A Novel Psychosocial Intervention for Motivational Negative Symptoms in Schizophrenia: Combined Motivational Interviewing and CBT
Abstract
Objective:
Negative symptoms are a primary cause of disability in schizophrenia for which there are no established pharmacotherapies. This study evaluated a novel psychosocial intervention that combined two evidence-based practices—motivational interviewing and cognitive-behavioral therapy (MI-CBT)—for the treatment of motivational negative symptoms.
Methods:
Seventy-nine participants with schizophrenia and moderate to severe negative symptoms were included in a randomized controlled trial comparing the 12-session MI-CBT treatment with a mindfulness control condition. Participants were assessed at three time points through the study period, which included 12 weeks of active treatment and 12 weeks of follow-up. The primary outcome measures were motivational negative symptoms and community functioning; the secondary outcomes included a posited biomarker of negative symptoms: pupillometric response to cognitive effort.
Results:
Compared with the control group, participants in the MI-CBT group showed significantly greater improvements in motivational negative symptoms over the acute treatment period. Their gains relative to baseline were maintained at follow-up, although the differential benefit relative to control subjects was attenuated. There were nonsignificant effects toward improvements in community functioning and differential change in the pupillometric markers of cognitive effort.
Conclusions:
The results show that combining motivational interviewing with CBT yields improvements in negative symptoms, a feature of schizophrenia generally thought of as resistant to intervention. Motivational negative symptoms not only responded to the novel treatment, but the gains were maintained over the follow-up period. Implications for future studies and for improving the generalization of the negative symptom gains to daily functioning domains are discussed.
Schizophrenia is associated with a high rate of disability, and it is one of the most expensive per-patient psychiatric conditions (1). While there have been advances in pharmacological interventions for positive symptoms, they have had a minimal impact on daily functional outcomes for those living with the illness. Motivational factors, commonly conceptualized and measured as negative symptoms, are a primary determinant of functional disability in schizophrenia (2, 3).
Negative symptoms generally refer to the absence or diminution of normal functions such as emotional expression and goal-oriented activities. An important distinction exists between expressive negative symptoms, that is, reduced display of affect and speech, and motivational negative symptoms, that is, lowered volition, drive, and capacity for pleasure (4). The motivational negative symptoms have the greatest impact on outcomes and are consistently predictive of poorer future social and occupational outcomes (2, 3). Despite their importance in predicting outcomes, negative symptoms are less frequently a target for psychopharmacological studies and are more difficult to treat than positive symptoms. Apart from pharmacological studies, recent research indicates that psychosocial treatments may have promise for motivational deficits.
Cognitive-behavioral therapy (CBT) is a structured, action-oriented approach to changing maladaptive thoughts and problematic behaviors that interfere with functioning. According to a cognitive-behavioral model of schizophrenia, motivational negative symptoms are influenced by two component processes: defeatist beliefs (e.g., “Why bother trying if I won’t be perfect”) and avoidant behavior (e.g., social withdrawal) (2, 6, 7). Preliminary studies support the efficacy of CBT for negative symptoms (e.g., 6, 8–10); however, these interventions are typically time and resource intensive. Moreover, a potential barrier to delivering therapies such as CBT is that individuals may have low motivation to engage with and adhere to treatment. Recognition of the importance of motivation for effective CBT has led to integrating motivational interviewing and CBT (MI-CBT), which has shown positive results for a variety of disorders, including substance use, depression, anxiety, and physical health–related behaviors (12). Motivational interviewing is a well-established, evidence-based approach, effective for initiating and maintaining behavioral changes across a range of clinical populations (5). It is an ideal companion to systemized therapies such as CBT because it provides an effective framework from which a clinician can work to augment intrinsic motivation and encourage the client to view the treatment benefits as worth the “cost.” In serious mental illness, motivational interviewing has been used in brief applications that precede other interventions to enhance treatment engagement (13) and improve outcomes for targets, including substance misuse and psychiatric symptoms (14–16). However, integrated MI-CBT has not, to our knowledge, been tested as an approach for treating motivational negative symptoms or improving functioning in schizophrenia.
Beyond treatment of negative symptoms, a secondary goal is to identify accurate and sensitive tools to measure motivation deficits in psychiatric research. Neurophysiological indicators may provide objective clinical trial endpoints to facilitate treatment development. One such indicator is pupil dilation. The variation in the size of the pupil in response to psychological stimuli is a reliable index of attentional effort (17). We selected pupillometry as the preferred neurophysiological approach for this study because of the extensive pupillometric research in schizophrenia for assessing attention, cognitive control, and, more recently, task-based motivation (e.g., 18–22). A notable gap in this literature is that pupil size has not been evaluated in relation to motivational negative symptoms in a randomized controlled trial for schizophrenia.
We report here a controlled study of a novel group-based psychosocial intervention for the motivational negative symptoms of schizophrenia. The primary outcomes consisted of one proximal measure, motivational negative symptoms, and one more distal measure, community functioning. We expected significantly greater gains in the treatment condition during the 12-week acute intervention period compared with the active control. We also examined a physiological secondary outcome measure: pupillary response, as a biological marker of motivated effort on a cognitive task. Expressive negative, depressive, positive, and agitation symptoms were examined to evaluate specificity of effects. Durability was assessed at 12 weeks posttreatment.
Methods
Participants
Ninety-nine participants were recruited from outpatient clinics at the Veterans Affairs Greater Los Angeles Healthcare System. Selection criteria included DSM-5 (23) diagnosis of schizophrenia; age 18–65 years; no clinically significant neurological disease; no history of serious head injury; no evidence of moderate or greater current substance use disorder; no history of intellectual or developmental disability; clinical stability (i.e., no inpatient hospitalizations during the 3 months prior to enrollment, not meeting criteria for a major depressive episode in the 8 weeks prior to enrollment, and no changes in antipsychotic medication type in the 4 weeks prior to enrollment); and moderate to severe levels of motivational negative symptoms (defined as a score ≥15 out of 36 on the motivation and pleasure subscale of the Clinical Assessment Interview for Negative Symptoms [CAINS MAP] [4, 24] in the 4 weeks preceding study entry). Baseline data from this sample have been reported previously (21, 22).
Design
This study was approved by the Institutional Review Board at the Veterans Affairs Greater Los Angeles Healthcare System. Written informed consent was obtained from all participants after they were provided a complete description of study procedures. Of the 99 potential participants who gave initial consent, 79 individuals with schizophrenia and moderate to severe negative symptoms met inclusion criteria and agreed to participate in the trial (Figure 1). Participants were randomized in cohorts to either the treatment or active control; cohort sizes were allowed to range from five to eight to allow good group dynamics while minimizing delays in treatment initiation, and a block procedure was used to maintain balance of the arms throughout the study. Participants were assessed at baseline, at end of treatment (12 weeks), and at end of follow-up (24 weeks). The diagnostic interview and cognitive assessment were performed only at baseline; all three assessment points included pupillometry and measures of clinical symptoms and community functioning. Assessors were blind to treatment condition.
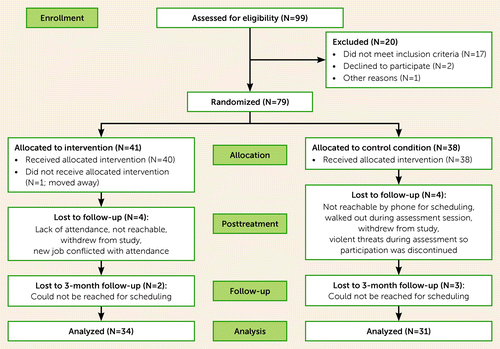
FIGURE 1. CONSORT flowchart for a controlled trial of combined motivational interviewing and cognitive-behavioral therapy
Treatment and Control Procedures
Both groups received 12 1-hour weekly group treatment sessions. The treatment and control conditions were matched for location, format, duration, peer interaction, didactic instruction, worksheets, and homework. All sessions were led by two cofacilitators. Participants received compensation after each session and each assessment ($15/hour).
MI-CBT condition.
The treatment manual was adapted from published motivational interviewing and CBT texts, as well as the dual-diagnosis motivational interviewing manual (25), which was designed for use in people with substance use and psychotic disorders. The sessions integrate a motivational interviewing approach focused on building motivation for goals, establishing a commitment to change, and resolving ambivalence with CBT-based behavioral activation, problem solving, and cognitive restructuring exercises. Each session followed the same structure: 1) weekly check-in: review of homework to share past-week progress and identify behavioral and cognitive facilitators and impediments to successful completion; 2) skills training: role-playing and problem-solving exercises to address behavioral challenges, and, if applicable, cognitive restructuring exercises to identify and modify unhelpful cognitions (e.g., defeatist performance beliefs); and 3) goal setting: collaborative development of an individualized homework assignment of behavioral steps for the upcoming week. See Table S1 in the online supplement for a detailed description of the sessions.
Control condition.
An adapted mindfulness-based stress reduction protocol was utilized as an active control. Session content included short videos, didactics, and experiential practice. Participants employed a variety of types of mindfulness in sessions that followed the same format as the treatment condition: 1) weekly check-in: review homework to share daily progress with practicing mindfulness; 2) skills training: introduction of new mindfulness techniques and group practice, using recorded audio or scripts read by facilitators; and 3) goal setting: collaborative development of individualized homework assignments of mindfulness techniques to practice in various settings throughout the week. Participants in this condition completed weekly mindfulness logs to record daily practice.
Treatment Quality Control and Fidelity Monitoring
The study principal investigator (PI) (L.F.R.) was the primary facilitator for 88% of the treatment and control cohorts. Facilitators, including the PI, did not conduct assessments, which were performed blind to condition. For the two cohorts not run by the PI (one treatment and one control), the trained doctoral-level cofacilitators met with the PI weekly for supervision. Two sessions of each cohort were observed and assessed for fidelity by a senior clinical psychologist (S.G.) for those led by the PI, or by the PI for the other two; session leaders were rated on adherence to specific motivational interviewing and CBT components, as well as general group facilitation skills. The ratings were uniformly high for both conditions (>90 out of 100) across all 16 cohorts, indicating excellent mastery of the therapeutic techniques and fidelity to the protocols.
Assessments
Symptoms
Clinical symptoms were evaluated at all three time points using the Clinical Assessment Interview for Negative Symptoms (CAINS) (4) and the expanded UCLA version of the Brief Psychiatric Rating Scale (BPRS) (26). The CAINS is a 13-item instrument that yields two subscales measuring negative symptom factors: the motivation and pleasure (MAP) subscale (one of our primary outcomes), which measures motivational negative symptoms, and the expression subscale, which measures experiential negative symptoms. The BPRS is a 24-item instrument with multiple subscales. We used the CAINS expression subscale and the BPRS subscales for positive symptoms, depression, and agitation to characterize the sample and examine the specificity of treatment effects.
Cognition
The MATRICS Consensus Cognitive Battery (MCCB) (27) was used to assess baseline cognitive functioning. It includes 10 tests measuring seven domains: speed of processing, attention/vigilance, working memory, verbal memory, visual memory, reasoning and problem solving, and social cognition. Standardized t scores were computed for each of the seven domains, correcting for age and gender. The composite score was based on the average t score from each of the domains and was used to characterize the sample.
Community Functioning
The Specific Level of Functioning Scale (SLOF) (28) assessed community functioning at the three time points. The SLOF is a 43-item report of daily behavior and functioning across a range of domains; the primary outcome variable was obtained by summing the interpersonal relationships and work skills subscales.
Pupillometry
The cognitive Deck Choice Effort Task–Pupillometry Version has been described in detail elsewhere (21, 22). Briefly, participants respond rapidly to a series of yellow or blue digits by indicating either whether the number is odd or even or whether it is lesser than or greater than five (with the question being determined by the color). There are two conditions: “easy,” in which 90% of the digits are a single color, and “hard,” in which the color changes for 90% of the trials, thereby requiring frequent switching between odd/even and lesser/greater-than decisions. Participants completed nine blocks of each condition type (easy, hard). Throughout the task, pupillometry data were acquired continuously at 220 Hz via a Viewpoint head-fixed infrared eye tracker (Arrington Research, Scottsdale, Ariz.). Periods of invalid data (e.g., during eyeblinks or saccades) were identified algorithmically and replaced with interpolated data (for additional details, see reference 22). A pupil dilation ratio trajectory was calculated for each task block by dividing pupil size at each time point during the block by the mean pupil size across the 1-second period just prior to block onset (29, 30). We then calculated an overall trajectory for each task condition by taking the median pupil ratio for each time point across all blocks in that condition. Finally, we calculated the mean pupil dilation ratio by averaging the values from the median trajectories across time, excluding the period corresponding to the first digit, to yield two dependent measures indicating pupil ratios for the easy and hard conditions.
Data Analysis
Preliminary analyses examined the two groups for baseline differences in demographic characteristics, cognition, symptoms, attendance, and the outcome measures.
We then examined the efficacy and durability of treatment effects by testing for group differences in the longitudinal trajectories of the primary outcome measures using generalized linear mixed models (GLMMs) with group (MI-CBT, control) as the between-subject factor, time (0, 12, 24 weeks) as the within-subject factor, and a group-by-time interaction, along with subject-level random intercepts. GLMMs properly account for the correlations induced by repeated measurements within subjects and automatically handle missing data, producing unbiased parameter estimates as long as incomplete observations are missing at random (31). We fitted a single model to each outcome, encompassing both the acute treatment and follow-up periods, to maximize power and obtain consistent estimates across time points. Our aims correspond to particular contrasts within those models. Specifically, our primary efficacy hypotheses concern the group-by-time interactions from 0 to 12 weeks, corresponding to differential effects of MI-CBT compared with the control condition over the acute treatment period. Our secondary durability hypotheses, evaluated for outcomes showing acute treatment effects, involve two contrasts: the 0- to 24-week interaction, which, if significant, would imply a continued advantage of MI-CBT relative to the control condition at the end of the follow-up period, and the 12- to 24-week interaction, which would indicate whether there was a significant attenuation of that treatment benefit. Post hoc within-group contrasts provide estimates of the change in each treatment arm over the acute intervention and follow-up periods. Following current guidelines (32), the primary models were fitted without covariate adjustments.
The same GLMM approach was used to examine 1) the efficacy and durability of treatment for the secondary outcome measures related to pupillometry, and 2) the specificity of treatment effects by looking at tertiary outcomes, including experiential negative symptoms from the CAINS and the BPRS subscales. Finally, tolerability ratings were examined with one-way analyses of variance. Analyses for the two primary outcome variables were corrected for multiple comparisons using a Bonferroni adjustment (at an alpha of 0.025); all other analyses were considered exploratory. Effect sizes for the mixed models are reported as Cohen’s f2, which indexes explained versus unexplained variability; values of 0.02, 0.15, and 0.35 are generally considered small, medium, and large, respectively (33).
Results
Preliminary Analyses
There were no significant differences between the groups at baseline in demographic characteristics, symptoms, cognition, or the primary and secondary outcome variables (Table 1). The groups also did not differ significantly in treatment session attendance (average of about 9.7 out of 12 sessions in each arm).
| MI-CBT Group (N=41) | Control Group (N=38) | |||
|---|---|---|---|---|
| Characteristic or Measure | N | % | N | % |
| Male | 39 | 95 | 36 | 95 |
| Hispanic | 4 | 10 | 5 | 13 |
| Race | ||||
| Asian | 2 | 5 | 1 | 3 |
| Black | 23 | 56 | 20 | 53 |
| White | 13 | 32 | 14 | 37 |
| Other | 1 | 3 | 2 | 5 |
| Mean | SD | Mean | SD | |
| Age (years) | 54.8 | 8.8 | 54.1 | 8.2 |
| Education (years) | 12.7 | 1.7 | 13.1 | 1.2 |
| Sessions (0–12) | 9.3 | 3.1 | 10.0 | 2.2 |
| Homework (0–10) | 5.8 | 3.7 | 6.7 | 3.6 |
| MCCB score | 32.9 | 12.8 | 33.3 | 9.6 |
| UPSA-2 score | 36.8 | 6.6 | 36.3 | 5.7 |
TABLE 1. Demographic and attendance data for participants in a controlled trial of combined motivational interviewing and cognitive-behavioral therapya
Analyses of Treatment Efficacy and Durability
Estimated marginal means, tests of the key efficacy and durability contrasts, and corresponding effect sizes are summarized in Table 2 for all primary and secondary variables. Figure 2 shows the outcome trajectories for outcome measures that showed significant or near-significant acute treatment effects. Figure S1 in the online supplement shows the raw means for the primary outcome variable that showed significant effects.
| Estimated Marginal Means | Statistics | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | Endpoint | Follow-Up | Acute Treatment Effect (0–12 weeks) | Durability of Treatment Effect (12–24 weeks) | Durability of Treatment Effect (0–24 weeks) | ||||||||||
| Measure and Group | Mean | SE | Mean | SE | Mean | SD | F | p | f2 | F | p | f2 | F | p | f2 |
| MAP | 10.5 | 0.001 | 0.10 | 5.42 | 0.02 | 0.05 | 0.59 | 0.45 | 0.01 | ||||||
| Control | 2.11 | 0.12 | 2.05 | 0.13 | 1.75 | 0.13 | |||||||||
| MI-CBT | 2.09 | 0.11 | 1.42 | 0.13 | 1.59 | 0.13 | |||||||||
| SLOF | 1.81 | 0.072 | 0.02 | 1.26 | 0.21 | 0.01 | 0.45 | 0.65 | 0.01 | ||||||
| Control | 46.9 | 1.47 | 45.90 | 1.61 | 47.63 | 1.63 | |||||||||
| MI-CBT | 45.7 | 1.44 | 48.37 | 1.49 | 47.35 | 1.56 | |||||||||
| Pupil-easy | 5.20 | 0.025 | 0.05 | 2.57 | 0.11 | 0.03 | 0.30 | 0.59 | 0.00 | ||||||
| Control | 0.993 | 0.008 | 0.988 | 0.008 | 0.991 | 0.009 | |||||||||
| MI-CBT | 0.982 | 0.007 | 1.01 | 0.008 | 0.988 | 0.008 | |||||||||
| Pupil-hard | 3.26 | 0.074 | 0.03 | 1.02 | 0.32 | 0.01 | 0.49 | 0.49 | 0.01 | ||||||
| Control | 1.01 | 0.008 | 0.99 | 0.009 | 1.003 | 0.010 | |||||||||
| MI-CBT | 0.99 | 0.008 | 1.02 | 0.009 | 1.008 | 0.009 | |||||||||
| DAS | 0.02 | 0.882 | 0.00 | 1.07 | 0.30 | 0.01 | 1.47 | 0.23 | 0.01 | ||||||
| Control | 47.7 | 2.8 | 47.6 | 2.9 | 48.49 | 3.0 | |||||||||
| MI-CBT | 50.2 | 2.7 | 49.6 | 2.8 | 46.99 | 2.8 | |||||||||
| EXP Neg | 1.41 | 0.238 | 0.01 | 0.00 | 0.96 | 0 | 0.18 | 0.28 | 0.01 | ||||||
| Control | 1.47 | 0.16 | 1.21 | 0.17 | 1.21 | 0.18 | |||||||||
| MI-CBT | 1.51 | 0.16 | 0.97 | 0.16 | 0.98 | 0.17 | |||||||||
| BPRS | |||||||||||||||
| Positive | 1.56 | 0.215 | 0.01 | 0.01 | 0.91 | 0 | 1.22 | 0.27 | 0.01 | ||||||
| Control | 2.13 | 0.14 | 2.03 | 0.15 | 1.92 | 0.15 | |||||||||
| MI-CBT | 2.33 | 0.14 | 1.99 | 0.14 | 1.91 | 0.15 | |||||||||
| Depression | 0.14 | 0.712 | 0.001 | 2.41 | 0.12 | 0.02 | 3.8 | 0.05 | 0.03 | ||||||
| Control | 2.01 | 0.12 | 1.95 | 0.13 | 2.06 | 0.14 | |||||||||
| MI-CBT | 2.02 | 0.12 | 1.89 | 0.13 | 1.67 | 0.13 | |||||||||
| Agitation | 0.17 | 0.680 | 0.001 | 0.01 | 0.92 | 0 | 0.09 | 0.77 | 0.00 | ||||||
| Control | 1.23 | 0.07 | 1.29 | 0.08 | 1.24 | 0.08 | |||||||||
| MI-CBT | 1.33 | 0.07 | 1.35 | 0.07 | 1.30 | 0.08 | |||||||||
TABLE 2. Treatment effects for primary, secondary, and specificity measures across baseline, endpoint, and follow-up assessments in a controlled trial of combined motivational interviewing and cognitive-behavioral therapya
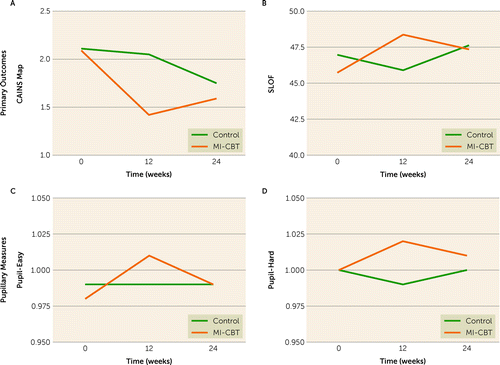
FIGURE 2. Overall mixed models for primary outcomes and pupillary measures in a controlled trial of combined motivational interviewing and cognitive-behavioral therapya
aCAINS MAP=motivation and pleasure subscale of the Clinical Assessment Instrument for Negative Symptoms; MI-CBT=motivational interviewing and cognitive-behavioral therapy; SLOF=Specific Level of Functioning Scale.
Primary Outcome Measures
On the CAINS MAP, there was an acute treatment effect (0- to 12-week interaction: F=10.50, df=1,111, p=0.0016, f2=0.10) whereby the MI-CBT group showed a significant improvement (F=26.02, df=1,111, p<0.0001, f2=0.23) but the control group did not (F=0.16, df=1,111, p=0.6855, f2=0.001). In terms of durability, at follow-up the MI-CBT group still showed substantial (albeit somewhat reduced) gains relative to baseline (F=15.02, df=1,111, p=0.0002, f2=0.14). However, the benefit of MI-CBT relative to the control condition was attenuated (12- to 24-week interaction: F=5.42, df=1,111, p=0.0217, f2=0.049; 0- to 24-week interaction: F=0.59, df=1,111, p=0.445, no longer significant), as the control group was also somewhat improved by 24 weeks (F=6.6, df=1,111, p=0.0111, f2=0.06). On the SLOF, there was an acute treatment effect for the 0- to 12-week interaction favoring MI-CBT, although it fell short of significance (F=1.81, df=1,119, p=0.0722, f2=0.015). There were no other significant effects.
Secondary Outcomes and Specificity Analyses
As shown in Table 2, on the easy trials of the pupillometry task, there was a significant differential treatment effect during the acute period, such that the MI-CBT group showed an increase in average pupil dilation from baseline to posttreatment assessment, whereas the control group did not (0- to 12-week interaction: F=5.20, df=1,00, p=0.0248, f2=0.052; MI-CBT group change: F=7.70, df=1,00, p=0.0066, f2= 0.077; control group change: F=0.24, df=1,00, p=0.6274, f2=0.024). However, these effects did not persist over follow-up. On the hard trials, there were effects that fell short of significance suggesting differential change over the acute treatment period (0- to 12-week interaction: F=3.26, df=1,00, p=0.0742, f2=0.033), such that dilation diameter increased from baseline to posttreatment assessment in the treatment group but not in the control group.
To examine the specificity of the MI-CBT effect on motivational negative symptoms, we fitted parallel models for the CAINS expressive negative symptoms and BPRS positive, depression, and agitation symptoms. There was no evidence of differential treatment effects on any of these measures.
Tolerability Ratings
Participants reported uniformly high tolerability ratings (rated on scales from 0 to 10). Across groups, there were high mean ratings for how much they liked the groups (mean=9.2, SD=1.1), how enthusiastic they considered the trainers (mean=8.9, SD=1.4), how well the trainers knew the material (mean=9.4, SD=1.1), how much the treatment helped their daily lives (mean=8.6, SD=1.7), and how much the treatment helped with symptoms (mean=8.6, SD=1.8). There were no significant between-group differences.
Discussion
In this examination of a novel application of MI-CBT for motivational negative symptoms in schizophrenia, the treatment was well tolerated and feasible to administer. Participants attended the majority of sessions, completed the majority of homework assignments, and reported enjoying and benefiting from the intervention. Attrition was low and fidelity ratings were uniformly high. We found a robust treatment effect for MI-CBT that was partially maintained at follow-up for the proximal primary outcome, motivational negative symptoms. For the distal primary outcome, community functioning, we found an acute treatment effect showing improvement in the expected direction, although it did not reach statistical significance. For the pupillary biomarker measures, in one condition we found a significant interaction during the acute treatment phase, and in the other an effect that fell short of significance, suggesting that participants who received the MI-CBT intervention more effectively modulated their effort exertion on the task than those in the control condition. This study provides initial evidence, for the efficacy of motivational interviewing and CBT integrated into one cohesive treatment and applied in a group format to remediate motivational negative symptoms in schizophrenia. Our results are encouraging for psychosocial interventions aiming to improve quality of life for people with schizophrenia and indicate specific directions for future clinical research.
Our findings are consistent with recent pharmacological and neuromodulation studies showing that negative symptoms may be malleable (34, 35). Notably, this study shows significant gains from a psychosocial intervention that is highly resource efficient. Other recent psychosocial modalities have been more demanding. For example, they may require 36–52 sessions (8, 9); rely on individual rather than group format (8); require handheld digital technology (9, 11, 36); involve home visits and in vivo trainings (37); or primarily target neurophysiology without bridges to daily functioning (34, 38). The present intervention is one that can be replicated and disseminated generally without excessive staff or equipment.
Although MI-CBT was effective in improving motivational negative symptoms, the treatment effect was less substantial on functioning. There are two possible explanations for this pattern of findings. First, the theoretical model of motivational change that underlies motivational interviewing posits a stage-like cycle of increasing motivation and activating behavior change (5, 25). Our results indicate that the intervention supports participants through early stages (i.e., attitudes about the worthiness of making changes) but less so in later stages (i.e., sustained lifestyle and behavioral changes). Twelve weeks is a relatively short time to actualize significant alterations in domains like social connectedness or work status, and the improvements in these areas might have been more robust with a longer follow-up. Relatedly, the intervention may serve a catalytic effect within a critical window. That is, it is possible to have a treatment effect window during the weeks of active intervention and the months immediately following, in which participants are more likely to engage in other important intervention modalities that could facilitate enhanced functioning (e.g., social skills training, occupational interventions). Future studies should examine variable treatment length, with a focus on a longer intervention that incorporates booster sessions.
Second, the SLOF may be a suboptimal measure to evaluate gradual improvements in functioning. Daily functioning is a complex construct, and a measure that detects small improvements may be more appropriate for assessing meaningful change than one designed to assess global outcomes. Many participants reported meaningful and positive behavioral changes in their exit interviews. For instance, one participant reported decreasing from 30 to 15 cigarettes per day; another received a delayed eye surgery after setting the goal to improve their health; and another attained sobriety. One participant identified physical health and weight loss as her goal and established a support network, began exercising, and improved self-care and mood. A participant who had smoking cessation as a goal and cited friends who still smoked as a barrier established a new social support group of like-minded residents at his supported-living facility. Thus, it appears that meaningful changes in daily functioning were occurring for many participants, and a more granular assessment may detect intervention-related changes.
Pupillary response is a well-established indicator of effort allocation on cognitive tasks; thus, we explored its potential as a biomarker of clinical change in the treatment of motivational negative symptoms. The results suggest that it may be sensitive to treatment-related changes. Importantly, participants who received MI-CBT modulated their physiological effort exertion more effectively following the 12-week intervention than at baseline compared with control participants. This finding is encouraging given the dearth of meaningful biomarkers and objective indices of negative symptoms. While this is the first study, to our knowledge, to examine changes in pupillary response on an effort task over a psychosocial motivational intervention for schizophrenia, other studies have shown change in pupil response to stimulation following cognitive control training, when combined with pharmacological intervention (39).
In summary, this was a rigorously controlled treatment study using an innovative and accessible intervention, and the results support the efficacy of MI-CBT for improving motivational negative symptoms. As mentioned, study limitations include the relatively short follow-up period and a potentially insufficient measurement of community functioning. In addition, the generalizability of findings from this chronically ill, predominantly male sample of veterans is unclear. The lack of an extrapyramidal side effect scale to assess primary versus secondary negative symptoms is a potential limitation. That participants were paid for attendance in the sessions also limits generalizability of feasibility to clinical settings, and the comparison with an active intervention may underestimate the treatment effect. Evidence for the efficacy of negative symptom interventions is growing, and our results provide additional support. A critical direction for future research is to translate these gains to lasting improvements in daily functioning.
1 : The social and economic burden of treatment-resistant schizophrenia: a systematic literature review. Int Clin Psychopharmacol 2014; 29:63–76Crossref, Medline, Google Scholar
2 : From perception to functional outcome in schizophrenia: modeling the role of ability and motivation. Arch Gen Psychiatry 2012; 69:1216–1224Crossref, Medline, Google Scholar
3 : Prognostic variables at intake and long-term level of function in schizophrenia. Am J Psychiatry 2006; 163:433–441Link, Google Scholar
4 : Development and psychometric validation of the Clinical Assessment Interview for Negative Symptoms (CAINS). Schizophr Res 2011; 132:140–145Crossref, Medline, Google Scholar
5 : Motivational Interviewing: Preparing People for Change. New York, Guilford, 2002 Google Scholar
6 : Improvement in negative symptoms and functioning in cognitive-behavioral social skills training for schizophrenia: mediation by defeatist performance attitudes and asocial beliefs. Schizophr Bull 2018; 44:653–661Crossref, Medline, Google Scholar
7 : Defeatist beliefs as a mediator of cognitive impairment, negative symptoms, and functioning in schizophrenia. Schizophr Bull 2009; 35:798–806Crossref, Medline, Google Scholar
8 : Randomized trial to evaluate the efficacy of cognitive therapy for low-functioning patients with schizophrenia. Arch Gen Psychiatry 2012; 69:121–127Crossref, Medline, Google Scholar
9 : Randomized clinical trial of cognitive behavioral social skills training for schizophrenia: improvement in functioning and experiential negative symptoms. J Consult Clin Psychol 2014; 82:1173–1185Crossref, Medline, Google Scholar
10 : Six-month follow-up of recovery-oriented cognitive therapy for low-functioning individuals with schizophrenia. Psychiatr Serv 2017; 68:997–1002Link, Google Scholar
11 : Efficacy of PRIME, a mobile app intervention designed to improve motivation in young people with schizophrenia. Schizophr Bull 2018; 44:1010–1020Crossref, Medline, Google Scholar
12 : Motivational Interviewing and CBT: Combining Strategies for Maximum Effectiveness. New York, Guilford, 2017 Google Scholar
13 : Motivational interviewing to increase cognitive rehabilitation adherence in schizophrenia. Schizophr Bull 2016; 42:327–334Crossref, Medline, Google Scholar
14 : Motivational interviewing–based compliance/adherence therapy interventions to improve psychiatric symptoms of people with severe mental illness: meta-analysis. J Am Psychiatr Nurses Assoc 2019; 25:122–133Crossref, Medline, Google Scholar
15 : Randomized controlled trial of motivational interviewing, cognitive behavior therapy, and family intervention for patients with comorbid schizophrenia and substance use disorders. Am J Psychiatry 2001; 158:1706–1713Link, Google Scholar
16 : Motivational interviewing and treatment adherence among psychiatric and dually diagnosed patients. J Nerv Ment Dis 1999; 187:630–635Crossref, Medline, Google Scholar
17 : Pupillometric measures of cognitive and emotional processes. Int J Psychophysiol 2004; 52:1–6Crossref, Medline, Google Scholar
18 : Motivation modulates visual attention: evidence from pupillometry. Front Psychol 2013; 4:59Crossref, Medline, Google Scholar
19 : Oculomotor and pupillometric indices of pro- and antisaccade performance in youth-onset psychosis and attention deficit/hyperactivity disorder. Schizophr Bull 2010; 36:1167–1186Crossref, Medline, Google Scholar
20 : Pupillometric measures of attentional allocation to target and mask processing on the backward masking task in schizophrenia. Psychophysiology 2009; 46:510–520Crossref, Medline, Google Scholar
21 : Pupillary responses to a cognitive effort task in schizophrenia. Schizophr Res 2018; 199:53–57Crossref, Medline, Google Scholar
22 : Pupillary change on a cognitive effort task in schizophrenia: associations with cognition and motivation. Int J Psychophysiol 2020; 155:1–7Crossref, Medline, Google Scholar
23 : Structured Clinical Interview for DSM-5–Research Version (SCID-5 for DSM-5, Research Version; SCID-5-RV). Washington, DC, American Psychiatric Association, 2015 Google Scholar
24 : The Clinical Assessment Interview for Negative Symptoms (CAINS): final development and validation. Am J Psychiatry 2013; 170:165–172Link, Google Scholar
25 : Dual diagnosis motivational interviewing: a modification of motivational interviewing for substance-abusing patients with psychotic disorders. J Subst Abuse Treat 2002; 23:297–308Crossref, Medline, Google Scholar
26 : Brief Psychiatric Rating Scale (BPRS) expanded version: scales, anchor points, and administration manual. Int J Methods Psychiatr Res 1993; 3(App 1):227–243 Google Scholar
27 : MATRICS Consensus Cognitive Battery. Los Angeles, MATRICS Assessment, 2006 Google Scholar
28 : SLOF: a behavioral rating scale for assessing the mentally ill. Soc Work Res Abstr 1983; 19:9–21Crossref, Medline, Google Scholar
29 : Test-retest repeatability of the pupil light response to blue and red light stimuli in normal human eyes using a novel pupillometer. Front Neurol 2011; 2:10Crossref, Medline, Google Scholar
30 : The pupillary light response reveals the focus of covert visual attention. PLoS One 2013; 8:e78168Crossref, Medline, Google Scholar
31 : Multilevel Analysis: An Introduction to Basic and Advanced Multilevel Modeling, 2nd ed. London, Sage, 2012 Google Scholar
32 : A source of false findings in published research studies: adjusting for covariates. JAMA Psychiatry 2015; 72:961–962Crossref, Medline, Google Scholar
33 : Statistical Power Analysis for the Behavioral Sciences, 2nd ed. Hillsdale, NJ, Lawrence Erlbaum Associates, 1988 Google Scholar
34 : Repetitive transcranial magnetic stimulation for negative symptoms of schizophrenia: review and meta-analysis. J Clin Psychiatry 2010; 71:411–418Crossref, Medline, Google Scholar
35 : Cariprazine in the management of negative symptoms of schizophrenia: state of the art and future perspectives. Future Neurol 2020; 15:4 Crossref, Google Scholar
36 : Development of the Motivation and Skills Support (MASS) social goal attainment smartphone app for (and with) people with schizophrenia. J Behav Cogn Ther 2020; 30:23–32Crossref, Medline, Google Scholar
37 : A randomized pilot study of MOtiVation and Enhancement (MOVE) training for negative symptoms in schizophrenia. Schizophr Res 2015; 165:175–180Crossref, Medline, Google Scholar
38 : Repetitive transcranial magnetic stimulation as an adjunctive treatment for negative symptoms and cognitive impairment in patients with schizophrenia: a randomized, double-blind, sham-controlled trial. Neuropsychiatr Dis Treat 2019; 15:1141–1150Crossref, Medline, Google Scholar
39 : You gotta work at it: pupillary indices of task focus are prognostic for response to a neurocognitive intervention for rumination in depression. Clin Psychol Sci 2014; 2:455–471 Crossref, Google Scholar



