d-Cycloserine, an NMDA Glutamate Receptor Glycine Site Partial Agonist, Induces Acute Increases in Brain Glutamate Plus Glutamine and GABA Comparable to Ketamine
To the Editor: Ketamine, an N-methyl-d-aspartate glutamate receptor (NMDAR) antagonist, is effective acutely for major depression (1), but development of alternative NMDAR antagonists is limited by incomplete understanding of mechanism and optimal dosage. Based on preclinical and clinical studies (1, 2), we hypothesize that NMDAR antagonism will produce an acute increase in GABA and glutamate plus glutamine (Glx) as an early event in antidepressant action. Using proton magnetic resonance spectroscopy (1H MRS), we found acute ketamine treatment in depressed patients produced rapid, transient elevations (approximately 40%) of both Glx and GABA levels in the medial prefrontal cortex (1), suggesting that this is an initial step in the antidepressant action cascade for NMDAR antagonists. We used the same methods (1) to assess the effects of high-dose d-cycloserine (1000 mg), an NMDAR glycine site partial agonist, on brain GABA and Glx in healthy subjects. Compared with ketamine, d-cycloserine has a slower (onset within 2–4 weeks) but comparable antidepressant effect (3). d-cycloserine has a dose-dependent biphasic effect on the NMDAR, potentiating function at low dosages (<100 mg), but functioning as a net antagonist at dosages >500 mg (4).
After baseline 1H MRS scans, six healthy subjects (age=33 years, SD=4 years; four males and two females) were administered 1000 mg of oral d-cycloserine, followed by serial 1H MRS scans for up to 90 minutes to measure Glx and GABA levels. Areas under the curve computed using the trapezoidal rule yielded large effect sizes for both Glx (69%, SD=48%, d=1.44) and GABA (39%, SD=31%, d=1.26) without inducing overt psychosis (Figure 1, next page). There was an overall main effect on Glx (F5,22.8=2.9, p=0.034), with an initial peak at approximately 35 minutes postdose (increase of 23%, SD=5%) and a second peak 75–90 minutes postdose (increase of 20%, SD=7%). For GABA, there was a comparable, trend-level main effect (F5,23.3=2.5, p=0.058; increase of 16%, SD=5%).
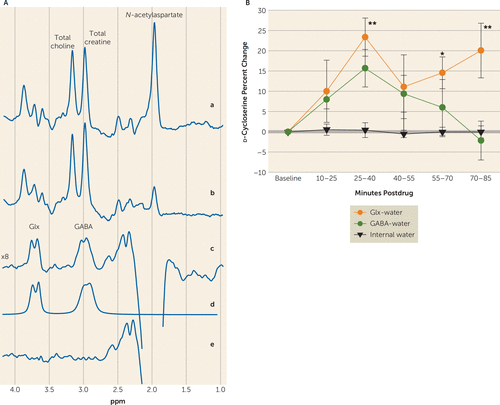
FIGURE 1. Sample In Vivo Human Brain GABA and Glutamate Plus Glutamine (Glx) Spectra, and Percentage Change of Concentrations Measured by Proton Magnetic Resonance Spectroscopy (1H MRS)a
a Panel A illustrates medial prefrontal cortex GABA and Glx detection with the J-editing 1H MRS technique. Traces in (a) and (b) represent single-voxel subspectra of the J-editing method acquired in 15 minutes, respectively, with the editing pulses off and on, and 580 total interleaved averages. The spectrum in (c) is the difference between spectra in (a) and (b) and shows the edited brain GABA and Glx resonances. Model fitting of the experimental spectrum in (c) was used to obtain the peak GABA and Glx areas as shown in the spectrum in (d). The spectrum in (e) shows the residual of the difference between spectra in (c) and (d). Panel B shows the percentage change of 1H MRS-measured GABA-water and Glx-water concentrations in healthy controls in the medial prefrontal cortex following d-cycloserine. Frame duration was 13 minutes and 20 seconds. Significant group increases, denoted by ** for p<0.01 and by * for p<0.05, relative to predrug baseline levels for Glx were observed. No clinically significant side effects, change in vital signs, or behavioral symptoms as measured by the Positive and Negative Syndrome Scale were observed. Error bars denote standard deviation from the mean. The overall significance on the mixed model was p=0.03.
d-cycloserine increased both neurotransmitter levels comparably to ketamine, and this may be the initial step in the antidepressant cascade of NMDAR antagonism. Future studies should examine the relationship of these neurotransmitters to the degree of the antidepressant effect of d-cycloserine. Targeting the NMDAR and AMPA receptors may have specific advantages in treatment-resistant depression. The more rapid clinical improvement seen with ketamine may be due to other pharmacological effects that distinguish it from d-cycloserine, and these need to be identified to facilitate development of more rapidly acting antidepressants.
1 : A pilot in vivo proton magnetic resonance spectroscopy study of amino acid neurotransmitter response to ketamine treatment of major depressive disorder. Mol Psychiatry 2015; 21:320–327Crossref, Medline, Google Scholar
2 : Bidirectional homeostatic regulation of a depression-related brain state by gamma-aminobutyric acidergic deficits and ketamine treatment. Biol Psychiatry 2016; 80:457–468Crossref, Medline, Google Scholar
3 : Single-dose ketamine followed by daily d-cycloserine in treatment-resistant bipolar depression. J Clin Psychiatry 2015; 76:737–738Crossref, Medline, Google Scholar
4 : Actions of d-cycloserine at the N-methyl-d-aspartate-associated glycine receptor site in vivo. Neuropharmacology 1991; 30:1167–1171Crossref, Medline, Google Scholar



