Pain as a Predictor of Opioid Use Disorder in a Nationally Representative Sample
Abstract
Objective:
The authors sought to ascertain the relationship between moderate and more severe pain and prescription opioid use disorders in the noninstitutionalized U.S. population.
Method:
A structural equation model was used to assess prospectively the interdependency of pain and prescription opioid use disorder at waves 1 (2001–2002) and 2 (2004–2005) of the National Epidemiologic Survey on Alcohol and Related Conditions. Pain was measured with a 5-point scale of pain-related interference in daily activities and dichotomized as “no pain” (no or little interference) or “pain” (moderate to extreme interference). Prescription opioid use disorder was assessed with a structured interview (the Alcohol Use Disorder and Associated Disabilities Interview Schedule–DSM-IV version). Other covariates included age, sex, anxiety or mood disorders, and family history of drug, alcohol, and behavioral problems.
Results:
In the structural equation model, pain and prescription opioid use disorders were significantly associated with one another at baseline and at 3-year follow-up. However, whereas pain at baseline was also significantly associated with prescription opioid use disorder at follow-up, prescription opioid use disorder at baseline was not associated with pain at follow-up. These associations were independent of several background demographic and clinical characteristics. The path for pain interference was associated with a 41% relative increase in the risk of developing a prescription opioid use disorder.
Conclusions:
Painful conditions contribute to the risk of prescription opioid use disorders. To help reduce the incidence of prescription opioid abuse and dependence among adults with moderate to severe pain, careful monitoring and consideration of nonopioid alternative treatments is warranted.
The high prevalence of chronic pain and increases in the prevalence and adverse events associated with opioid prescriptions have brought to the fore the need to examine the relationship between chronic pain and prescription opioid use disorders (1, 2). Pain is a prevalent condition, with recent estimates suggesting that chronic pain affects approximately one-third of the U.S. population and constitutes one of the most common symptoms for which patients seek medical attention (3). It is associated with intense personal suffering, high rates of disability, and an annual economic burden surpassing half a trillion dollars due to costs of medical treatment and productivity losses (4).
Concerns about undertreatment of pain have led to growth in opioid prescriptions and an increase in the prevalence of prescription opioid use disorders, which themselves pose risks of premature mortality (5, 6). Despite the clinical, public health, and policy relevance of prescription opioid use disorder, little is known about its relationship to pain. Information about these relationships bears directly on the development of guidelines and legislation to ensure the safe treatment of pain and minimize the risk of addiction to prescription opioids (7, 8). Several cross-sectional studies have indicated that pain is associated with an increased risk of prescription opioid use disorders (9, 10), and concerns have been raised that individuals with opioid use disorders may develop abnormal pain sensitivity or hyperalgesia (11). Surprisingly, however, no study has examined prospectively the relationship between pain and prescription opioid use disorders in a nationally representative sample.
We sought to address this gap in knowledge with prospective data from the National Epidemiological Survey on Alcohol and Related Conditions (NESARC), a large, nationally representative sample. We examined whether the presence of pain increased the risk of developing a prescription opioid use disorder 3 years later and, conversely, whether prescription drug use disorders increased the subsequent risk of developing pain, after adjusting for several relevant demographic and clinical covariates.
Method
Sample
The 2001–2002 NESARC (wave 1), and the 2004–2005 follow-up (wave 2), is a nationally representative survey of the noninstitutionalized adult U.S. population conducted by the U.S. Census Bureau under the direction of the National Institute on Alcoholism and Alcohol Abuse, as described elsewhere (12–14). The wave 1 response rate was 81%. Excluding ineligible respondents (e.g., deceased), the wave 2 response rate was 86.7%, resulting in a cumulative response rate of 70.2% (N=34,653). Wave 2 NESARC weights include a component that adjusts for nonresponse, demographic factors, and psychiatric diagnoses to ensure that the wave 2 sample approximated the target population, that is, the original sample minus attrition between the two waves (12).
Assessment
All diagnoses for waves 1 and 2 were made using the Alcohol Use Disorder and Associated Disabilities Interview Schedule–DSM-IV version (AUDADIS-IV) (15). Consistent with previous reports, nonmedical use of a prescription opioid was defined as using a prescription drug “without a prescription, in greater amounts, more often, or longer than prescribed, or for a reason other than a doctor said you should use them” during the 12 months preceding the interview. More than 30 symptom items are used by the AUDADIS-IV to define 12-month prescription opioid use disorders (abuse or dependence) according to DSM-IV criteria. The NESARC also collected information for other substance use disorders (nicotine dependence, alcohol use disorders, and drug use disorders, including other prescription drug use disorders). The reliability of the AUDADIS-IV prescription opioid use questions (kappa=0.66) and that of associated substance use disorder diagnoses (kappa values, 0.53–0.84) are well documented in both clinical (16) and general population (17) samples. The prescription opioid use disorder diagnosis also has established concurrent and predictive validity related to impairment and treatment seeking (18).
Mood disorders included major depressive disorder, dysthymia, bipolar I disorder, and bipolar II disorder. Anxiety disorders included panic disorder, social anxiety disorder, specific phobia, and generalized anxiety disorder. AUDADIS-IV methods to diagnose these disorders have been described in detail elsewhere (19). Test-retest reliabilities for AUDADIS-IV mood, anxiety, and personality disorders in the general population and in clinical settings were fair to good. Convergent validity was good to excellent for all mood, anxiety, and personality disorder diagnoses, showing good agreement with psychiatrist reappraisals (kappa values, 0.64–0.68) (20). As in our previous work (21), family histories of alcohol use disorders, drug use disorders, antisocial personality disorder, and depression were sought for first-degree relatives and were also assessed with the AUDADIS-IV, using readily observable manifestations of the disorders. The test-retest reliability for AUDADIS family history variables is very good to excellent (14).
Assessment of Pain
Consistent with previous national studies (4), pain was assessed using the pain item of the Medical Outcomes Study 12-item Short-Form Health Survey (SF-12) (22), a valid measure that is commonly used in population surveys and is sensitive to change (23, 24). The pain item measures the amount to which pain interfered with daily activities during the past month, on a 5-point scale (not at all, a little bit, moderately, quite a bit, and extremely) (25). The pain measure was collapsed into two levels to indicate whether pain was associated with no or little interference (“no pain”) or moderate to extreme interference (“pain”) (2). In preliminary analyses, pain interference was associated with lower scores on each of the other 11 items of the SF-12 (all p values, <0.001) and increased the likelihood of all medical conditions assessed in the NESARC (odds ratios, 2.8–7.6; all p values, <0.001). This confirmed previous analyses indicating that greater interference of the SF-12 pain item was associated with higher health care expenditures, more missed days of work, lower productivity (4), and greater probability and frequency of nonmedical use of prescription drugs, even after adjusting for multiple comorbidities (9).
Statistical Analysis
Wave 1 descriptive demographic and clinical characteristics were compared between individuals with and without pain and between individuals with and without a prescription opioid use disorder. Group differences were evaluated with chi-square or t tests.
In separate analyses, odds ratios and adjusted odds ratios, controlling for age, sex, other substance use disorders, and mood or anxiety disorders at wave 1, were then used to assess the strength of associations at wave 1 between pain severity and prescription opioid use disorder.
We built our model for the prospective relationship between pain and prescription opioid use disorder in two steps. First, a series of logistic regression models were used to examine whether pain at wave 1 was associated with the prevalence and incidence of prescription opioid use disorders at wave 2 and whether prescription drug use disorder at wave 1 was associated with the prevalence and incidence of pain at wave 2. Incidence was defined as the number of new cases reported between wave 1 and wave 2. Exploratory analyses examined whether there were any interactions between time of pain assessment (wave 1 versus wave 2) and likelihood of prescription drug use disorders at wave 2. All logistic regression models were adjusted for age, sex, other substance use disorders, and mood or anxiety disorders at wave 1, which have been shown to be associated both with pain (2–4) and with prescription opioid use disorders (26, 27). In addition, the prevalence models adjusted for current prescription opioid use disorder or level of pain at wave 1, as appropriate.
In the second step, because of the bidirectional relationship between pain and prescription opioid use disorders, we used structural equation models to model the interdependency between pain and prescription opioid use disorders at waves 1 and 2. Jointly modeling pain and prescription use disorders avoids simultaneous equation bias (28), which can result when they are modeled in separate, unlinked regressions. Because the prevalence of pain and prescription drug use disorders, along with several covariates in the model, differs by sex (3, 29), we used multiple group structural equation model analyses to examine whether the model was invariant by sex, comparing a model with all parameters free to differ across sex to one in which they were all the same. Invariance was determined by finding this chi-square difference test nonsignificant (p>0.05). Additional structural equation models were examined substituting specific criteria (tolerance and withdrawal) or an indicator of using prescription opioids longer or at greater doses than prescribed for prescription opioid use disorder at wave 2. Logistic regression models were fitted and adjusted odds ratios estimated with 95% confidence intervals using SUDAAN. The structural equation models were fitted with standardized probit regression coefficients and p values in Mplus, using weighted least squares, an estimator that is robust for nonnormality. Because the structural equation models being estimated were fully saturated (i.e., all cross-sectional and longitudinal associations between pain and prescription opioid use disorder at both waves were freely modeled such that the degrees of freedom was zero), standard structural equation model fit statistics provide no additional information. Both SUDAAN and Mplus take into account the complex design features of the NESARC.
Results
Bivariate Associations
At wave 1, the prevalence of pain was 18.76% (N=6,943) and the prevalence of prescription opioid use disorders was 0.33% (N=104). Individuals with prescription opioid use disorders were younger than those without, were more likely to be male, and were more likely to have another current substance use disorder, a mood or anxiety disorder, and a family history of alcohol use disorders, drug use disorders, depression, and antisocial personality disorder. Individuals with pain were more likely to be older than those without and were more likely to be female, but they were also more likely than those without pain to have a current substance use disorder, a mood or anxiety disorder, and a family history of alcohol use disorders, drug use disorders, depression, and antisocial personality disorder (Table 1).
| Prescription Opioid Use Disorder | Pain Interference | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Characteristic | Present (N=104) | Absent (N=34,549) | t or χ2 | p | Present (N=6,943) | Absent (N=27,522) | t or χ2 | p | ||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||||
| Age at wave 1 (years) | 35.19 | 14.41 | 45.11 | 17.33 | –6.01 | <0.001 | 51.77 | 17.70 | 43.53 | 16.88 | 23.53 | <0.001 |
| % | N | % | N | % | N | % | N | |||||
| Sex | 6.15 | 0.02 | 75.15 | <0.001 | ||||||||
| Male | 63.48 | 58 | 47.87 | 14,506 | 41.56 | 2,477 | 49.38 | 12,008 | ||||
| Female | 36.52 | 46 | 52.13 | 20,043 | 58.44 | 4,466 | 50.62 | 15,514 | ||||
| Family history | ||||||||||||
| Alcohol use disorders | 61.21 | 57 | 34.38 | 12,193 | 12.20 | <0.001 | 42.00 | 2,943 | 32.93 | 9,294 | 112.06 | <0.001 |
| Drug use disorders | 30.05 | 33 | 16.61 | 5,939 | 6.30 | 0.01 | 20.79 | 1,461 | 15.77 | 4,500 | 51.65 | <0.001 |
| Depression | 52.44 | 57 | 32.58 | 11,076 | 9.78 | 0.003 | 38.18 | 2,543 | 31.55 | 8,578 | 68.69 | <0.001 |
| Antisocial personality disorder | 34.35 | 34 | 17.87 | 6,177 | 6.61 | 0.01 | 21.86 | 1,493 | 17.12 | 4,714 | 43.78 | <0.001 |
| Any other 12-month substance use disorder | 74.53 | 76 | 18.12 | 5,912 | 33.20 | <0.001 | 21.82 | 1,385 | 17.61 | 4,600 | 36.08 | <0.001 |
| Any 12-month mood or anxiety disorder | 50.39 | 54 | 16.10 | 5,873 | 18.44 | <0.001 | 24.57 | 1,748 | 14.38 | 4,172 | 153.55 | <0.001 |
TABLE 1. Background Characteristics of NESARC Respondents, by Prescription Opioid Use Disorder and Pain Interference Status at Wave 1a
Pain was reported by 38.27% of individuals with prescription opioid use disorders and 18.69% of those without (odds ratio=2.70, 95% CI=1.66–4.39). After adjustment for the background demographic and clinical characteristics, a strong association persisted between prescription opioid use disorder and pain (adjusted odds ratio=2.38, 95% CI=1.51–3.76).
Logistic regression models indicated that pain at baseline was associated with both prevalence (odds ratio=2.05, 95% CI=1.33–3.18) and incidence (odds ratio=2.15, 95% CI=1.37–3.37) of prescription opioid use disorders, and the odds did not significantly change after adjustment for demographic and clinical covariates (adjusted odds ratio=2.17, 95% CI=1.35–3.48, and adjusted odds ratio=2.26, 95% CI=1.40–3.65, respectively). By contrast, while a prescription opioid use disorder at baseline was associated with prevalence of pain at wave 2 (odds ratio=1.84, 95% CI=1.04–3.25), it was not significantly associated with incidence (odds ratio=1.78, 95% CI=0.75–4.24), and neither association was significant after adjustment for demographic and clinical covariates (adjusted odds ratio=1.72, 95% CI=0.95–3.13, and adjusted odds ratio=1.90, 95% CI=0.76–4.70, respectively) (Table 2). There were no significant interactions between time of pain assessment and likelihood of prescription drug use disorder at wave 2 either in unadjusted models (odds ratio=0.96, 95% CI=0.39–2.38) or in models that adjusted for age, sex, other substance use disorders, or any mood or anxiety disorder at wave 1 (odds ratio=0.99, 95% CI=0.40–2.50).
| Association | Odds Ratio | 95% CI | Adjusted Odds Ratiob | 95% CI |
|---|---|---|---|---|
| Wave 1 prescription opioid use disorder predicting: | ||||
| Prevalence of wave 2 pain interference | 1.84 | 1.04–3.25 | 1.72 | 0.95–3.13 |
| Incidence of pain interference | 1.78 | 0.75–4.24 | 1.90 | 0.76–4.70 |
| Wave 1 pain interference predicting: | ||||
| Prevalence of wave 2 prescription opioid use disorder | 2.05 | 1.33–3.18 | 2.17 | 1.35–3.48 |
| Incidence of prescription opioid use disorder | 2.15 | 1.37–3.37 | 2.26 | 1.40–3.65 |
TABLE 2. Prospective Associations of the Prevalence and Incidence of Pain Interference and Prescription Opioid Use Disorders in the NESARCa
Structural Equation Model
In the model, which adjusted for demographic and clinical characteristics, prescription opioid use disorder and pain at wave 1 each were associated with themselves at wave 2 (for wave 1 pain with wave 2 pain, β=0.53, p<0.001; for wave 1 opioid use disorder with wave 2 opioid use disorder, β=0.25, p=0.002). Moreover, the adjusted correlation between pain and opioid use disorder was substantial at both wave 1 and wave 2. However, whereas prescription opioid use disorder at wave 1 did not lead to pain at wave 2, pain at wave 1 led to prescription opioid use disorder at wave 2 (Figure 1, Table 3). The predicted risk of developing wave 2 prescription opioid use disorder was 0.44% for persons without and 0.62% for those with wave 1 pain interference, an increase of 41% (data not shown). The model was invariant by sex (χ2=46.64, df=34, p=0.07). Post hoc analyses indicated that pain at wave 1 was significantly associated with opioid tolerance (β=0.17, p=0.002) and withdrawal (β=0.24, p<0.001) at wave 2, and the association approached significance for taking opioids longer or at greater doses than prescribed (β=0.12, p=0.07).
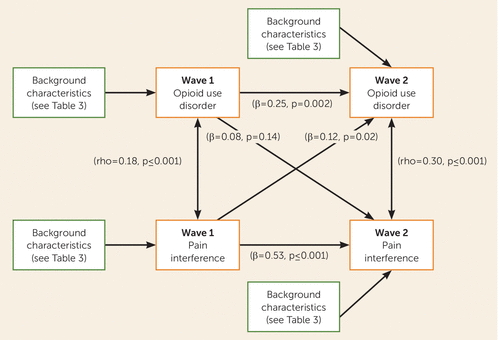
FIGURE 1. Pathways From Opioid Use Disorder and Pain Interference, NESARC Wave 1 (2001–2002) to Wave 2 (2004–2005)a
a NESARC=National Epidemiologic Survey on Alcohol and Related Conditions. Pain interference denotes moderate to extreme interference; opioid use disorder denotes prescription opioid use disorders.
| Wave 1 | Wave 2 | |||||||
|---|---|---|---|---|---|---|---|---|
| Prescription Opioid Use Disorder | Pain Interference | Prescription Opioid Use Disorder | Pain Interference | |||||
| Characteristic | β | p | β | p | β | p | β | p |
| Age | –0.13 | 0.002 | 0.29 | <0.001 | –0.22 | <0.001 | 0.18 | <0.001 |
| Male | 0.09 | 0.02 | –0.06 | <0.001 | 0.07 | 0.07 | –0.03 | 0.01 |
| Family history | ||||||||
| Alcohol use disorder | 0.10 | 0.04 | 0.04 | <0.001 | 0.05 | 0.16 | 0.03 | 0.007 |
| Drug use disorder | –0.01 | 0.72 | 0.03 | 0.006 | 0.04 | 0.29 | 0.001 | 0.95 |
| Depression | 0.03 | 0.50 | 0.01 | 0.24 | 0.06 | 0.20 | –0.004 | 0.69 |
| Antisocial personality disorder | –0.002 | 0.95 | 0.02 | 0.06 | –0.03 | 0.38 | 0.03 | 0.004 |
| Other 12-month substance use disorderb | 0.24 | <0.001 | 0.09 | <0.001 | 0.09 | 0.02 | 0.02 | 0.13 |
| Any 12-month mood or anxiety disorderc | 0.14 | <0.001 | 0.13 | <0.001 | 0.05 | 0.09 | 0.05 | <0.001 |
TABLE 3. Structural Model Correlations and Probabilities of Background Demographic and Clinical Characteristics With Prescription Opioid Use Disorder and Pain Interference at NESARC Waves 1 and 2a
Having a mood or anxiety disorder or family history of alcohol use disorder or antisocial personality disorder at wave 1 was associated with pain and prescription opioid use disorder at wave 1. A family history of drug use disorders was further associated with pain at wave 1. In the structural model, age and sex were associated with both prescription opioid use disorder and pain at wave 1, but in opposite directions. Whereas male sex was directly associated and age inversely associated with prescription opioid use disorder, age was directly associated and male sex inversely associated with pain at wave 1. A family history of antisocial personality disorder at wave 1 was also associated with prescription opioid use disorder at wave 2. Having a mood or anxiety disorder and having a family history of alcohol use disorder or a depression disorder at wave 1 was directly associated and male sex inversely associated with pain at wave 2. Age continued to be directly associated with pain and inversely associated with prescription opioid use disorder at wave 2 (Table 3).
Discussion
In a nationally representative sample assessed twice 3 years apart, pain and prescription opioid use disorders were associated with one another at both time points. However, whereas pain at baseline was associated with prescription opioid use disorder at follow-up, prescription opioid use disorder at baseline was not associated with pain at follow-up. The results were consistent across different modeling strategies, indicating the robustness of the findings. Several demographic and clinical correlates were also directly associated with both pain and prescription opioid use disorders. Furthermore, older age decreased the risk of prescription opioid use disorder but increased the risk of pain, whereas male as compared with female sex increased the risk of prescription opioid use disorder but decreased the risk of pain.
Our first major finding was an association between pain at baseline and disorder-level prescription opioid use at follow-up that was independent of the demographic and clinical factors. The path for pain interference was associated with a 41% relative increase in the risk of developing a prescription opioid use disorder. This prospective association extends results from cross-sectional studies that have documented a link between pain and prescription opioid use disorders (10). Persistent pain may lead some individuals to use prescription opioids in patterns different from what their prescribing physician intended, leading to tolerance and withdrawal symptoms and eventually to opioid abuse or dependence. Pain, an extremely powerful motivator, may also lead individuals to discount the long-term risks of their actions in an urgent effort to suppress pain. Because both pain and opioids can activate dopamine release in their acute phase, they may share some neurobiological mechanisms in the brain reward and motivational systems (30). Complex biological interactions related to pain or inflammation, which often accompanies pain, may also alter opioid receptors and increase the risk of addiction (31).
In order to reduce the long-term risk of prescription opioid use dependence in individuals with chronic pain, ongoing assessment of pain, consideration of alternative treatments, and treatment of comorbid medical or psychiatric conditions may be useful (32). Use of tamper-resistant medications or partial opioid agonists such as buprenorphine may also help provide adequate treatment while minimizing the abuse potential (33). Outside of supervised settings, reducing opioid use in patients with chronic pain is often clinically challenging and quite difficult to achieve and maintain (34). Our findings highlight the need to provide evidence-based treatment for individuals in pain and to develop more effective nonopioid alternative treatments for those who do not respond to existing options.
Our second major finding was that prescription opioid use disorder was not significantly associated with pain at 3-year follow-up after accounting for potential confounding covariates. This contrasts with findings from cross-sectional studies of clinical samples of individuals in methadone programs (35) and in some (36), although not all (37), studies of patients with acute perioperative exposure to opioids. However, our prospective results are consistent with findings from patients with chronic pain (11, 38) and with the results of the only randomized trial prospectively examining opioid-induced hyperalgesia. In that trial, patients with chronic back pain treated with oral morphine were no more likely than those assigned to placebo to develop hyperalgesia (39).
Consistent with previous studies, several demographic and clinical factors were associated with prescription opioid use disorders and pain (10, 40, 41). These findings converge to highlight the complexity of factors that influence the development and maintenance of pain and prescription opioid use disorders and the challenge of studying these phenomena. From the clinical and preventive perspective, these clinical and demographic characteristics identify subgroups at increased risk who should be screened for pain and prescription opioid abuse. They may also reflect overlaps in the etiology of pain, prescription opioid use disorders, and other psychiatric disorders (42), and they support recent interest in developing transdiagnostic approaches to psychiatric disorders and symptoms (43, 44).
This study has several limitations. First, the NESARC sampled individuals age 18 and older, and the relationship between pain and prescription opioid use disorders may differ in younger individuals. Second, pain was assessed only at two time points 3 years apart, precluding the assessment of the relationship between pain and prescription opioid use disorder using other time frames (e.g., lifetime) or over longer periods. Furthermore, the data were collected a decade ago. Nevertheless, the NESARC remains the most recent nationally representative cohort of U.S. adults. Third, the NESARC did not assess inmate populations, which may have a higher prevalence of substance use disorders (45). Fourth, the assessment of nonmedical use of prescription opioids, although extensive, was not exhaustive. In addition, it included two nonopioid medications (celecoxib and rofecoxib). However, because these medications do not have addictive potential, they are unlikely to have led to prescription drug use disorders and thus to have biased the estimates of the study. Fifth, the NESARC did not ask how individuals obtained their medications. Sixth, as in any complex model, the estimates of the associations should be interpreted with caution and taking into account that they are not independent associations, but rather adjusted for the other covariates in the model. Finally, the NESARC assessed pain with a single item and did not query about the location or duration of the pain.
In a large nationally representative sample, pain predicted opioid use disorder. We hope that this finding helps to focus research and practice on development and use of nonopioid strategies for pain management.
1 : Major increases in opioid analgesic abuse in the United States: concerns and strategies. Drug Alcohol Depend 2006; 81:103–107Crossref, Medline, Google Scholar
2 : Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC, National Academies Press, 2011Google Scholar
3 : The prevalence of chronic pain in United States adults: results of an Internet-based survey. J Pain 2010; 11:1230–1239Crossref, Medline, Google Scholar
4 : The economic costs of pain in the United States. J Pain 2012; 13:715–724Crossref, Medline, Google Scholar
5 : Medication-assisted therapies: tackling the opioid-overdose epidemic. N Engl J Med 2014; 370:2063–2066Crossref, Medline, Google Scholar
6 : Pharmaceutical overdose deaths, United States, 2010. JAMA 2013; 309:657–659Crossref, Medline, Google Scholar
7 : Interagency Guideline on Opioid Dosing for Chronic Non-Cancer Pain: An Educational Pilot to Improve Care and Safety With Opioid Treatment. Olympia, Washington State Department of Labor and Industries, 2010Google Scholar
8 : CDC Guideline for Prescribing Opioids for Chronic Pain: United States, 2016. MMWR Recomm Rep 2016; 65:1–49 (https://www.cdc.gov/mmwr/volumes/65/rr/rr6501e1.htm)Google Scholar
9 : Physical pain, common psychiatric, and substance use disorders, and the non-medical use of prescription analgesics in the United States. Drug Alcohol Depend 2009; 100:63–70Crossref, Medline, Google Scholar
10 : The prevalence of mental health and pain symptoms in general population samples reporting nonmedical use of prescription opioids: a systematic review and meta-analysis. J Pain 2012; 13:1029–1044Crossref, Medline, Google Scholar
11 : Opioid-induced hyperalgesia in humans: molecular mechanisms and clinical considerations. Clin J Pain 2008; 24:479–496Crossref, Medline, Google Scholar
12 : Source and Accuracy Statement: The Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. Bethesda, Md, National Institute on Alcohol Abuse and Alcoholism, 2007Google Scholar
13 : Source and Accuracy Statement: Wave 1 of the 2001–2002 National Epidemiologic Survey of Alcohol and Related Conditions (NESARC). Bethesda, Md, National Institute on Alcohol Abuse and Alcoholism, 2003Google Scholar
14 : The Alcohol Use Disorder and Associated Disabilities Interview Schedule-IV (AUDADIS-IV): reliability of alcohol consumption, tobacco use, family history of depression, and psychiatric diagnostic modules in a general population sample. Drug Alcohol Dep 2003; 71:7–16Crossref, Medline, Google Scholar
15 : The Alcohol Use Disorder and Associated Disabilities Interview Schedule–DSM-IV Version. Bethesda, Md, National Institute on Alcohol Abuse and Alcoholism, 2001Google Scholar
16 : The Alcohol Use Disorder and Associated Disabilities Interview Schedule (AUDADIS): reliability of alcohol and drug modules in a clinical sample. Drug Alcohol Depend 1997; 44:133–141Crossref, Medline, Google Scholar
17 : The Alcohol Use Disorder and Associated Disabilities Interview schedule (AUDADIS): reliability of alcohol and drug modules in a general population sample. Drug Alcohol Depend 1995; 39:37–44Crossref, Medline, Google Scholar
18 : The National Epidemiologic Survey on Alcohol and Related Conditions (NESARC) waves 1 and 2: review and summary of findings. Soc Psychiatry Psychiatr Epidemiol 2015; 50:1609–1640Crossref, Medline, Google Scholar
19 : Sociodemographic and psychopathologic predictors of first incidence of DSM-IV substance use, mood, and anxiety disorders: results from the wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. Mol Psychiatry 2009; 14:1051–1066Crossref, Medline, Google Scholar
20 : The Spanish Alcohol Use Disorder and Associated Disabilities Interview Schedule (AUDADIS): reliability and concordance with clinical diagnoses in a Hispanic population. J Stud Alcohol 1999; 60:790–799Crossref, Medline, Google Scholar
21 : Differences among major depressive disorder with and without co-occurring substance use disorders and substance-induced depressive disorder: results from the National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry 2012; 73:865–873Crossref, Medline, Google Scholar
22 : A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care 1996; 34:220–233Crossref, Medline, Google Scholar
23 : Effect of first episode axis I disorders on quality of life. J Nerv Ment Dis 2014; 202:271–274Crossref, Medline, Google Scholar
24 : Quality of life following remission of mental disorders: findings from the National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry 2013; 74:e445–e450Crossref, Medline, Google Scholar
25 : SF-36 Health Survey: Manual and Interpretation Guide. Boston, New England Medical Center, Health Institute, 1993Google Scholar
26 : Mood and anxiety disorders and their association with non-medical prescription opioid use and prescription opioid-use disorder: longitudinal evidence from the National Epidemiologic Study on Alcohol and Related Conditions. Psychol Med 2012; 42:1261–1272Crossref, Medline, Google Scholar
27 : The latent structure and predictors of non-medical prescription drug use and prescription drug use disorders: a national study. Drug Alcohol Depend 2013; 133:473–479Crossref, Medline, Google Scholar
28 : Econometric Analysis, 5th ed. Boston, Prentice Hall, 2003Google Scholar
29 : Changes in the prevalence of non-medical prescription drug use and drug use disorders in the United States: 1991–1992 and 2001–2002. Drug Alcohol Depend 2007; 90:252–260Crossref, Medline, Google Scholar
30 : The missing p in psychiatric training: why it is important to teach pain to psychiatrists. Arch Gen Psychiatry 2011; 68:12–20Crossref, Medline, Google Scholar
31 : Opioid and chemokine receptor crosstalk: a promising target for pain therapy? Nat Rev Neurosci 2015; 16:69–78Crossref, Medline, Google Scholar
32 : Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J Pain 2009; 10:113–130Crossref, Medline, Google Scholar
33 : Abuse-deterrent formulations and the prescription opioid abuse epidemic in the United States: lessons learned from oxycontin. JAMA Psychiatry 2015; 72:424–430Crossref, Medline, Google Scholar
34 : Interventions for the reduction of prescribed opioid use in chronic non-cancer pain. Cochrane Database Syst Rev 2013; 9:CD010323Medline, Google Scholar
35 : Methadone maintenance patients are cross-tolerant to the antinociceptive effects of morphine. Pain 2001; 93:155–163Crossref, Medline, Google Scholar
36 : Acute opioid tolerance: intraoperative remifentanil increases postoperative pain and morphine requirement. Anesthesiology 2000; 93:409–417Crossref, Medline, Google Scholar
37 : Intraoperative remifentanil infusion does not increase postoperative opioid consumption compared with 70% nitrous oxide. Anesthesiology 2005; 102:398–402Crossref, Medline, Google Scholar
38 : Oral opioid use alters DNIC but not cold pain perception in patients with chronic pain: new perspective of opioid-induced hyperalgesia. Pain 2008; 139:431–438Crossref, Medline, Google Scholar
39 : Analgesic tolerance without demonstrable opioid-induced hyperalgesia: a double-blinded, randomized, placebo-controlled trial of sustained-release morphine for treatment of chronic nonradicular low-back pain. Pain 2012; 153:1583–1592Crossref, Medline, Google Scholar
40 : Motives for nonmedical use of prescription opioids among high school seniors in the United States: self-treatment and beyond. Arch Pediatr Adolesc Med 2009; 163:739–744Crossref, Medline, Google Scholar
41 : Correlates of extramedical use of OxyContin versus other analgesic opioids among the US general population. Drug Alcohol Depend 2009; 99:58–67Crossref, Medline, Google Scholar
42 : The downward spiral of chronic pain, prescription opioid misuse, and addiction: cognitive, affective, and neuropsychopharmacologic pathways. Neurosci Biobehav Rev 2013; 37:2597–2607Crossref, Medline, Google Scholar
43 : The structure of common mental disorders. Arch Gen Psychiatry 1999; 56:921–926Crossref, Medline, Google Scholar
44 : Mapping common psychiatric disorders: structure and predictive validity in the National Epidemiologic Survey on Alcohol and Related Conditions. JAMA Psychiatry 2013; 70:199–208Crossref, Medline, Google Scholar
45 : The effect of inmate populations on estimates of DSM-IV alcohol and drug use disorders in the United States. Am J Psychiatry 2010; 167:473–474Link, Google Scholar



