In Search of Biomarkers to Guide Interventions in Autism Spectrum Disorder: A Systematic Review
Abstract
Objective:
The aim of this study was to catalog and evaluate response biomarkers correlated with autism spectrum disorder (ASD) symptoms to improve clinical trials.
Methods:
A systematic review of MEDLINE, Embase, and Scopus was conducted in April 2020. Seven criteria were applied to focus on original research that includes quantifiable response biomarkers measured alongside ASD symptoms. Interventional studies or human studies that assessed the correlation between biomarkers and ASD-related behavioral measures were included.
Results:
A total of 5,799 independent records yielded 280 articles for review that reported on 940 biomarkers, 755 of which were unique to a single publication. Molecular biomarkers were the most frequently assayed, including cytokines, growth factors, measures of oxidative stress, neurotransmitters, and hormones, followed by neurophysiology (e.g., EEG and eye tracking), neuroimaging (e.g., functional MRI), and other physiological measures. Studies were highly heterogeneous, including in phenotypes, demographic characteristics, tissues assayed, and methods for biomarker detection. With a median total sample size of 64, almost all of the reviewed studies were only powered to identify biomarkers with large effect sizes. Reporting of individual-level values and summary statistics was inconsistent, hampering mega- and meta-analysis. Biomarkers assayed in multiple studies yielded mostly inconsistent results, revealing a “replication crisis.”
Conclusions:
There is currently no response biomarker with sufficient evidence to inform ASD clinical trials. This review highlights methodological imperatives for ASD biomarker research necessary to make definitive progress: consistent experimental design, correction for multiple comparisons, formal replication, sharing of sample-level data, and preregistration of study designs. Systematic “big data” analyses of multiple potential biomarkers could accelerate discovery.
Autism spectrum disorder (ASD) is a neurodevelopmental syndrome defined in DSM-5 by a combination of symptoms across two clinical domains: “persistent deficits in social communication and social interaction” and “restricted, repetitive patterns of behavior, interests, or activities,” which can include sensory processing difficulties (1). It is a common disorder, currently estimated to affect almost 2% of 8-year-old children, leading to substantial morbidity (2).
Biomarkers are characteristics that can be measured accurately and reproducibly in individual patients to provide objective and quantifiable metrics of clinically relevant processes (3). They can reflect typical biological processes, pathogenic processes, or responses to an exposure or intervention (4) and include molecular, physiological, and anatomical measures (5).
To date, no biomarkers have passed the multi-step approval process of the U.S. Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) Biomarker Qualification Program (BQP) to be “qualified” for use in ASD. The U.S. Food and Drug Administration and National Institutes of Health Biomarker Working Group generated the “Biomarkers, Endpoints, and Other Tools” (BEST) resource to harmonize biomarker terminology. BEST defines several biomarker categories based on use case, such as diagnostic and safety biomarkers. Here, we focus on response biomarkers, defined by BEST as “a biomarker used to show that a biological response, potentially beneficial or harmful, has occurred in an individual who has been exposed to a medical product or an environmental agent” (4). Given the strong evidence for pathological vulnerability during fetal and perinatal development (6, 7), the challenges of early detection of social deficits, and the paucity of somatic treatments that target ASD-defining deficits (8), more reliable, biologically based assays would be transformative. In light of the substantial recent progress in the genetics and biology of ASD and the associated promise of identifying novel molecular treatment targets (9), identifying reliable response biomarkers in ASD could revolutionize the field, providing a standardized metric to assess and refine therapeutic strategies (5).
There is ample reason to be optimistic that response biomarkers can be found. Over the past decade, substantial progress has been made in identifying specific genes that dramatically increase the risk for ASD (6). Moreover, the study of these definitive molecular risk factors, both individually and collectively, have identified a wide range of potential biological mechanisms (6, 10) and also provided evidence that these genes converge to disrupt a smaller number of molecular pathways, cell types, and circuits in particular brain regions at specific points in development, resulting in the clinical phenotype (6, 11–15). Indeed, while the genetic contribution to ASD has been defined in only a minority of affected individuals, findings to date strongly suggest that markers of altered biological processes are likely identifiable, whether there is contribution from rare large-effect mutations, common polygenic inheritance, or environmental factors, all of which play a role in ASD pathogenesis (16).
The heterogeneity of ASD etiology is mirrored in the clinic in the heterogeneity of symptoms and comorbid disorders. The diagnosis itself is defined by a constellation of behaviors in multiple combinations (17), and while all cases must exhibit impairments in the two DSM-5-defined clinical domains, other important features, such as sensory processing impairment, cognitive impairment, language delay, or stereotypic behavior, are present only in subsets of patients. This extensive heterogeneity and the absence of a clearly defined proxy for ASD symptoms in experimental model systems complicate the search for biomarkers, the pursuit of causal relationships, and the development of therapeutics.
To catalog and evaluate progress in identifying a response biomarker in ASD, we performed a systematic literature review with the objective of documenting which biomarkers have been tested and whether any biomarkers showed evidence of replication. We identified 1,057 original research articles that include a biomarker in individuals with ASD or in ASD-related animal models (Figure 1; see also Table S1 in the online supplement). To focus on response biomarkers, an in-depth review led us to a subset of 280 articles that included both a biomarker and a measurement of ASD symptoms, either with a therapeutic intervention or showing evidence of the relationship between the biomarker and ASD behaviors. This analysis demonstrates that, to date, no ASD response biomarkers meet the exacting standards necessary to inform clinical trials and highlights key methodological imperatives necessary for the field to achieve success.
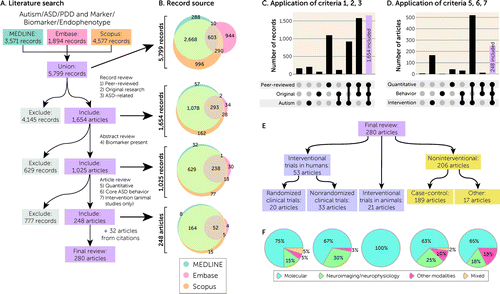
FIGURE 1. Overview of database searcha
aAs shown in panel A, 5,799 independent records were identified from searches of MEDLINE, Embase, and Scopus. Applying seven inclusion criteria in three steps yielded 248 articles, with an additional 32 articles included from citations, giving a final review set of 280 articles. In panel B, at each step of the application of inclusion criteria, the overlap between the three databases is shown by Venn diagrams. In panels C and D, the number of records that met or did not meet combinations of criteria 1–3 or criteria 5–7 is shown in UpSet plots. A complete record of this process is recorded in Table S1 in the online supplement. In panel E, the 280 articles are subdivided by experimental design. Panel F shows the proportions of the types of biomarkers assessed in each of the five types of experimental design. ASD=autism spectrum disorder; PDD=pervasive developmental disorder.
Methods
A systematic search of the literature was performed following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (18), although we note that our study was not registered in advance and includes an additional 32 articles identified from citations of the reviewed papers that were not captured by our initial database search (see Table S2 in the online supplement). In April 2020, three databases (MEDLINE, Embase, and Scopus) were searched for relevant articles from January 1, 1900, to February 29, 2020, with the terms (autism, ASD, pervasive developmental disorder, or PDD) and (biomarker, marker, or endophenotype). For exact search terms, methods, and the results of these searches, see the Methods section and Table S1 in the online supplement. Our initial search identified 3,571 MEDLINE records, 1,894 Embase records, and 4,577 Scopus records (Figure 1A). Duplicate records within and across these databases were identified to yield a total of 5,799 independent records (Figure 1; see also Table S1 in the online supplement).
Two authors (A.A. and A.S.J.) independently applied a first round of filtering based on the title, article type, and language of the record, using three inclusion criteria:
Criterion 1: The article must be peer-reviewed and published in English.
Criterion 2: The article must describe original research.
Criterion 3: The article must focus on nonsyndromic ASD, although it may use a different term, such as pervasive developmental disorder or Asperger syndrome.
Applying these criteria, we retained 1,654 records (28.5%, 1,654/5,799) and excluded 4,145 records (Figure 1A–C). The majority of exclusions (70.2%, 2,908/4,145) were due to not meeting criterion 3 (focus on ASD), followed by criterion 2 (original research) in 1,121 of the remaining 1,237 (90.6%). For the 1,654 records retained, we assessed our fourth inclusion criterion by reading the abstract and, if necessary, the full article:
Criterion 4: The article must describe new data assessing at least one biomarker.
Applying this fourth criterion identified 1,025 records of biomarkers in ASD (62.0%, 1,025/1,654) and excluded 629 (Figure 1A and 1B). For all 1,025 records, we assessed the full article to apply three additional criteria:
Criterion 5: The biomarker assessed must have been both quantifiable and potentially variable (i.e., not fixed/structural).
Criterion 6: The research must have included a measure of ASD severity using behavioral measures or scales to assess social-communication and/or repetitive and restrictive behaviors. Noninterventional studies must also assess the association between these measures and the biomarker.
Criterion 7: If the research was based on animal models, there must have been an intervention.
To assess these criteria, each article was assessed independently by at least two of the authors (A.A., A.S.J., M.P., M.B., E.U.), and discrepancies were reviewed by an additional author. Applying the remaining criteria yielded a final sample of 248 articles (24.2%, 248/1,025) and excluded 777 (Figure 1A, 1B, and 1D). The majority of exclusions (94.3%, 733 of 777) were due to criterion 6 (requirement for a measure of ASD severity), with 567 of the 733 being case-control studies for which the study needed to assess the relationship between the biomarker and ASD severity for inclusion (Figure 1D). We note that these 777 excluded articles may nevertheless provide insights into potential ASD diagnostic biomarkers for future study, and we include a complete list of these articles in Table S1 in the online supplement. From the citations of these 248 articles, we identified another 32 articles that met our seven inclusion criteria but had not been identified by the initial search, due to the absence of the words “biomarker,” “marker,” or “endophenotype” in the title or abstract, to yield 280 articles for final review. Table S1 in the online supplement details all 5,799 articles and the outcome as these criteria were applied sequentially, plus the 32 articles identified from references cited by the articles we reviewed. We developed a standardized data extraction form, which five authors (A.A., A.S.J., M.P., M.B., and E.U.) used to extract data manually from all 280 eligible studies. If data were unclear or ambiguous, a consensus decision was taken by all five authors. The following metrics were extracted: biomarkers, study design, sample size, trial registration number if applicable (i.e., interventional study), ASD diagnostic criteria, inclusion and exclusion criteria, intervention (including dose and duration), primary outcomes, behavioral measures, and participants’ age, sex, and cognitive ability (see Tables S3 and S4 in the online supplement). For the most frequently analyzed biomarkers, we also extracted the following outcomes: statistical association for the biomarker, direction of effect for the biomarker, whether the biomarker correlated with behavioral symptoms, and, for interventional studies, whether the intervention led to behavioral improvement. Where multiple outcomes were stated, the result based on the largest sample size was recorded. Missing data were recorded as “not stated.” For response biomarkers with consistent directions of effect across multiple studies (Tables 1 and 2), we also extracted t statistics or means and standard deviations in cases and controls; studies in which these metrics were not reported were excluded from this step.
| Biomarker and Referenceb | Tissue or Measurement | Sample Size (ASD/control) | Age (years) (range, or mean and SD of ASD group) | Biomarker Outcome | ASD Behavior Measure | Biomarker and Behavior Correlation |
|---|---|---|---|---|---|---|
| Glutathione | ||||||
| Geier et al., 2009 | Plasma | 148 (28/120) | 2–13 | CT>ASD | CARS | No |
| Adams et al., 2011 | Plasma | 99 (55/44) | 5–16 | CT>ASD | ATEC, PDDBI, SAS | Yes |
| Lakshmi et al., 2011 | Plasma | 90 (45/45) | 4–12 | CT>ASD | CARS | No |
| Frye et al., 2013 | Plasma | 72 (18/54) | 8.2 (3.1) | CT>ASD | SRS, VABS | No |
| Vijayashankar et al., 2014 | Saliva | 60 (40/20) | 4–12 | CT>ASD | CARS | Yes |
| Durieux et al., 2016 | Brain, MRS | 50 (21/29) | 18–50 | Not different | ADI-R, ADOS | No |
| Meguid et al., 2018 | Plasma | 120 (90/30) | 2–7 | CT>ASD | CARS | Yes |
| IL-6 | ||||||
| Emanuele et al., 2010 | Serum | 50 (22/28) | 19–42 | ASD>CT | ADI-R-soc, VABS | Not reported |
| Ashwood et al., 2011a | PBMC | 72 (37/32) | 2–5 | Not stated | AbBC, ADOS, regression, VABS | No |
| Ashwood et al., 2011b | Plasma | 223 (97/87/39DD) | 2–5 | ASD>DD, CT | ADI-R, regression | Yes |
| Napolioni et al., 2013 | Plasma | 50 (25/25) | 0–25 | Not different | SRS | No |
| Ricci et al., 2013 | Serum | 58 (29/29) | 2–21 | ASD>CT | CARS | No |
| Yang et al., 2015 | Plasma | 64 (33/31) | 4.5 (1.3) | ASD>CT | CARS | Yes |
| Ferguson et al., 2016 | Serum | 120 (120/0)c | 6–18 | NA | VABS | Yes |
| Jácome et al., 2016 | Plasma | 32 (17/15) | 3–9 | ASD>CT | CARS | Yes |
| Careaga et al., 2017 | PBMC, LPS stimulated | 50 (50/16) | 2–4 | Not stated | ADOS | Yes |
| Guloksuz et al., 2017 | Plasma | 75 (40/35) | 7.0 (4.0) | Not different | CARS, regression | Not reported |
| Krakowiak et al., 2017 | Blood spot | 303 (214/62/27DD) | Neonatal | Not different | VABS | No |
| Masi et al., 2017 | Plasma | 144 (113/31)d | 2–18 | Not stated | SRS | No |
| Ning et al., 2019 | Serum | 204 (102/102) | 4.5 (1.3) | ASD>CT | CARS | Yes |
| Prosperi et al., 2019 | Plasma | 85 (85/0)c | 2–6 | NA | Regression | No |
| BDNF | ||||||
| Correia et al., 2010 | Platelet-rich plasma | 196 (146/50) | 2–18 | ASD>CT | CARS | Yes |
| Mansour et al., 2010 | Serum | 40 (20/20) | 3–12 | CT>ASD | CARS | No |
| Ray et al., 2011 | Plasma | 39 (21/18) | 2–18 | CT>ASD | CARS | No |
| Ricci et al., 2013 | Serum | 58 (29/29) | 2–21 | ASD>CT | CARS | No |
| Zhang et al., 2014 | Serum | 120 (60/60) | 3.8 (1.2) | ASD>CT | CARS | Yes |
| Segura et al., 2015 | Whole blood, RNA | 31 (21/10) | 12–44 | ASD>CT | ToM | No |
| Wang et al., 2015 | Serum | 150 (75/75) | 4.0 (1.3) | ASD>CT | CARS | Yes |
| Meng et al., 2017 | Serum | 164 (82/82) | 4.0 (1.3) | ASD>CT | CARS | Yes |
| Francis et al., 2018 | Serum | 71 (45/26) | 2.5–3.5 | CT>ASD | ADOS, CARS, VABS | No |
| Serotonin | ||||||
| Alabdali et al., 2014 | Platelet-free plasma | 82 (52/30) | 3–12 | CT>ASD | CARS, SRS | Yes |
| Yang et al., 2015 | Whole blood | 64 (33/31) | 12.4 (2.4) | ASD>CT | CARS | Yes |
| Russo et al., 2013 | Plasma | 77 (48/29) | 8.0 (ND) | ASD>CT | Pfeiffer Q | Yes |
| Alabdali et al., 2014 | Plasma | 82 (52/30) | 3–12 | ASD>CT | CARS, SRS | Yes |
| GABA | ||||||
| Brix et al., 2015 | Brain, anterior cingulate | 38 (14/24) | 10.2 (1.9) | Not different | ASSQ | Yes |
| Carvalho Pereira et al., 2018 | Brain, mOFC | 34 (20/14) | 10–18 | Not different | ADI-R | Yes |
| Meguid et al., 2018 | Plasma | 120 (90/30) | 2–7 | ASD>CT | CARS | Yes |
| Saleem et al., 2020 | Plasma | 118 (54/64) | 5.6 (2.1) | Not different | CARS | No |
| Oxytocin | ||||||
| Alabdali et al., 2014 | Platelet-free plasma | 82 (52/30) | 3–12 | CT>ASD | CARS, SRS | Yes |
| Oztan et al., 2018 | CSF | 72 (36/36) | 1.5–9 | Not stated | ADOS | No |
| Aita et al., 2019 | Serum | 79 (54/25) | 16–60 | Not stated | CARS, RBS-R | Yes |
| Alpha power | ||||||
| Van Hecke et al., 2009 | EEG, right temporal-parietal | 33 (19/14) | 8–12 | Not different | SRS, SSRS | No |
| Cornew et al., 2012 | EEG, absolute and relative power | 50 (27/23) | 6–15 | ASD>CT | SRS | Yes (relative power) |
| Kozhushko et al., 2018 | EEG, group independent components | 112 (42/70) | 4–9 | Not different | Self-devised scale (psychological function) | No |
| Keehn et al., 2017 | EEG, posterior left, central, right | 40 (19/21) | 12–17 | CT>ASD | ADOS, SRS | No |
| Sutton et al., 2005 | EEG, central and parietal | 43 (23/20) | 9–14 | ASD>CT | ASSQ, ASAS, BASC | Not reported |
| Fixation time (proportional looking times) | ||||||
| Bacon et al., 2019 | GeoPref, SocPref, JA | 49 (49/0)e | 1.9f (ND) | NA | ADOS, VABS, SRS | Yes (GeoPref) |
| Shic et al., 2019 | Direct gaze, speech | 97 (50/47) | 1.96 (0.26) | CT>ASD | ADOS | Yes |
| Åsberg et al., 2017 | Dynamic emotional faces | 115 (57/58) | 10–44 | CT>ASD | ADOS, ADI-R, AQ | No |
| Swanson et al., 2013 | Congruent/incongruent gaze | 45 (21/24) | 3.58–9.41 | Not different | ADOS, SRS | Yes (high SRS) |
| Wang et al., 2018 | Dyadic bid and sandwich | 301 (112/36/163DD) | 1.86 (0.25) | CT>ASD | ADOS, ADI-R | Yes |
| Klin et al., 2002 | Eyes in dynamic social scene | 30 (15/15) | 15.1 (7.2) | CT>ASD | ADOS-social affect, VABS | Yes |
| Frazier et al., 2016 | ARI | 79 (40/39) | 3–8 | Not different | ADOS, SRS, CBCL | Yes |
| Murias et al., 2018 | Social vs. nonsocial | 25 (25/0)c,g | 2–6 | NA | ADOS, VABS, BASC, PDDBI | Yes |
| Laidi et al., 2017 | Eyes avatars | 59 (33/26) | 18–64 | Not stated | ADOS | No |
| Fractional anisotropy | ||||||
| Lin et al., 2019 | DTI | 24 (13/11) | 8.9 (1.9) | ASD>CT | ADI-R, ADOS | No |
| Gibbard et al., 2013 | DTI | 50 (25/25) | 18.8–33.3 | CT>ASD | AQ | Yes |
| Barnea-Goraly et al., 2010 | DTI | 37 (13/11) (+13Sib) | 10.5 (2) | CT>ASD | ADI-R, ADOS | No |
| Lange et al., 2010 | DTI | 60 (30/30) | 7–28 | Not different | SRS | No |
| Functional activation (after biological motion) | ||||||
| Moessnang et al., 2020 | Whole-brain, fMRI | 394 (205/189) | 6–30 | Not different | SRS | Yes |
| Kaiser et al., 2010 | Whole-brain, fMRI | 62 (25/17) (+20Sib) | 4–17 | CT>ASD | SRS | Yes |
| Björnsdotter et al., 2016 | Whole-brain, fMRI | 114 (59/55) | 4.0–17.7 | CT>ASD | SRS, VABS, ADOS | Yes |
| Functional connectivity (resting state) | ||||||
| Jung et al., 2014 | DMN, fMRI | 40 (19/21) | 16–40 | CT>ASD | AQ | Yes |
| Neufeld et al., 2018 | DMN, fMRI | 150 (29/121)h | 8–23 | Not different | SRS-2 | Yes |
| Yerys et al., 2015 | DMN, fMRI | 44 (22/22) | 8–13 | CT>ASD (post DMN) | ADOS | Yes |
| Jann et al., 2015 | DMN, fMRI | 39 (17/22) | 13.8 (2.0) | CT>ASD (post DMN) | ADOS, SRS | Yes |
| Respiratory sinus arrhythmia | ||||||
| Van Hecke et al., 2009 | Cardiorespiratory | 33 (19/14) | 8–12 | CT>ASD | ADOS, SRS | Yes |
| Patriquin et al., 2011 | Cardiorespiratory | 23 (23/0)c | 4–7 | NA | SICS, SRS, SSP | Yes |
| Klusek et al., 2013 | Cardiorespiratory | 107 (40/28/39FXS) | 4–15 | Not different | ADOS, CASL-PJ, PRS-SA | No |
| Guy et al., 2014 | Cardiorespiratory | 36 (14/22) | 14.2 (3.7) | CT>ASD | ADOS, BRIEF, SRS, VABS | Yes |
TABLE 1. Most cited biomarkers: noninterventional studiesa
| Biomarker and Referenceb | Modality | Study Design | Intervention | Sample Size (drug/placebo) | Age (years) (range, or mean and SD of ASD group) | Biomarker Outcome | ASD Behavior Measures | Behavioral Improvement | Biomarker×Behavior Correlationc |
|---|---|---|---|---|---|---|---|---|---|
| Glutathione | |||||||||
| Bertoglio et al., 2010 | Plasma | RDBPC, crossover | Methyl B12 | 30/0 | 3–8 | Not different | AbBC, CARS, CBCL, PIA-CV | No | Not tested |
| Al-Ayadhi et al., 2013 | Plasma | RDBPC | Camel milk | 49/11 | 2–12 | Post>pre | CARS, WSQ | Yes | Not tested |
| Faber et al., 2015 | Plasma | Open-label | Cleanroom | 10/0 | 3–12 | Not stated | ATEC, CARS, GARS-2, PDDBI | No | Not tested |
| Wink et al., 2016 | Plasma | RDBPC | N-acetylcysteine | 16/15 | 4–12 | Post>pre | SRS, VABS | No | Not tested |
| IL-6 | |||||||||
| Tsilioni et al., 2015 | Serum | Open-label | Diet | 40/0 | 4–10 | Pre>post | VABS | Yes | Yes |
| Melamed et al., 2018 | Blood | Open-label | Immunoglobulin, intravenous | 17/0 | 4–14 | Not different | ADOS, SRS | Yes | Not tested |
| BDNF | |||||||||
| Makkonen et al., 2011 | CSF | Open-label | Fluoxetine | 13/0 | 5–16 | Pre>post | ATEC | Yes | Not tested |
| Pardo et al., 2013 | CSF, plasma | Open-label | Minocycline | 11/0 | 3–5 | Pre>post | CGI-anchored, VABS | No | Not tested |
| Hellings et al., 2015 | Serum | Open-label | Loxapine | 16/0 | 13–65 | Post>pre | RBS-R | Yes | Not tested |
| Serotonin | |||||||||
| Bent et al., 2018 | Urine | Open-label | Sulforaphane | 21/0 | 5–22 | Not stated | AbBC, SRS | Yes | Yes |
| GABA | |||||||||
| Connery et al., 2018 | Plasma | Open-label | Immunoglobulin, intravenous | 31/0 | 9.7 (4.4) | Not different | AbBC, SRS | Yes | Not tested |
| Oxytocin | |||||||||
| Andari et al., 2010 | Plasma | RDBPC, crossover | Oxytocin, intranasal | 33/0 | 17–39 | Post>pre | Social behavior | Yes | Not tested |
| Parker et al., 2017 | Plasma | RDBPC | Oxytocin, intranasal | 32/0 | 6–12 | Not stated | SRS | Yes | Yes |
| Alpha power | |||||||||
| Kang et al., 2019 | EEG (resting state) | Open-label | rTMS treatment | 16/16 | 7.8 (2.1) | Post>pre | AuBC | Yes | Not tested |
| Murias et al., 2018 | EEG (social vs. nonsocial) | Open-label | Autologous umbilical cord blood | 25/0 | 2–6 | Post>pre | VABS | Yes | Yes |
| Fixation time (proportional looking times) | |||||||||
| Bradshaw et al., 2019 | Eyes, mouth, face; social vs. nonsocial | Pilot, randomized | PRISM | 12/11 | 1.5–4 | Not different | ADOS, VABS | No | No |
| Umbricht et al., 2017 | Social vs. nonsocial | RDBPC, crossover | V1a receptor antagonist, intravenous | 19/0 | 18–45 | Post>pre (social) | ASR, RMET, SCIT, AbBC reduced | Yes | Not tested |
| Andari et al., 2010 | Mouth, eyes, face | RDBPC, crossover | Oxytocin, intranasal | 20/0 | 17–39 | Post>pre | Social behavior | Yes | Yes |
| Fractional anisotropy | |||||||||
| Carpenter et al., 2019 | DTI | Open-label | Autologous umbilical cord blood | 19/0 | 2–6 | Post>pre | VABS | Yes | Yes |
| Functional activation (after biological motion) | |||||||||
| Ventola et al., 2015 | Whole brain | Other CT | PRT | 15/0 | 4.5–7.7 | Post>pre (in 1 of 2 groups) | ADOS, SRS-2 | Yes | Yes |
| Yang et al., 2016 | Whole brain | Open-label | PRT | 20/0 | 5.90 (1.07) | Not stated | SRS | Yes | Yes |
| Yang et al., 2017 | Whole brain | Pilot | VR-SCT | 17/0 | 18–31 | Not stated | ACS-SP, SAT | Yes | Yes |
TABLE 2. Most cited biomarkers: interventional studiesa
Data Analysis and Statistical Methods
Power calculations (Figure 2) were performed using the TTestIndPower function in the Python “statsmodels” library to perform a two-sided t test with alpha values of 0.05 (nominal) and 5.3×10–5 (after Bonferroni correction for 940 biomarkers). Biomarkers reported in more than one study are displayed as co-publication networks using Cytoscape with the default “ForceDirected” layout. For response biomarkers with consistent directions of effect across multiple studies (i.e., glutathione), Cohen’s d was estimated from the t statistic (using the t2d function in the Python “psych” library) or mean and standard deviations (see Methods in the online supplement) and converted to Hedges’ g* with 95% confidence intervals (see Methods and Table S7 in the online supplement). Hedges’ g* values were represented alongside 95% confidence intervals and sample size to provide insight into potential sample size biases (Figure 3D and 3E). Given the small number of studies and their heterogeneous designs, the data were not subjected to meta-analysis or statistical assessment of heterogeneity, robustness, or bias.
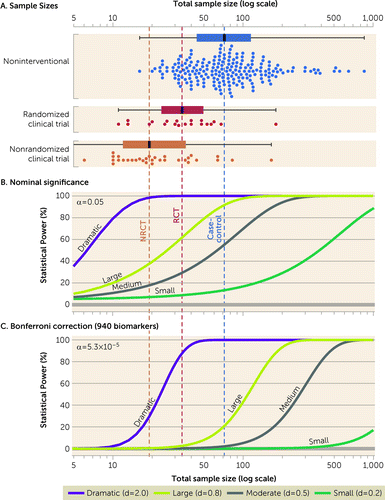
FIGURE 2. Sample sizes of biomarker studies alongside statistical power calculationsa
aIn panel A, total sample size (x-axis, logarithmic scale) was calculated by adding all cases and controls in each study. Studies are divided by design (see Figure 1), with a box plot showing the median sample size (thick vertical black line and vertical dashed lines), the interquartile range (filled box), and data points within 1.5 interquartile ranges (whiskers). Each point of the swarm plot below the corresponding box plot shows the total sample size of one of the 259 papers reviewed that included human subjects. In panel B, the curves show the statistical power against total sample size using a two-sided t test with equal numbers of cases and controls (ratio=1) at nominal significance (an alpha of 0.05) for four effect sizes: dramatic (Cohen’s d of 2), large (Cohen’s d of 0.8), moderate (Cohen’s d of 0.5), and small (Cohen’s d of 0.2). In panel C, the analysis in panel B is repeated, correcting for all 940 biomarkers with the Bonferroni method (an alpha of 5.3×10–5). RCT=randomized clinical trial; NRCT=nonrandomized clinical trial.
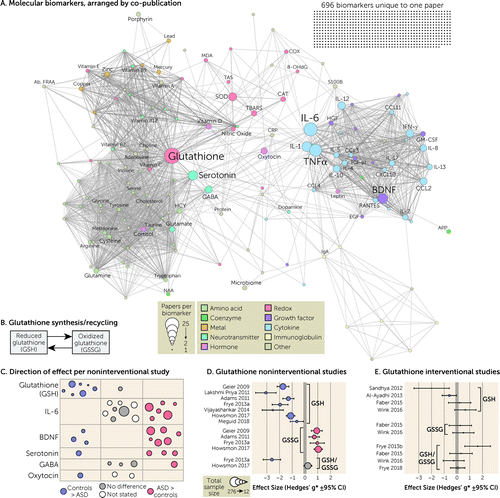
FIGURE 3. Molecular biomarkersa
aIn panel A, the 846 unique molecular biomarkers are represented as nodes (circles) with sizes corresponding to the number of articles in which they are assayed and colors representing functional categories. Edges (lines) represent co-analysis of the two biomarkers in the same article, and the network is displayed with a force-directed layout. As shown in panel B, when reduced glutathione (GSH) is oxidized, it forms a dimer called oxidized glutathione (GSSG). In panel C, the direction of effect (color and location) and total sample size (circle size) are shown for six frequently assayed biomarkers in noninterventional studies; each circle represents one study (see Table 1). Panel D shows the effect size and 95% confidence interval for the difference in glutathione between ASD cases and controls; a positive effect size represents a higher concentration in cases. Panel E shows the effect size and 95% confidence interval for the difference in glutathione between treated ASD and untreated ASD cases; a positive effect size represents a higher concentration in those receiving the treatment. Hedges’ g* is similar to Cohen’s d, using the same scale but with a correction for small sample sizes (see Methods in the online supplement). Full citations for the studies listed in panels D and E are included in Tables S4 and S2, respectively, in the online supplement. Ab, FRAA=folate receptor–alpha autoantibody; APP=amyloid precursor protein; ASD=autism spectrum disorder; BDNF=brain-derived neurotrophic factor; CAT=catalase; CCL11=C-C motif chemokine ligand 11 (eotaxin); CCL2=C-C motif chemokine ligand 2 (MCP-1); CCL3=C-C motif chemokine ligand 3 (MIP-1α); CCL4=C-C motif chemokine ligand 4 (MIP-1-beta); CCL5=C-C motif chemokine ligand 5 (RANTES); CRP=C-reactive protein; CXCL10=C-X-C motif chemokine ligand 10 (IP-10); EGF=epidermal growth factor; GABA=γ-aminobutyric acid; GM-CSF=granulocyte-macrophage colony stimulating factor (CSF2); GSH=glutathione; GSSG=oxidized glutathione; HCY=homocysteine; HGF=hepatocyte growth factor; IFN-γ=interferon gamma; IL=interleukin; MDA=malondialdehyde; NAA=N-acetylaspartate; SOD=superoxide dismutase; TBARS=thiobarbituric acid reactive substances; TGF-β1=transforming growth factor beta 1; TNFα=tumor necrosis factor–alpha; TOS=total oxidant stress.
Results
Our final review included 280 articles (see Table S1 in the online supplement), of which 53 were human studies that included an intervention, 206 were human studies without an intervention, and 21 were interventional animal studies (Figure 1E and 1F). The 53 human interventional studies included 20 randomized and 33 nonrandomized clinical trials, most of which were open-label trials. The noninterventional studies were mostly case-control design (189/206), and the remaining 17 included longitudinal and cross-sectional cohorts. Across all 280 studies, we divided the biomarkers into three groups (Figure 1E and 1F; see also Table S5 in the online supplement): molecular (e.g., glutathione, serotonin); neuroimaging and neurophysiological (e.g., functional MRI, EEG, eye tracking); and other modalities (e.g., heart rate). In total, we identified 940 unique biomarkers across all included studies, of which 846 were molecular, 82 were neuroimaging or neurophysiological, and 12 were other modalities (see Tables S5 and S6 in the online supplement).
Sample Sizes and Multiple Comparisons
To provide insight into the level of evidence provided by the biomarker literature reviewed, we considered the statistical power at varying levels of biomarker effect size alongside the total sample size in the human studies reviewed (Figure 2). The power calculation was initially performed with a two-sided t test at nominal significance (i.e., an alpha of 0.05) (Figure 2B). Candidate gene studies have highlighted the perils of relying on nominal significance as an appropriate threshold when numerous groups are engaging in parallel discovery efforts (19, 20). Appropriate correction for multiple comparisons has led to replicable findings in genomics, and therefore we repeated the power calculation using the Bonferroni method to correct for all 940 biomarkers assessed (i.e., an alpha of 5.3×10–5) (see Tables S5 and S6 in the online supplement). Noninterventional studies were generally larger than clinical trials (Figure 2A). Most noninterventional and randomized clinical trials were adequately powered to identify a biomarker with a large effect size (a Cohen’s d ≥0.8) at nominal significance (Figure 2B); however, few were capable of identifying a moderate effect size (a Cohen’s d ≥0.5) or a biomarker with large effect size after correction for multiple biomarkers (Figure 2C). Nonrandomized clinical trials included the fewest samples and were only powered to identify biomarkers with dramatic effects (a Cohen’s d ≥2.0) at nominal significance (Figure 2B).
Molecular Biomarkers
The most frequently assayed group of biomarkers was molecular (Figure 3; see also Table S5 in the online supplement), the great majority of which were measured peripherally via blood, although there was considerable variation both between and within individual biomarkers. We identified 846 unique molecules across 189 articles, the majority of which (696 molecules, 82.2%) were unique to one article (Figure 3A). By considering the frequency and overlap of the remaining 150 molecules across the articles, we generated a network of molecule co-publication. Two major groups were apparent, one made up of cytokines and growth factors (e.g., interleukin-6 [IL-6], brain-derived neurotrophic factor [BDNF]) and the other a combination of amino acids, neurotransmitters (e.g., cysteine, serotonin, GABA), and hormones (e.g., vitamin D). Between these two groups were molecules relating to reduction-oxidation (redox), including glutathione, the most frequently assayed molecule. Most papers reported an association between a molecular marker and ASD diagnostic status. True biomarkers should have a consistent association with ASD symptoms, including magnitude and direction of effect, across multiple analyses, and therefore we collated the reported outcomes of the most frequently analyzed molecules across the main molecular classes (Figure 3C, Tables 1 and 2). While the direction of effect was usually reported, the magnitude of effect, summary statistics that enabled the magnitude to be calculated (e.g., mean and standard deviation), or individual-level values often were not, preventing meta- or mega-analysis.
Redox metabolism.
Glutathione was assessed in 25 of the reviewed papers (Figure 3). All measures were peripheral, and 14 included sufficient details to quantify the magnitude of observed effect (Figure 3D and 3E; see also Table S7 in the online supplement). A decrease in reduced glutathione, generally studied in plasma (Table 1), was reported in children diagnosed with ASD in seven of eight case-control cohorts (Figure 3C), with a median Hedges’ g* (which is equivalent to Cohen’s d with a sample size correction) of −1.77 (Figure 3D, Table 1; see also Table S7 in the online supplement). Four of these papers also reported a corresponding increase in oxidized glutathione in ASD cases (Figure 3D). The glutathione results were the most consistent in direction and magnitude of effect size of any of the biomarkers examined in depth. Interventional studies, mostly aimed at reducing oxidative stress, showed inconsistent results (Figure 3E and Table 2; see also Table S8 in the online supplement).
Cytokines.
Thirty-three papers described assays of cytokine immune signaling molecules in ASD, with IL-6 being the most frequently assayed (21 papers). All papers used peripheral measures, except one that assayed CSF. Fourteen papers compared IL-6 levels between ASD cases and controls (Table 1). Eight did not report any IL-6 association, and the remaining six reported an IL-6 increase (proinflammatory state), which was correlated with the severity of ASD symptoms (stereotypies or social impairment), especially in those with regressive ASD (21) or with gastrointestinal disorders (22). Two interventional studies, aimed at reducing systemic inflammation, reported improvements in ASD-related behaviors, one of which was accompanied by an IL-6 reduction (Table 2; see also Table S8 in the online supplement).
Growth factors.
Thirty-three papers reported on growth factors, often in tandem with cytokines; all were assayed peripherally except one that was assayed in CSF. BDNF was the most frequently assayed growth factor, with 14 papers reporting levels in serum or plasma and one in CSF. Nine case-control studies all reported association with ASD, although the direction of effect varied: an increase was associated with ASD in six, and a decrease in three. Those reporting an increase tended to be larger (mean total sample size of 120 vs. 50) and included more severe cases (Table 1). Three interventional studies also showed inconsistent direction of effect for BDNF (Table 2; see also Table S8 in the online supplement).
Neurotransmitters.
Neurotransmitters were measured in 25 papers (two in brain via spectroscopy, and the remainder all peripheral), of which 14 included serotonin and 10 included GABA (Figure 3). Three of four case-control analyses reported higher serotonin levels in ASD cases, while one showed the opposite (Table 1, Figure 3C); one interventional study in humans reported correlation with behavior (Table 2). The remaining studies, many of which were interventional studies in animal models, showed inconsistent effects on serotonin levels (see Table S8 in the online supplement). For GABA, one case-control study of plasma levels reported higher levels in children with ASD, while another did not; two brain spectroscopy analyses showed no differences. A single interventional trial reported no association between GABA levels and ASD.
Hormones.
Twenty-eight of the reviewed papers included hormonal assays, with oxytocin being the most frequently assayed. Most assays were peripheral, with the exception of two analyses of CSF (one quantifying oxytocin and vasopressin, and the other, leptin). One of three case-control papers reported an association, with oxytocin being lower in individuals with ASD (Table 1, Figure 3C). The two intervention studies conducted in humans assessed intranasal oxytocin, and while both reported some degree of improvement in social behavior, only one reported increased levels of oxytocin (Table 2) (23, 24). Two more interventional studies were conducted in animal models. Again, both showed improvement in the behavioral measures assessed, while only one reported an increase in the biomarker (see Table S8 in the online supplement).
Neurophysiological Biomarkers
The second most frequently assayed category of putative biomarkers was neurophysiological (46 articles) (Figures 1F and 4B). Within this category, the two main approaches were either EEG and magnetoencephalography (MEG), with 28 papers describing 50 unique biomarkers, or eye tracking, with 19 papers describing 21 unique biomarkers. Across EEG and MEG analyses, the most frequently assessed biomarker was alpha power (Tables 1 and 2, Figure 4), which showed inconsistent results across studies, with variable directions of effect if an association was detected. N170 has been proposed as a diagnostic biomarker in ASD (25); our review included three case-control analyses that assess correlation of N170 with severity (see Table S6 in the online supplement). Within the eye-tracking papers, fixation time was the most frequently assayed outcome. Seven case-control studies assessed the proportion of looking time, although the visual stimulus varied widely. Four of the seven reported control subjects looking longer at social behaviors, speech, or eyes, while three did not (Table 1, Figure 4). One of three interventional studies showed correlation with severity.
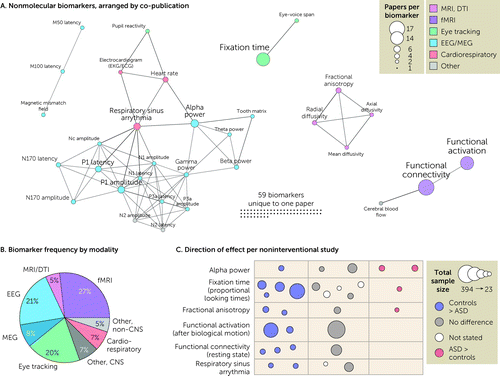
FIGURE 4. Nonmolecular biomarkersa
aIn panel A, the 94 unique nonmolecular biomarkers are represented as nodes (circles) with size corresponding to the number of articles in which they are assayed and colors representing biomarker categories. Edges (lines) represent co-analysis of the two biomarkers in the same article, and the network is displayed with a force-directed layout. Panel B shows the distribution of the total number of papers split by modality. In panel C, the direction of effect (location and color) and total sample size (circle size) are shown for the six most frequently assayed biomarkers; each circle represents one study (see Table 1). ASD=autism spectrum disorder; DTI=diffusion tensor imaging; MEG=magnetoencephalography.
Neuroimaging Biomarkers
The third most frequently assayed category was neuroimaging, with 35 articles (Figures 1F and 4B), including 25 that detailed functional MRI analyses, most of which assessed resting-state functional connectivity or task-related functional activation (Figure 4; see also Table S6 in the online supplement). Of the remaining neuroimaging articles, six described diffusion tensor imaging, with fractional anisotropy being the most common metric (Figure 4), and the direction of effect varying across the four case-control studies and one open-label trial (Tables 1 and 2, Figure 4). The remainder of the neuroimaging studies applied diverse neuroimaging techniques (see Table S6 in the online supplement).
Seventeen functional connectivity studies assessed brain networks in the resting state or, occasionally, during a task. Five studies assayed whole-brain network connectivity, with four different analytical approaches and no replicated findings. Of these, two used independent component analysis, but the connectivity patterns detected did not overlap (26, 27). Another study combined two multisite repositories to assess 654 individuals (28). This study employed a Bayesian model to identify three ASD-associated “factors,” all within the default network. Only the first factor correlated with ASD symptoms and was defined by hypoconnectivity within and between perceptual-motor networks and hyperconnectivity between perceptual-motor and association networks and between somatomotor and subcortical regions. The other 11 studies assessed specific networks; only the default mode network and the salience network were studied in more than one. All four default mode network studies (Tables 1 and 2, Figure 4) reported decreased connectivity between the medial prefrontal cortex and the posterior cingulate cortex, which was negatively correlated with social impairment in only two. The salience network studies did not show replicated patterns or symptomatic correlation.
Across the 13 functional activation studies, the tasks most frequently assessed were attention to biological motion versus scrambled motion and social psychological tasks, such as nonverbal judgments after viewing a situation or game. Whole-brain analyses were generally used for initial discovery, with specific region-task combinations followed up in subsequent targeted analyses. Task activation correlated with ASD severity most frequently in the superior temporal sulcus, parieto-temporal junction, and medial and inferior prefrontal cortices (Table 1; see also Table S6 in the online supplement). Regional activation was included in five clinical trials, conducted by two research groups, focusing on oxytocin and pivotal response treatment, respectively. Promising results from a randomized clinical trial of a single intranasal dose of oxytocin under laboratory conditions (29) were followed by improvement in Autism Diagnostic Observation Scale (ADOS) reciprocity scores following 6 weeks of treatment (30), which correlated with greater task-independent resting-state functional connectivity between the anterior cingulate cortex and dorsomedial prefrontal cortex. However, larger trials have failed to replicate improvements in ADOS score with intranasal oxytocin (31).
Physiological Biomarkers
The final category of biomarkers we identified were physiological measures outside the CNS (e.g., movement, heart rate). Eleven papers included such measures, with respiratory sinus arrhythmia (RSA), the physiological change in heart rate with breathing, being the most frequently assayed. Four papers assessed RSA, of which two reported reduced RSA in individuals with ASD (Table 1).
Discussion
Through a systematic review of quantitative biomarkers in ASD, we identified 280 papers that detailed analyses of 940 potential response biomarkers. The majority of papers reported an association between a biomarker and ASD, yet no biomarkers have been qualified by the CDER BQP. Furthermore, biomarkers assessed multiple times mostly reveal both inconsistent evidence of ASD association and variable direction of effect (Figures 3 and 4, Tables 1 and 2). These discrepancies suggest a replication crisis, as observed in other fields of biomedical research (32, 33). Our review identifies small sample sizes (Figure 2), inadequate correction for multiple comparisons (Figure 2), and the absence of replication cohorts as contributory factors. Given the high degree of positive findings despite minimal replication, it is likely that there is also a substantial publication bias, although this was not assessed statistically due to the limited reporting of quantitative outcomes in many analyses.
Against this background, distinguishing biomarkers that show true association with ASD symptoms is challenging. Mega-analysis of individual-level biomarker values would facilitate clear comparisons across biomarkers and assessment of biomarker-wide significant results; however, few studies include individual-level data. Similarly, meta-analysis, based on summary statistics, would also allow consistent comparisons across the field; surprisingly, many studies did not include these metrics. Furthermore, the heterogeneity of study design (e.g., phenotypes, demographic characteristics, methods) complicates comparison across multiple biomarkers. Well-known developmental changes are rarely taken into account in study designs (30). Replication should distinguish true positive associations, focusing on the most frequently assayed biomarkers in each class; the majority do not show consistent results (Figures 3 and 4).
The most consistent results were lower levels of reduced glutathione in ASD, observed in seven of eight case-control cohorts, with corresponding changes in oxidized glutathione (34). The median effect size of −1.77 (Hedges’ g*) (Figure 3D) is substantial. This pattern could be consistent with true ASD association, or it could arise by chance from the 940 biomarkers assayed and/or publication bias. A rigorous, large-scale, preregistered analysis is needed to resolve this question; with this effect size, the results should be definitive. Glutathione is considered to be a marker of oxidative stress in ASD, a hypothesis based on early findings of increased lactic acid in some children with ASD and the high frequency of ASD in children with genetic defects in mitochondrial enzymes (35). To date, no gene related to oxidative stress has been associated with ASD through common or rare variation, suggesting that a causal role is unlikely, although it is possible that oxidative stress reflects nonspecific systemic dysfunction.
Many of the biomarkers assessed were cytokines, aiming to detect a proinflammatory state. Inflammation is a component of the maternal immune activation and lipopolysaccharide animal models that induce social impairments, and gene expression analyses of the postmortem human cortex show a consistent upregulation of coexpressed modules enriched for immune-related genes in ASD (14, 36, 37), but neither common nor rare variation associated with ASD implicates immune processes. Proinflammatory IL-6 was the most frequently studied cytokine, and most studies failed to identify an association with ASD (Figure 3), but those that did consistently reported higher IL-6 in ASD cases, often correlated with severity. In a meta-analysis of proinflammatory cytokines in ASD, interferon gamma, IL-1β, IL-6, and tumor necrosis factor–alpha reached nominal significance (38); however, these results did not survive correction for the 21 cytokines assayed, let alone the 940 identified in this review.
The growth factor BDNF and the neurotransmitter serotonin both play critical roles in neurodevelopment and neurophysiology and highlight the importance of developmental age. Both BDNF and serotonin have some evidence to support a role as diagnostic biomarkers (39–42), although physiological levels and correlation with ASD vary across development (41, 43). Hyperserotonemia was one of the first biomarkers implicated in ASD, with early studies showing that one-third of subjects with ASD showed increased blood levels (44), a finding that has been validated in a recent meta-analysis (45). Subsequent human and animal studies showed a physiological reduction in serotonin with increasing age. This age-related reduction is attenuated in autistic individuals (43, 46), so that ASD-related hyperserotonemia is most apparent in late childhood (47). While the papers we reviewed all report an association of BDNF and serotonin levels with ASD, the direction of effect varies (Table 1, Figure 3C). Prior evidence of ASD association may have been incorrect, or, alternatively, the heterogeneous ages of individuals in these cohorts may have masked or augmented the ASD-related differences.
The extensive variability of biomarkers, both within and between individuals, presents one of the biggest challenges to biomarker research. Likely sources of variability include symptom severity, comorbidities, developmental age, sex, ancestry, genetic variation, environmental conditions (e.g., diet, medications, infections), time of day, sample processing methods, tissue assayed, and experimental assay. The blood-brain barrier is expected to be a major source of variability, leading to different results if molecular biomarkers are assayed centrally or peripherally. This will vary between molecules; for example, plasma-brain correlation has been demonstrated for BDNF in rodents, but not in humans (48), while such correspondence has not been established for GABA (49) or most other biomarkers. Differences between central and peripheral assays may also occur across development, as the permeability of the blood-brain barrier varies with maturity. Within the central or peripheral compartments, the tissue assayed is also critical. For example, platelets store serotonin, so platelet-rich blood provides more accurate assays. Where possible, recording likely sources of variability enables their inclusion as covariates in statistical models—for example, correcting for population stratification based on genotypic data. For unrecognized variables, careful experimental design, including selection of cases and controls, is critical. In discovery cohorts, large sample sizes are essential to overcome this variability, while longitudinal analyses in the same individuals can help delineate the major sources of intra- and interindividual variability in validation studies. Identifying these key covariates will be critical to define the homogeneous cohorts in which a specific biomarker may augment a clinical trial.
Neuroimaging and neurophysiological studies have clear potential for detecting biomarkers, although we found that sample sizes have been modest (the median total sample size was 44; see Table S6 in the online supplement), and a myriad of techniques, instruments, tasks, brain regions, and data processing methods pose additional challenges to distinguishing true biomarkers (Figure 4). Both alpha power and fixation time, the most frequently assayed biomarkers in neurophysiological studies, showed inconsistent direction of effect across studies (Table 1, Figure 4C). As for neuroimaging, both task activation and functional connectivity studies implicated brain regions that are generally considered part of the “social brain” (50), including the medial prefrontal and temporal cortices. Activation of regions in the medial prefrontal cortex and the parieto-temporal junction replicate across different study designs and show changes that correlate with ASD symptoms in clinical trials (29, 30, 51). Functional connectivity analyses show consistent ASD association, and some correlation with ASD symptoms, for the default-mode network, including decreased connectivity between the medial prefrontal cortex and the posterior cingulate cortex (52–55).
The variability issues that embroil neuroimaging markers are compounded by variability in data acquisition methods, such as differences in hardware, image acquisition sequence parameters, and tasks, and differences in data-analytic approaches, including data postprocessing and quality control, as well as focus on various brain regions. While there is some replication in studies implicating social brain regions and the default mode network, the heterogeneity between studies prevents clear conclusions from being drawn at this time. Data repositories, such as the Autism Brain Imaging Data Exchange (ABIDE), and open-source data analysis pipelines (e.g., ABIDE imaging masks and analysis pipelines) are enabling a new generation of larger-scale neuroimaging analyses with clear correction for multiple comparisons and open data sets for replication (56). It remains to be seen whether these initial findings prove to be robustly replicated in subsequent studies and whether some of the methodological difficulties are best overcome by pooling multiple studies or designing large-scale studies with consistent methods.
Animal models enable direct measurement of brain tissue and greater control over experimental conditions. All biomarkers assessed in animals were molecular in the studies we reviewed, and many overlapped with the molecules and classes most frequently assayed in human studies (Figure 3; see also Table S8 in the online supplement). The model most frequently used in the reviewed studies was the BTBR mouse, which is defined solely on ASD-like behaviors (57). The reliance on purely behavioral phenotypes, particularly in rodents, has generally not been productive for illuminating the biology of psychiatric phenotypes, with rare exceptions (58). In ASD, particularly given the discovery of dozens of large-effect mutations that can subserve the creation of “construct-valid” animal models, reliance on the BTBR mouse model has come under increasing scrutiny and at present has questionable relevance to the human syndrome. In addition, several environmentally induced models, including maternal immune activation and exposure to valproic acid, have been studied, but it is unclear to what extent these model common etiological mechanisms of ASD in humans. After applying our review criteria (Figure 1), none of the animal models included were based on ASD-associated genes. Relaxing our exclusion criteria by discounting criteria 6 and 7 (Figure 1) identified another 53 animal studies, but even here, only a few genetic models were used (e.g., 16p11.2, MECP2, FMR1; see Table S1 in the online supplement). With CRISPR-Cas9 gene editing and biorepositories, genetic model systems are an underutilized resource in biomarker discovery, including genetic animal models and isogenic and patient-derived human cell culture models.
Limitations of the Review
While we identified over 1,000 original research articles that included putative biomarkers in ASD, we focused on about one-quarter of these—those that were most relevant to the response biomarker classes defined by BEST (4) (see Methods and Table S1 in the online supplement), by selecting articles that assessed whether biomarkers correlate with ASD symptom intensity. We note that biomarkers can overlap between classes; for example, a diagnostic biomarker that distinguished individuals with high or low ASD liability might also change in relation to a behavior of interest. However, since no ASD biomarker in any class has been qualified by CDER BQP, and since assessing correlation with symptom severity is a logical follow-up analysis for promising diagnostic biomarkers, it is unlikely that in-depth analysis of the other 750 articles would change our main conclusions. We note that we did not systematically assess whether these conclusions generalize to other biomarker classes.
Our search is also limited by the accuracy and sensitivity of ASD severity measures, especially in the older studies. We cast a wide net, including instruments the authors of each study utilized as a measure of autistic severity, unless the measure was solely assessing global functioning or disability. Consequently, some of the measures included are confounded by behavioral or intellectual impairment. Severity measures and validation efforts continue to improve (59); it remains to be seen whether currently available metrics can detect the modest short-term changes that are likely to be necessary for evaluating biomarkers or therapeutic response.
Our most in-depth analysis focused on 12 biomarkers assayed across multiple papers (Table 1), allowing us to assess replication in order to distinguish true biomarkers. An alternative strategy would be to rank all biomarkers assayed by effect size or p value to find the most promising. However, we found the analytic methods and summary statistics reported to be too heterogeneous and incomplete for this approach; it is possible that true biomarkers are included among those studied but do not currently stand out from the crowd (see Table S4 and S5 in the online supplement).
Finally, we note several items missing from the PRISMA checklist, specifically that the review and protocol were not registered in advance, that we included 32 articles identified from citations but not the initial search, and that formal analyses of risk of bias, robustness, and heterogeneity were not performed, given the heterogeneity of the studies reviewed.
Future Studies
The ASD biomarker field is reminiscent of the era of candidate gene discovery, in which technological and biobank limitations necessitated a focus on small numbers of loci in small cohorts, which in turn led to a replication crisis (19, 20). Many of the lessons learned from candidate gene approaches are transferable to biomarkers, including the need for larger sample sizes, appropriate multiple comparisons (Figure 2), and replication cohorts. Correcting statistical significance thresholds to reflect all biomarkers assayed in a study is a bare minimum. A higher statistical threshold is required to overcome publication bias. Ideally, this threshold would be based on the total number of effective tests across all biomarkers, estimated from the degree of interdependence between biomarkers (60). Until such estimates can be made, family-wise error correction (e.g., Bonferroni) for all biomarkers tested to date, currently about 1,000, is a simple and conservative approach. Alternatively, widespread sharing of individual-level data and key metrics (e.g., tissue, collection conditions, assay, demographic characteristics, deep phenotyping) would allow false discovery rates to be estimated in multi-biomarker mega-analyses. The widespread data sharing required would be simplified by the adoption of community-wide standards, for example, using standardized ontologies and machine-readable file formats to share biomarker results and key metrics. Once true biomarkers are identified for specific subgroups of individuals with ASD, specific developmental stages, or specific symptoms, their usefulness for monitoring change due to specific interventions could be tested in clinical trials. Only when the evidence for the biomarker is comparable to the confidence in existing behavioral measures of ASD severity will biomarkers become a useful outcome measure for interventional studies.
Based on our review, we provide a list of recommendations to help identify and distinguish true ASD biomarkers (Box 1). Many of these recommendations are already being applied in some recent neuroimaging studies and through the CDER BQP process initiated by the Autism Biomarkers Consortium for Clinical Trials. Advances in genetics, such Mendelian randomization (61) and CRISPR-derived model systems, and in technology, such as proteomics and metabolomics, have the potential to greatly accelerate the hunt for biomarkers. Although ASD biomarkers remain elusive, there is immense potential if community-wide efforts can be paired with rigorous scientific methodology.
BOX 1. Recommendations for ASD biomarker research
Individual biomarker values should be shared for each sample to enable mega-analyses, ideally in standardized machine-readable formats.
Key demographic, deep phenotypic, and experimental data should be shared for each sample, including age, sex, self-reported and/or genotypic ancestry, medications, comorbid conditions (including intellectual disability, seizures, and motor delay), and tissue assayed, time of day, and experimental assay. Sharing these details would enable them to be included as covariates and to identify more homogeneous subsets.
Biomarkers and metadata should be registered against standard schema or anatomical atlases (e.g., Chemical Entities of Biological Interest, PRotein Ontology) to facilitate comparisons and mega- or meta-analyses.
Consortia should promote consistent methodologies and larger cohorts with consistent phenotypic and genotypic data.
Case-control biomarker discovery cohorts should be adequately powered and should include an independent replication cohort.
A clear statement should be included about the approach to multiple comparisons, both in the paper and across the biomarker field (equivalent to genome-wide significance).
As is standard practice in human interventional trials, noninterventional biomarker studies should be preregistered to define the research plan prior to data collection.
Once suitable biomarkers are identified, cohorts selected for clinical trials should be tailored to both the biomarker and the intervention.
1. : Diagnostic and Statistical Manual of Mental Disorders, 5th ed. Washington, DC, American Psychiatric Association, 2013Crossref, Google Scholar
2. : Prevalence of autism spectrum disorder among children aged 8 years: autism and developmental disabilities monitoring network, 11 sites, United States, 2016. MMWR Surveill Summ 2020; 69:1–12Crossref, Google Scholar
3. : What are biomarkers? Curr Opin HIV AIDS 2010; 5:463–466Crossref, Medline, Google Scholar
4. FDA-NIH Biomarker Working Group: BEST (Biomarkers, EndpointS, and other Tools) resource, 2016 https://www.ncbi.nlm.nih.gov/books/NBK326791/Google Scholar
5. : Biomarker definitions and their applications. Exp Biol Med (Maywood) 2018; 243:213–221Crossref, Medline, Google Scholar
6. : Large-scale exome sequencing study implicates both developmental and functional changes in the neurobiology of autism. Cell 2020; 180:568–584.e23Crossref, Medline, Google Scholar
7. : Coexpression networks implicate human midfetal deep cortical projection neurons in the pathogenesis of autism. Cell 2013; 155:997–1007Crossref, Medline, Google Scholar
8. : Autism spectrum disorder. Nat Rev Dis Primers 2020; 6:5Crossref, Medline, Google Scholar
9. : Lost in translation: traversing the complex path from genomics to therapeutics in autism spectrum disorder. Neuron 2018; 100:406–423Crossref, Medline, Google Scholar
10. : Identification of common genetic risk variants for autism spectrum disorder. Nat Genet 2019; 51:431–444Crossref, Medline, Google Scholar
11. : The Psychiatric Cell Map Initiative: a convergent systems biological approach to illuminating key molecular pathways in neuropsychiatric disorders. Cell 2018; 174:505–520Crossref, Medline, Google Scholar
12. : De novo mutations revealed by whole-exome sequencing are strongly associated with autism. Nature 2012; 485:237–441Crossref, Medline, Google Scholar
13. : Synaptic, transcriptional, and chromatin genes disrupted in autism. Nature 2014; 515:209–215Crossref, Medline, Google Scholar
14. : Integrative genomics identifies a convergent molecular subtype that links epigenomic with transcriptomic differences in autism. Nat Commun 2020; 11:4873Crossref, Medline, Google Scholar
15. : Linking autism risk genes to disruption of cortical development. Cells 2020; 9:2500Crossref, Medline, Google Scholar
16. : Most genetic risk for autism resides with common variation. Nat Genet 2014; 46:881–885Crossref, Medline, Google Scholar
17. : Diagnosing autism in neurobiological research studies. Behav Brain Res 2013; 251:113–124Crossref, Medline, Google Scholar
18. : Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Plos Med 2009; 6:e1000097Crossref, Medline, Google Scholar
19. : No evidence that schizophrenia candidate genes are more associated with schizophrenia than noncandidate genes. Biol Psychiatry 2017; 82:702–708Crossref, Medline, Google Scholar
20. : How genome-wide association studies (GWAS) made traditional candidate gene studies obsolete. Neuropsychopharmacology 2019; 44:1518–1523Crossref, Medline, Google Scholar
21. : Altered T cell responses in children with autism. Brain Behav Immun 2011; 25:840–849Crossref, Medline, Google Scholar
22. : Plasma cytokine profiling in sibling pairs discordant for autism spectrum disorder. J Neuroinflammation 2013; 10:38Crossref, Medline, Google Scholar
23. : Promoting social behavior with oxytocin in high-functioning autism spectrum disorders. Proc Natl Acad Sci U S A 2010; 107:4389–4394Crossref, Medline, Google Scholar
24. : Intranasal oxytocin treatment for social deficits and biomarkers of response in children with autism. Proc Natl Acad Sci U S A 2017; 114:8119–8124Crossref, Medline, Google Scholar
25. : Letter of Intent (LOI) Decision Letter: N170 to upright human faces. May 6, 2019. https://www.fda.gov/media/127494/download Google Scholar
26. : Altered functional connectivity in children with low-function autism spectrum disorders. Front Neurosci 2019; 13:806Crossref, Medline, Google Scholar
27. : Salience network-based classification and prediction of symptom severity in children with autism. JAMA Psychiatry 2013; 70:869–879Crossref, Medline, Google Scholar
28. : Reconciling dimensional and categorical models of autism heterogeneity: a brain connectomics and behavioral study. Biol Psychiatry 2020; 87:1071–1082Crossref, Medline, Google Scholar
29. : Mitigation of sociocommunicational deficits of autism through oxytocin-induced recovery of medial prefrontal activity: a randomized trial. JAMA Psychiatry 2014; 71:166–175Crossref, Medline, Google Scholar
30. : Clinical and neural effects of six-week administration of oxytocin on core symptoms of autism. Brain 2015; 138:3400–3412Crossref, Medline, Google Scholar
31. : Effect of intranasal oxytocin on the core social symptoms of autism spectrum disorder: a randomized clinical trial. Mol Psychiatry 2020; 25:1849–1858Crossref, Medline, Google Scholar
32. : Why most published research findings are false. Plos Med 2005; 2:e124Crossref, Medline, Google Scholar
33. : Editors’ introduction to the special section on replicability in psychological science: a crisis of confidence? Perspect Psychol Sci 2012; 7:528–530Crossref, Medline, Google Scholar
34. : The role of glutathione redox imbalance in autism spectrum disorder: a review. Free Radic Biol Med 2020; 160:149–162Crossref, Medline, Google Scholar
35. : Mitochondrial dysfunction in autism spectrum disorders: a systematic review and meta-analysis. Mol Psychiatry 2012; 17:290–314Crossref, Medline, Google Scholar
36. : Single-cell genomics identifies cell type-specific molecular changes in autism. Science 2019; 364:685–689Crossref, Medline, Google Scholar
37. : Transcriptome analysis reveals dysregulation of innate immune response genes and neuronal activity-dependent genes in autism. Nat Commun 2014; 5:5748Crossref, Medline, Google Scholar
38. : A meta-analysis of pro-inflammatory cytokines in autism spectrum disorders: effects of age, gender, and latitude. J Psychiatr Res 2019; 115:90–102Crossref, Medline, Google Scholar
39. : Meta-analysis of BDNF levels in autism. Cell Mol Neurobiol 2017; 37:949–954Crossref, Medline, Google Scholar
40. : Brain-derived neurotrophic factor levels in autism: a systematic review and meta-analysis. J Autism Dev Disord 2017; 47:1018–1029Crossref, Medline, Google Scholar
41. : Association of peripheral blood levels of brain-derived neurotrophic factor with autism spectrum disorder in children: a systematic review and meta-analysis. JAMA Pediatr 2016; 170:1079–1086Crossref, Medline, Google Scholar
42. : Peripheral brain-derived neurotrophic factor in autism spectrum disorder: a systematic review and meta-analysis. Sci Rep 2016; 6:31241Crossref, Medline, Google Scholar
43. : Significance of abnormalities in developmental trajectory and asymmetry of cortical serotonin synthesis in autism. Int J Dev Neurosci 2005; 23:171–182Crossref, Medline, Google Scholar
44. : Free serotonin in plasma: autistic children and their first-degree relatives. Biol Psychiatry 1988; 24:488–491Crossref, Medline, Google Scholar
45. : Blood serotonin levels in autism spectrum disorder: a systematic review and meta-analysis. Eur Neuropsychopharmacol 2014; 24:919–929Crossref, Medline, Google Scholar
46. : Developmental changes in brain serotonin synthesis capacity in autistic and nonautistic children. Ann Neurol 1999; 45:287–295Crossref, Medline, Google Scholar
47. : The developmental disruptions of serotonin signaling may involved in autism during early brain development. Neuroscience 2014; 267:1–10Crossref, Medline, Google Scholar
48. : Blood BDNF concentrations reflect brain-tissue BDNF levels across species. Int J Neuropsychopharmacol 2011; 14:347–353Crossref, Medline, Google Scholar
49. : Brain GABA levels across psychiatric disorders: a systematic literature review and meta-analysis of (1)H-MRS studies. Hum Brain Mapp 2016; 37:3337–3352Crossref, Medline, Google Scholar
50. : Fractionating theory of mind: a meta-analysis of functional brain imaging studies. Neurosci Biobehav Rev 2014; 42:9–34Crossref, Medline, Google Scholar
51. : Social brain activation during mentalizing in a large autism cohort: the Longitudinal European Autism Project. Mol Autism 2020; 11:17Crossref, Medline, Google Scholar
52. : Default mode network in young male adults with autism spectrum disorder: relationship with autism spectrum traits. Mol Autism 2014; 5:35Crossref, Medline, Google Scholar
53. : Altered resting perfusion and functional connectivity of default mode network in youth with autism spectrum disorder. Brain Behav 2015; 5:e00358Crossref, Medline, Google Scholar
54. : Alterations in resting state connectivity along the autism trait continuum: a twin study. Mol Psychiatry 2018; 23:1659–1665Crossref, Medline, Google Scholar
55. : Default mode network segregation and social deficits in autism spectrum disorder: evidence from non-medicated children. Neuroimage Clin 2015; 9:223–232Crossref, Medline, Google Scholar
56. : Decreased interhemispheric functional connectivity rather than corpus callosum volume as a potential biomarker for autism spectrum disorder. Cortex 2019; 119:258–266Crossref, Medline, Google Scholar
57. : Behavioral and neuroanatomical phenotypes in mouse models of autism. Neurotherapeutics 2015; 12:521–533Crossref, Medline, Google Scholar
58. : Steroid hormone fluctuations and GABA(A)R plasticity. Psychoneuroendocrinology 2009; 34(suppl 1):S84–S90Crossref, Medline, Google Scholar
59. : Brief report: The ADOS calibrated severity score best measures autism diagnostic symptom severity in pre-school children. J Autism Dev Disord 2019; 49:2999–3006Crossref, Medline, Google Scholar
60. : Estimation of significance thresholds for genomewide association scans. Genet Epidemiol 2008; 32:227–234Crossref, Medline, Google Scholar
61. : Genetic drug target validation using Mendelian randomisation. Nat Commun 2020; 11:3255Crossref, Medline, Google Scholar



