Association of the Dysbindin Gene With Bipolar Affective Disorder
Abstract
Objective: In the study of bipolar affective disorder and schizophrenia, there is some evidence suggesting a phenotypic and genetic overlap between the two disorders. A possible link between bipolar affective disorder and schizophrenia remains arguable, however. The authors hypothesized that dysbindin, which is a probable susceptibility gene for schizophrenia, was associated with bipolar affective disorder and tested this hypothesis using a case-control design study. Method: Participants included 213 patients with bipolar I disorder and 197 comparison subjects. In each subject, 10 polymorphisms in the dysbindin gene were genotyped and assessed. Results: Two polymorphisms showed individual genotypic association with bipolar I disorder. Multiple marker haplotypes were more strongly associated, with the rarer of the two common haplotypes being overrepresented in the patients with bipolar affective disorder. A similar finding was reported in patients with schizophrenia in a previous study. Conclusions: Findings suggest that the human dysbindin gene may play a role in the susceptibility to bipolar affective disorder, which underscores a potentially important area of etiological overlap with schizophrenia. The existence of shared genetic risk factors will, in time, lead to changes in the current nosology of major psychoses.
Current nosology separates bipolar affective disorder and schizophrenia into nonoverlapping categories with distinct etiologies (the Kraepelinian dichotomy). This view is supported by the majority of family studies of psychosis. However, the alternative view, that psychosis is a continuum with etiological overlap existing between schizophrenia and bipolar affective disorder, is supported by gradually accumulating evidence from genetic epidemiology, including the results of some twin and family studies and, more recently, linkage and association analysis (1) . Two examples of such studies include a large population-based study in Denmark that found that patients with bipolar disorder have an elevated family history of both schizophrenia and schizoaffective disorder, albeit to a much lesser extent than bipolar disorder (2) , and a study that reported that the relatives of both patients with bipolar disorder and patients with schizophrenia may be at higher risk of developing unipolar depression and schizoaffective disorder, if not schizophrenia and bipolar disorder themselves (1) . These studies, taken together with other evidence from genetic epidemiology, suggest that bipolar affective disorder and schizophrenia are partially, but not fully, separable.
Psychotic features may represent a link, since they show familial clustering in bipolar affective disorder (3) , and families with psychotic bipolar disorder show a link to schizophrenia loci on chromosomes 22q and 13q (4) . Thus, it is important to determine whether genes implicated in the etiology of schizophrenia play a role in bipolar affective disorder and vice versa. Among these genes, there is strong evidence that the dysbindin gene on chromosome 6p is associated with schizophrenia (5) . There is modest evidence for a linkage between chromosome 6p and bipolar affective disorder, and stronger evidence that loci on chromosomes 6q and 6p interact to increase susceptibility for bipolar disorder (6). Given this and the other evidence suggesting etiological and phenotypic overlap between the two disorders, it is important to explore the potential role of this gene in the susceptibility to bipolar affective disorder. To explore this hypothesis, we analyzed the frequency of genetic variations in the dysbindin gene in a group of patients with bipolar I disorder relative to comparison subjects.
Method
Subjects were recruited through psychiatric hospitals in Scotland, met DSM-IV criteria for bipolar I disorder, and had been receiving lithium therapy for at least 3 years. All subjects were Caucasian with at least three of their four grandparents born in Scotland. Consensus diagnosis was determined by trained psychologists and psychiatrists based on case-note review and clinical interview using semistructured diagnostic questionnaires. The Operational Criteria Checklist for Psychotic Illness program was used to define diagnoses. Patients with schizoaffective disorder were excluded. Final decisions on any diagnostic disagreements ([N=31] for reasons of missing data or discrepancies between DSM-IV and International Classification of Diseases-10 diagnoses [N=31]) were made by one of the authors (D.St.C.) and corroborated by another (R.S.).
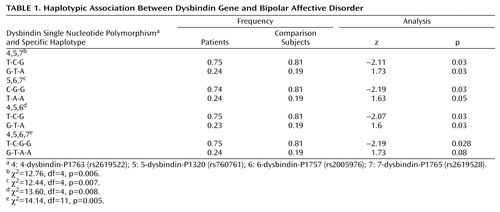
The following 10 dysbindin single nucleotide polymorphisms were analyzed from the previous study conducted by Straub and colleagues: 2-dysbindin-P1583 (rs909706); 3-dysbindin-P1578 (rs1018381); 4-dysbindin-P1763 (rs2619522); 5-dysbindin-P1320 (rs760761); 6-dysbindin-P1757 (rs2005976); 7-dysbindin-P1765 (rs2619528); 8-dysbindin-P1325 (rs1011313); 9-dysbindin-P1635 (rs3213207); 10-dysbindin-P1655 (rs2619539); and 11-dysbindin-P1287 (rs760666). An eleventh single nucleotide polymorphism (1-dysbindin-A [rs2619538]; see Williams and colleagues [7] ) in the promoter region was also genotyped. Genotyping was conducted using KBiosciences (http://www.kbioscience.co.uk), with a competitive allele specific polymerase chain reaction system.
Power analysis was performed using the genetic power calculator (http://statgen.iop.kcl.ac.uk/gpc/). Statistical analysis was performed using both χ 2 and Fisher’s exact test to analyze individual polymorphisms. GENECOUNTING software (version 1.3, March 2003) (8) was used to estimate the haplotype frequencies (with 1,000 permutations/simulations).
Results
All single nucleotide polymorphisms were in Hardy-Weinberg equilibrium. Our study group had 97% power to detect a heterozygote odds ratio of 2 at the 0.05 level for a risk allele frequency of 0.19. Single nucleotide polymorphisms P1757 and P1320 had p values <0.05 under a recessive genotypic model for the rare allele. Under this recessive genotypic model, P1320 was most significant, with an odds ratio of 0.33 (95% confidence interval=0.12–0.93), (χ 2 =4.39, df=1, p<0.04 [data not shown; available upon request]). Table 1 shows the most significant 3-marker haplotypes and empirical p values. The 4-5-6-7 haplotype was positive with a significance of p=0.005.
Discussion
We found an individual marker and haplotype-based association between the dysbindin gene and bipolar affective disorder. This finding concurs with the original association reported by Straub and colleagues (5) and van den Oord and colleagues (9) in that the less common alleles were greater in patients relative to healthy comparison subjects. Our finding does not, however, concur with the data reported by Schwab and colleagues (10) or Williams and colleagues (7) , since we found no evidence of a role for the common allele or the single nucleotide polymorphism 1-dysbindin-A (identified by Williams and colleagues) in the promoter region of the dysbindin gene.
It is unlikely that our clinical diagnostic procedures failed to fully separate schizophrenia from bipolar affective disorder, since 1) all patients had been receiving lithium therapy for at least 3 years, and thus subjects who had experienced first-break psychosis, in which the diagnostic boundaries are less certain, were not included, and 2) patients with schizoaffective disorder were specifically excluded.
Our finding provides additional molecular genetic evidence that a portion of the phenotypic overlap between schizophrenia and bipolar affective disorder is caused by the common effects of a shared gene, dysbindin. In addition, this may suggest a real molecular basis of the overlapping symptoms in both schizophrenia and bipolar affective disorder.
1. Berrettini W: Evidence for shared susceptibility in bipolar disorder and schizophrenia. Am J Med Genet C Semin Med Genet 2003; 15:59–64Google Scholar
2. Mortensen PB, Pedersen CB, Melbye M, Mors O, Ewald H: Individual and familial risk factors for bipolar affective disorders in Denmark. Arch Gen Psychiatry 2003; 60:1209–1215Google Scholar
3. Potash JB, Willour VL, Chiu YF, Simpson SG, MacKinnon DF, Pearlson GD, DePaulo JR Jr, McInnis MG: The familial aggregation of psychotic symptoms in bipolar disorder pedigrees. Am J Psychiatry 2001; 158:1258–1264Google Scholar
4. Potash JB, Zandi PP, Willour VL, Lan TH, Huo Y, Avramopoulos D, Shugart YY, MacKinnon DF, Simpson SG, McMahon FJ, DePaulo JR Jr, McInnis MG: Suggestive linkage to chromosomal regions 13q31 and 22q12 in families with psychotic bipolar disorder. Am J Psychiatry 2003; 160:680–686Google Scholar
5. Straub RE, Jiang Y, MacLean CJ, Ma Y, Webb BT, Myakishev MV, Harris-Kerr C, Wormley B, Sadek H, Kadambi B, Cesare AJ, Gibberman A, Wang X, O’Neill FA, Walsh D, Kendler KS: Genetic variation in the 6p22.3 gene DTNBP1, the human ortholog of the mouse dysbindin gene, is associated with schizophrenia. Am J Hum Genet 2002; 71:337–348Google Scholar
6. Schulze TG, Buervenich S, Badner JA, Steele CJ, Detera-Wadleigh SD, Dick D, Foroud T, Cox NJ, MacKinnon DF, Potash JB, Berrettini WH, Byerley W, Coryell W, DePaulo JR Jr, Gershon ES, Kelsoe JR, McInnis MG, Murphy DL, Reich T, Scheftner W, Nurnberger JI Jr, McMahon FJ: Loci on chromosomes 6q and 6p interact to increase susceptibility to bipolar affective disorder in the National Institute of Mental Health Genetics Initiative pedigrees. Biol Psychiatry 2004; 56:18–23Google Scholar
7. Williams NM, Preece A, Morris DW, Spurlock G, Bray NJ, Stephens M, Norton N, Williams H, Clement M, Dwyer S, Curran C, Wilkinson J, Moskvina V, Waddington JL, Gill M, Corvin AP, Zammit S, Kirov G, Owen MJ, O’Donovan MC: Identification in two independent samples of a novel schizophrenia risk haplotype of the dystrobrevin binding protein gene (DTNBP1). Arch Gen Psychiatry 2004; 61:336–344Google Scholar
8. Zhao JH, Lissarrague S, Essioux L, Sham PC: Genecounting: haplotype analysis with missing genotypes. Bioinformatics 2002; 18:1694–1695Google Scholar
9. van den Oord EJ, Sullivan PF, Jiang Y, Walsh D, O’Neill FA, Kendler KS, Riley BP: Identification of a high-risk haplotype for the dystrobrevin binding protein 1 (DTNBP1) gene in the Irish study of high-density schizophrenia families. Mol Psychiatry 2003; 8:499–510Google Scholar
10. Schwab SG, Knapp M, Mondabon S, Hallmayer J, Borrmann-Hassenbach M, Albus M, Lerer B, Rietschel M, Trixler M, Maier W, Wildenauer DB: Support for association of schizophrenia with genetic variation in the 6p22.3 gene, dysbindin, in sib-pair families with linkage and in an additional sample of triad families. Am J Hum Genet 2003; 72:185–190Google Scholar