Reduction of Symptoms by Valacyclovir in Cytomegalovirus-Seropositive Individuals With Schizophrenia
Abstract
OBJECTIVE: The study was an investigation of the effect of the antiviral medication valacyclovir on the symptoms of outpatients with persistent schizophrenia. METHOD: Oral valacyclovir, 1 g twice daily, was administered to 65 outpatients over 16 weeks along with their usual psychiatric medications. Changes in psychiatric symptoms were measured with the Positive and Negative Syndrome Scale and were tested for correlations with antibodies to potentially neurotropic human herpesviruses, as measured by immunoassay before the start of the therapy. RESULTS: There was a significant improvement in the psychiatric symptoms of individuals who were seropositive for cytomegalovirus. Improvement was not associated with antibodies to other herpesviruses or to a range of demographic and clinical variables. CONCLUSIONS: The replication of cytomegalovirus may contribute to the symptoms of schizophrenia in some individuals.
Schizophrenia is a pervasive neuropsychiatric disease of uncertain etiology. Epidemiological and immunohistochemical studies indicate that environmental factors such as infections with members of the family Herpesviridae may contribute to some cases of schizophrenia, most likely in combination with genetic determinants of disease susceptibility (1, 2). We investigated the effect of valacyclovir, a herpesvirus-inhibiting medication, on the symptoms of schizophrenia.
Method
Participants were recruited from the outpatient centers of the Sheppard Pratt Health System and other agencies in the Baltimore area. The inclusion criteria were a diagnosis of schizophrenia or schizoaffective disorder by DSM-IV criteria, age 18–65 years, current treatment with antipsychotic medication that conformed to the relevant Patient Outcomes Research Team recommendations (3), a stable medication regimen over the previous 21 days, the presence of residual symptoms attaining a total score of least 50 on the Positive and Negative Syndrome Scale (4), and the ability to provide informed consent. Persons were excluded from the study if they had mental retardation or an underlying nonpsychiatric medical disorder. Written informed consent was obtained from all study participants.
Before the initiation of therapy, blood samples were obtained for the measurement of antibodies to the following Herpesviridae: herpes simplex virus type 1, herpes simplex virus type 2, cytomegalovirus, Epstein-Barr virus, humanherpes type 6, and varicella-zoster virus. The assays were performed by using commercially available enzyme immunoassays (Viro-Immun Diagnostica, Oberrusel, Germany; Advanced Biotechnologies, Columbia, Md.) and assay methods that have been previously described (5). For each assay, the seropositivity of an individual was defined by comparing the reactivity generated by the sera in the immunoassay with the optical density generated by standard sera provided by the manufacturer. Samples that were reactive to cytomegalovirus by immunoassay were further tested with Western blotting by using previously described methods (6).
After a 2-week lead-in period in which participants were administered inactive medication, treatment was initiated with valacyclovir, 1 g administered orally twice a day for a maximum of 16 weeks. Both the valacyclovir and the inactive medication were administered in addition to the participants’ standard psychiatric medications. Assessments were made with the Positive and Negative Syndrome Scale at the onset of valacyclovir therapy and 2, 4, 8, 12, and 16 weeks after the initiation of therapy. Improvement in each score was calculated as a percentage decrease in the score from baseline. Measurements were performed by trained raters who were masked as to the viral antibody status of the participants.
Statistical analysis was performed by repeated-measures analysis of variance (ANOVA) of scores on the Positive and Negative Syndrome Scale of all 65 participants, with serologic status regarding the herpesvirus as the independent between-group factor and week of therapy as the within-group factor. The normality of the distribution of the scores was documented by tests for kurtosis and skewness. Covariant analyses were also performed by adding potentially confounding demographic and clinical variables as additional between-group factors. A critical value of alpha of 0.008 (0.05/6) was used in light of the testing for antibodies to six viruses.
Results
A total of 65 participants consented to participate in the study, started valacyclovir therapy, and had at least 1 follow-up measurement. The participants had a mean age of 42.7 years (SD=10.6); a total of 47 (72.3%) were male, and 60 (92.3%) were Caucasian. The mean duration of schizophrenia was 22.4 years (SD=10.4). A total of 58 participants completed 16 weeks of therapy. One patient withdrew because of a possible side effect of valacyclovir (rash); the other six dropouts withdrew voluntarily for reasons apparently unrelated to the valacyclovir medication.
We did not find an association between improvement in scores on the Positive and Negative Syndrome Scale and seropositivity for herpes simplex virus type 1, herpes simplex virus type 2, Epstein-Barr virus, varicella-zoster virus, or human herpesvirus 6. However, as depicted in Figure 1, individuals who were seropositive for cytomegalovirus displayed a significant improvement in overall symptoms, i.e., the Positive and Negative Syndrome Scale total symptom score (F=4.61, df=5, p<0.0005). The effect size of the improvement in overall symptoms in cytomegalovirus-seropositive individuals was 0.85. Some degree of improvement was noted in positive symptoms (F=2.48, df=5, p<0.04), negative symptoms (F=1.84, df=5, p<0.11), and general symptoms (F=2.46, df=5, p<0.04). Improvement was most evident after 8 weeks of valacyclovir therapy.
A total of 21 (32. 3%) of the participants were cytomegalovirus seropositive by enzyme immunoassay. The sera of these individuals also reacted to specific cytomegalovirus proteins on Western blot.
We also analyzed the effects of potential confounding variables on the interaction between cytomegalovirus seropositivity and improvement in the Positive and Negative Syndrome Scale total score. Cytomegalovirus seropositivity remained significantly associated with improvement when any of the following variables was added to the repeated-measures ANOVA model: age, duration of illness, number of hospitalizations, gender, race, marital status, initial Positive and Negative Syndrome Scale scores, diagnostic subtype, and type of antipsychotic medication.
Discussion
Valacyclovir, the orally administered l-valyl ester of acyclovir (acycloguanosine), is rapidly converted to acyclovir after ingestion. The administration of the prodrug rather than the native compound results in significantly higher levels of acyclovir in the blood and CNS (7). Acyclovir is converted to its monophosphate derivative by kinases encoded by several different herpesviruses but not by enzymes present in uninfected human cells. The monophosphate is further phosphorylated to the corresponding triphosphate by cellular enzymes, resulting in the inhibition of viral DNA synthesis and viral replication by a number of mechanisms. Clinical trials have documented that valacyclovir is effective in preventing infections due to herpes simplex virus type 1, herpes simplex virus type 2, and cytomegalovirus in susceptible individuals (8).
Our findings indicate that treatment with valacyclovir over 16 weeks can result in some degree of symptom improvement in individuals with schizophrenia who have serological evidence of cytomegalovirus infection. Valacyclovir may be more effective in this regard than native acyclovir, which has been previously reported to be ineffective in altering the symptoms of schizophrenia after oral administration (9). The biological mechanisms of the clinical response to valacyclovir are not known with certainty but are likely to be related to its effect on the replication of cytomegalovirus. Because we did not obtain cerebrospinal or other body fluids for virologic analyses, we could not directly define the relationship between cytomegalovirus replication within the CNS and clinical response to valacyclovir. We also could not determine the effect of latent cytomegalovirus infection on patient symptoms. The definition of these relationships should be the subject of future investigations.
Our exploratory study should be replicated with other groups of individuals with schizophrenia by means of a placebo-control format as well as additional dosing regimens and lengths of treatment. Controlled studies should also be performed with additional medications that have anticytomegalovirus activity to further define the relationship between cytomegalovirus infection and the symptoms of schizophrenia.
Received Oct. 31, 2002; revision received March 24, 2003; accepted June 20, 2003. From the Stanley Research Center, Sheppard Pratt Health Systems, Baltimore; and the Stanley Division of Developmental Neurovirology, Johns Hopkins Children’s Center, Johns Hopkins University School of Medicine. Address reprint requests to Dr. Yolken, Stanley Division of Developmental Neurovirology, Johns Hopkins University, Blalock 1105, 600 N. Wolfe St., Baltimore, MD 21287-4933; [email protected] (e-mail). Supported by the Stanley Medical Research Institute. The authors thank Dr. Glen Ford for performing the Western blot assays, Ms. Bogdana Krivogorsky for performing the radioimmunoassays, Ms. Ann Cusic for data entry and manuscript preparation.
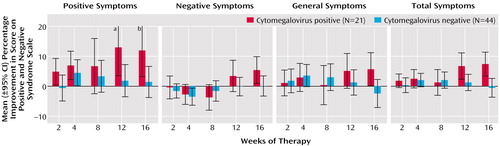
Figure 1. Improvement in Symptoms During Valacyclovir Therapy for Patients With Schizophrenia Who Were Seropositive or Seronegative for Cytomegalovirus
aThe maximum point of the 95% CI is 22.4%.
bThe maximum point of the 95% CI is 20.7%.
1. Yolken RH, Torrey EF: Viruses, schizophrenia, and bipolar disorder. Clin Microbiol Rev 1995; 8:131–145Crossref, Medline, Google Scholar
2. Rothschild DM, O’Grady M, Wecker L: Neonatal cytomegalovirus exposure decreases prepulse inhibition in adult rats: implications for schizophrenia. J Neurosci Res 1999; 57:429–434Crossref, Medline, Google Scholar
3. Lehman AF, Steinwachs DM: Translating research into practice: the schizophrenia Patient Outcomes Research Team (PORT) treatment recommendations. Schizophr Bull 1998; 24:1–10Crossref, Medline, Google Scholar
4. Kay SR, Fiszbein A, Opler LA: The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr Bull 1987; 13:261–276Crossref, Medline, Google Scholar
5. Buka SL, Tsuang MT, Torrey EF, Klebanoff MA, Bernstein D, Yolken RH: Maternal infections and subsequent psychosis among offspring. Arch Gen Psychiatry 2001; 58:1032–1037Crossref, Medline, Google Scholar
6. Grillner L: Screening of blood donors for cytomegalovirus (CMV) antibodies: an evaluation of different tests. J Virol Methods 1987; 17:133–139Crossref, Medline, Google Scholar
7. Perry CM, Faulds D: Valaciclovir: a review of its antiviral activity, pharmacokinetic properties and therapeutic efficacy in herpesvirus infections. Drugs 1996; 52:754–772Crossref, Medline, Google Scholar
8. Ormrod D, Scott LJ, Perry CM: Valaciclovir: a review of its long term utility in the management of genital herpes simplex virus and cytomegalovirus infections. Drugs 2000; 59:839–863Crossref, Medline, Google Scholar
9. DeLisi LE, Goldin LR, Nurnberger JI, Simmons-Alling S, Hamovit J, Dingman CW: Failure to alleviate symptoms of schizophrenia with the novel use of an antiviral agent, acyclovir (Zovirax). Biol Psychiatry 1987; 22:216–220Crossref, Medline, Google Scholar



