
There are important limitations associated with our results. Our sample of 91 families is small by standards of modern genomic efforts. Thus, we cannot exclude the possibility that our finding is a false positive that coincidentally appears to replicate that of Breen et al. (16), whose sample is much larger (>800 families). Additionally, our finding at 3p26-3p25 (highest peak at 24.9 cM) does not align with the meta-analyses results reported by McMahon et al. (33), who suggest that variants located at 3p21.1 (at approximately 70–72 cM) are associated with mood disorders. Our future efforts to localize the genetic variants influencing major depressive disorder will entail confirmatory analyses in other samples and additional genotyping in this region on chromosome 3p.
1. : The genetics of depression: a review. Biol Psychiatry 2006; 60:84–92Crossref, Medline, Google Scholar
2. : Genome-wide association for major depressive disorder: a possible role for the presynaptic protein piccolo. Mol Psychiatry 2009; 14:359–375Crossref, Medline, Google Scholar
3. : Genome-wide association study of major recurrent depression in the UK population. Am J Psychiatry 2010; 167:949–957Link, Google Scholar
4. : Poor replication of candidate genes for major depressive disorder using genome-wide association data. Mol Psychiatry (Epub ahead of print, March 30, 2010)Medline, Google Scholar
5. : Genome-wide association study of major depressive disorder: new results, meta-analysis, and lessons learned. Mol Psychiatry (Epub ahead of print, November 2, 2010)Google Scholar
6. : Genome-wide association study of recurrent early-onset major depressive disorder. Mol Psychiatry 2011; 16:193–201Crossref, Medline, Google Scholar
7. : Novel loci for major depression identified by genome-wide association study of Sequenced Treatment Alternatives to Relieve Depression and meta-analysis of three studies. Mol Psychiatry 2011; 16:202–215Crossref, Medline, Google Scholar
8. : Heavy smokers, smoking cessation, and clonidine: results of a double-blind, randomized trial. JAMA 1988; 259:2863–2866Crossref, Medline, Google Scholar
9. : Depression and depressive symptoms in smoking cessation. Compr Psychiatry 1990; 31:350–354Crossref, Medline, Google Scholar
10. : Nicotine withdrawal in women. Addiction 1997; 92:889–902Crossref, Medline, Google Scholar
11. : Nicotine withdrawal symptoms in adolescent and adult twins. Twin Res Hum Genet 2010; 13:359–369Crossref, Medline, Google Scholar
12. : Smoking cessation and the course of major depression: a follow-up study. Lancet 2001; 357:1929–1932Crossref, Medline, Google Scholar
13. : Smoking and major depression: a causal analysis. Arch Gen Psychiatry 1993; 50:36–43Crossref, Medline, Google Scholar
14. : Genetic linkage to chromosome 22q12 for a heavy-smoking quantitative trait in two independent samples. Am J Hum Genet 2007; 80:856–866Crossref, Medline, Google Scholar
15. : Linkage of nicotine dependence and smoking behavior on 10q, 7q and 11p in twins with homogeneous genetic background. Pharmacogenomics J 2008; 8:209–219Crossref, Medline, Google Scholar
16. : A genome-wide significant linkage for severe depression on chromosome 3: the Depression Network Study. Am J Psychiatry (published online May 16, 2011;
17. : A new, semi-structured psychiatric interview for use in genetic linkage studies: a report on the reliability of the SSAGA. J Stud Alcohol 1994; 55:149–158Crossref, Medline, Google Scholar
18. : A validity study of the SSAGA: a comparison with the SCAN. Addiction 1999; 94:1361–1370Crossref, Medline, Google Scholar
19.
20. : The CIDI-core substance abuse and dependence questions: cross-cultural and nosological issues: the WHO/ADAMHA Field Trial. Br J Psychiatry 1991; 159:653–658Crossref, Medline, Google Scholar
21. : Genetic linkage findings for DSM-IV nicotine withdrawal in two populations. Am J Med Genet B Neuropsychiatr Genet 2009; 150B:950–959Crossref, Medline, Google Scholar
22. : A high-resolution recombination map of the human genome. Nat Genet 2002; 31:241–247Crossref, Medline, Google Scholar
23. : MERLIN: rapid analysis of dense genetic maps using sparse gene flow trees. Nat Genet 2002; 30:97–101Crossref, Medline, Google Scholar
24. : Allele-sharing models: LOD scores and accurate linkage tests. Am J Hum Genet 1997; 61:1179–1188Crossref, Medline, Google Scholar
25. : A class of tests for linkage using affected pedigree members. Biometrics 1994; 50:118–127Crossref, Medline, Google Scholar
26. : A note on the calculation of empirical p values from Monte Carlo procedures. Am J Hum Genet 2002; 71:439–441Crossref, Medline, Google Scholar
27. : A note on calculation of empirical p values from Monte Carlo procedure. Am J Hum Genet 2003; 72:498–499Crossref, Medline, Google Scholar
28. : Common variants in the trichohyalin gene are associated with straight hair in Europeans. Am J Hum Genet 2009; 85:750–755Crossref, Medline, Google Scholar
29. : Snagger: a user-friendly program for incorporating additional information for tagSNP selection. BMC Bioinformatics 2008; 9:174Crossref, Medline, Google Scholar
30. : Genome-wide association of major depression: description of samples for the GAIN Major Depressive Disorder Study: NTR and NESDA biobank projects. Eur J Hum Genet 2008; 16:335–342Crossref, Medline, Google Scholar
31. : Genome-wide association study of recurrent major depressive disorder in two European case-control cohorts. Mol Psychiatry 2010; 15:589–601Crossref, Medline, Google Scholar
32. : Linkage analysis of smoking initiation and quantity in Dutch sibling pairs. Pharmacogenomics J 2004; 4:274–282Crossref, Medline, Google Scholar
33.



