Autism in a 15-Month-Old Child
Autism is a developmental disorder marked by severe deficits in reciprocal social interaction, communication, and imagination as well as repetitive and restricted patterns of interests and behaviors (1; DSM-IV-TR). Its early-onset symptom profile includes neurological findings (2) and structural and functional brain abnormalities (3); these findings and the chronicity of autism strongly argue for a biological basis, including genetic liabilities (4). Epidemiologic studies indicate rates of 16 children per 10,000 for more narrowly defined autism and up to 40 children per 10,000 for the more broadly defined group of conditions marked by substantial socialization deficits (5). Despite the presumed congenital nature of autism, its identification typically lags behind the onset of symptoms by several years (6–9). Epidemiologic studies currently place the mean age of diagnosis of autism at around 40 months (5).
Early identification is very important (10–12) given the current consensus that the beneficial impact of intervention programs increases with earlier treatment (13–15). This is likely a function of early neuroplasticity and the importance of early experiences in shaping behavioral and brain development (16, 17).
Over the past decade new information on autism as it appears in the second and third year of life has focused on social orientation and responsivity, communicative development (including both nonverbal communication and spoken language), imitation, and play and symbolic development (18–26) (see reference 27 for a review). However, specific knowledge of the crucial infancy period of development in individuals with autism is currently limited and includes a small number of retrospective studies based on parental report (6) as well as systematic analyses of home movies made by parents before the child was diagnosed (28–32). Despite methodological limitations, these studies have pointed to a series of converging findings. First, it is possible to differentiate infants who will be diagnosed as having autism from infants who will be diagnosed as having nonautistic global developmental delays and from typically developing children by the age of 12 months (28, 29, 31, 33) if not earlier (32). Second, a tendency to look at others less and to orient themselves less to people calling their names appears to be the most syndrome-specific deficit by the time children with autism are 1 year old (31, 32). Finally, there is some evidence that infants subsequently diagnosed with autism also display a series of sensory-processing and sensorimotor abnormalities (29) and use gestures and look at objects held by others less frequently than nonautistic subjects (31), although the syndrome specificity of these phenomena is not quite yet established.
An exceedingly small number of studies reported direct, contemporaneous observations of infants and toddlers with autism (34–37). The only prospective study from birth is a case report (34), which traced the child’s developmental abnormalities. During the first 6 months of life, there were difficulties in oral motor coordination and both hyper- and hypotonia. Interestingly, the child vocalized and responded socially to others by smiling and cooing. During the second half of the first year, the child continued to show sensorimotor difficulties and diminished oral-motor control and began to exhibit motor stereotypies associated with difficulties in social interaction. At 1 year of age the child met criteria for autism on a standardized diagnostic instrument.
A remarkably small number of published reports contain findings based on experimental (in contrast to observational) protocols of children younger than 3. These relate to abnormalities in response to eye-gaze cues (38), reduced visual focus on people in general and on more specific social cues (39, 40), and lack of mental representations of human action (39). None of these studies has been replicated to date. To our knowledge, there are no published studies to date using experimental protocols to test specific developmental hypotheses of the pathogenesis of this condition in infants and toddlers with autism.
There is a great need for systematic, directly obtained information on children with autism under the age of 3 years. The current definition of autism (DSM-IV-TR) was not researched among children younger than 3 (41), and there are concerns about its applicability and utility in the case of this ever-increasing group of children seen for clinical assessments (see references 42–44 for review).
This report describes the findings of an interdisciplinary developmental disabilities evaluation of a 15-month-old girl with autism. The evaluation included standardized measures of motor, cognitive, communication, and symbolic skills, adaptive behaviors, and autistic symptoms. It also included neurological and genetic workups. This child completed identical protocols at 15, 23, and 34 months. To our knowledge, this is the most comprehensive case study of a toddler with autism to date.
Case Presentation
The parents of “Helen,” a 15-month-old girl, sought a developmental disabilities evaluation because of their concern that she had stopped vocalizing and using words in the previous 2 or 3 months. They had been concerned about her development from shortly after her birth because we had diagnosed her brother, who is 22 months older, with autism when he was 2 years old.
Pregnancy and Perinatal Period
Helen’s mother had severe nausea as well as thyroid disease (treated with levothyroxine sodium) during pregnancy. Labor was induced 2 days before the expected date.
After delivery, Helen was kept in the newborn special care unit for a short period of time because her blood sugar was low, but she did well after discharge. She exhibited some colic and had difficulty with breast feeding but adjusted well to bottle-feeding. There was no report of unusual sensitivities during the first few months of life. Milestones were grossly in time. She smiled at 3 weeks of age, sat at 4 months, crawled at 6 months, and walked at 12 months. By that time, Helen was saying words such as “hi,” “baby,” “mommy,” and “daddy.” Normal development to this point was also documented by her pediatrician, who shared the parents’ concern given her older brother’s autism.
Shortly after 12 months Helen stopped using words. Her parents associated this loss with a series of four ear infections that occurred within a span of 2 or 3 months and were treated with antibiotics. Along with the loss of verbalizations, the parents noted that Helen became increasingly less socially engaged. She did not bring things to her parents (e.g., to show, to request). At 15 months, she developed aversion to bright lights and loud noises. Although reported to be still affectionate with her parents, she typically ignored other people. At 15 months, Helen had not had a hearing test, nor had she received any form of therapy or intervention.
Assessment Results
Helen’s clinical evaluations at 15, 23, and 34 months included 1) developmental assessment (Mullen Scales of Early Learning [45]), 2) a speech, language, and communication assessment including both parent-report measures such as the MacArthur Communicative Development Inventory (46) and direct measures such as the Communication and Symbolic Behavior Scales Developmental Profile (47), 3) a standardized diagnostic observational play protocol (module 1 of the Autism Diagnostic Observation Schedule [48]) and a measure of adaptive behavior (Vineland Adaptive Behavior Scales [49]). At the age of 15 and 34 months, a standardized parent interview (Autism Diagnostic Interview Toddler Version [50]) was conducted with Helen’s mother.
Assessments at 15 Months
On the developmental assessment, Helen’s scores were within the average range for nonverbal problem-solving skills (e.g., functional use of objects, object permanence, emerging shape discrimination skills) with performance at the 14-month level (Figure 1). Her scores for fine and gross motor skills (eye-hand coordination, scribbling in imitation, running, walking upstairs with assistance, throwing a ball) were also within average range, with performance at the 15- and 16-month levels, respectively. She exhibited significant delays in language development, however. Her receptive skills were at about the 5-month level, and her expressive skills were at the 9-month level, indicating delays of 6 to 8 months.
During testing Helen was unlikely to respond to simple verbal prompts (“sit,” “bye-bye”), verbal requests (“give me…,” “show me…”), even when paired with accompanying gestures or animated and playful vocalizations (including calling of her name). It was not possible to elicit a smile or vocalization without touching her. Helen did vocalize a great deal, but not in response or as an approach to other people. These vocalizations occurred primarily when she was exploring the environment alone or running aimlessly and did not appear to have a communicative purpose. Her vocalizations included consonant-vowel combinations but not words or word approximations. Helen’s only gestures were reaching for a desired object or pushing it away. These were unaccompanied by eye contact.
During the diagnostic play session (Autism Diagnostic Observation Schedule), Helen was extremely self-isolated. If left to her own devices, Helen perseverated on exploration of cause-effect toys (e.g., blocks, pop-up toys) and objects with special textures, ignoring the examiner’s bids for engagement, which ranged from incidental comments or facial/bodily gestures to highly animated, exaggerated, and sometimes intrusive (less than 5 inches from her face) vocalizations and gestures. Eye contact was very infrequent and could not be elicited in a predictable manner. Her smiles were mostly in response to her own actions (flicking a doll’s eye back and forth) or in response to sensory experiences (e.g., lights, shiny objects). Helen could reenact some simple actions modeled by the adult (e.g., dipping a bubble stick inside a little bottle with soapy water) but typically would not imitate the examiner’s entire goal-directed behavior (e.g., making soap bubbles) or the examiner’s style (e.g., exaggerating specific aspects of the movements). It was not possible to engage Helen in early anticipatory or imitative games (e.g., peek-a-boo), she did not appear to learn from the adult’s attitudes toward novel objects (e.g., she continued to be fearful of a mechanical bunny despite the examiner’s exaggerated displays of affection toward the toy animal), and, if forced to make eye contact, she typically focused on the periphery (e.g., hair, chin) of the approaching adult’s head or overly fixated on the adult’s mouth. She was not interested in nor did she explore any of the miniatures (e.g., people, furniture) or representational toys for any length of time. She mouthed the miniatures, very much like she mouthed nonrepresentational toys. There were no episodes of symbolic play. She vocalized infrequently. When frustrated, she made high-pitched sounds that were self-stimulatory rather than communicative. She was extremely independent, almost never using the adult to get something she wanted. She did not point to request something from or to show something to the adult or her parents, who were also in the room. She was not sensitive to exaggerated gaze cues or pointing gestures (i.e., she did not look in the direction of the adult’s gaze or pointing).
Her attention shifted quickly from object to object. If unable to secure a desired object, she mouthed what she had in her hands. She showed a number of self-stimulatory behaviors and unusual sensitivities. She flapped her arms and held her arms, hands, and fingers in unusual postures. Her disregard of the social approaches of others contrasted markedly with her acute awareness of inanimate details of the environment (e.g., finding a small piece of candy some 8 feet away from the play area). She was easily scared by noises and novel events (e.g., the noises of a balloon deflating), startling repeatedly until the adult discontinued the activity.
The speech, language, and communication assessment took place in a highly animated interactional environment to maximize social engagement and speech and communication production through familiar play routines (e.g., dancing, nursery rhymes, wind-up toys, blowing bubbles). By parent report, expressive and receptive language were delayed, with vocabulary production at 9 months, gesture production at 12 months, vocabulary comprehension at 8 months, and phrase comprehension at less than 8 months.
None of the gestures Helen displayed during assessment were conventional—no pointing, nodding, or showing were noted. Helen’s gestures consisted primarily of depictive and physical gestures, made to complete a physical routine (e.g., reenacting the motions of “Ring Around the Rosie” and pulling on the examiner’s hand to request falling “down”) or as a means to an end (e.g., moving a bubble jar to the examiner to see more bubbles) rather than communicative requests or attempts to share an experience with her play partner. In more spontaneous situations, Helen was extremely independent in getting desired objects and was likely to express frustration by moving the examiner’s hands away rather than making eye contact or otherwise directing her communication to the adult. By parent report, Helen was more likely to use her parents to obtain desired things in the environment, although she was also likely to discontinue this contact if her idiosyncratic gestures or vocalizations did not achieve the desired result. She sometimes sought physical proximity to her parents when distressed (e.g., when frightened of sudden noises), but not often. She was able to respond to verbal directions only in the context of strong visual cues (e.g., “give me” paired with an open palm and a toy bag), following predictable carrier phrases (e.g., “ready, set, go”), and routines (e.g., “if you’re happy and you know it, clap your hands”). Vocal and gestural imitation could be elicited only in these situations, and very inconsistently.
With regard to adaptive skills, Helen’s profile on the Vineland Adaptive Behavior Scales showed great scatter (Figure 2). At 15 months of age, daily living skills and motor skills were at or above age level (at 14- and 17-month levels, respectively); in contrast, there were significant delays in communication (10-month level) and, particularly, socialization (9-month level) skills. Helen’s parents reported somewhat higher receptive language skills in more familiar contexts (14 months) than those observed during the formal assessment and expressive language skills (approximately at the 8-month level). Her interpersonal relationships were at the 7-month level, and her play skills were at the 10-month level. Several social responses typically seen in the first 6 months of life were not present at 15 months of age (e.g., following a person moving around with her eyes, smiling to make social contact, looking for a familiar person when in need of attention).
Genetic and Neurological Evaluation
As noted, Helen’s brother had been diagnosed with a prototypical form of autism a short time before her birth. Otherwise there was no family history of autism or other developmental disorder in the immediate or extended family. Helen underwent genetic evaluations at 17 and 20 months. By report, Helen and her then 3.5-year-old brother with autism appeared to have lost some skills at around 12 months, although Helen lost words and social skills and her brother only the latter (he did not have words at that time). Both Helen and her brother are macrocephalic, which appears to be a family trait (the father’s head circumference is 58 cm [90th percentile], and the mother’s is 58 cm [>95th percentile]). Both children had accelerated head growth, although at different times. Both of them also showed accelerated weight and length gain, but not as dramatic as their head circumference. Helen’s head circumference went from the 50th percentile at 6 months to >95th percentile by 9 months (stabilizing thereafter), whereas her brother went from slightly below the 50th percentile at 2 weeks of age to the 95th percentile at 2 months and >95th percentile at 4 months (stabilizing thereafter). Helen’s weight was at the 75th percentile at 2 months, 90th percentile at 4 months, and >95th percentile at 6 months; her length was at the 75th percentile at 2 months and the 90th percentile at 4 months, continuing to fluctuate between the 75th and the 95th percentile until 24 months. Her brother’s weight was at the 95th percentile by 2 months and remained so until 24 months; his length was at about the 35th percentile at 2 months and fluctuated between the 75th and 95th percentile from 4 months to 24 months.
On examination at 15 months, Helen was essentially nondysmorphic, except for some minor distinctive features, including a prominent forehead, deep-set eyes, flat nasal bridge, and tapered digits (features that were similar to her brother’s). Results of head ultrasound were normal. There were no neurocutaneous lesions to suggest either tuberous sclerosis or neurofibromatosis. A series of genetic and metabolic tests were performed, and the results were all normal. These included fragile X polymerase chain reaction, subtelomeric fluorescence in situ hybridization (for chromosomal microdeletions), karyotype, urine purine and pyrimidine analysis, guanidinoacetate (for creatine synthesis defects presenting as developmental arrest, often with seizures), urine organic acids (for organic acidurias and mitochondrial disorders), fasting plasma and amino acids (for mitochondropathies and aminoacidopathies), lactate/pyruvate, and creatine phosphokinase (additional screening for mitochondrial disorders). Helen’s brother had results on normal antigliadin and antiendomysial antibody screens for celiac sprue (carried out because of his alternating constipation and diarrhea). He also had normal results on EEG and brain magnetic resonance imagining scan, carried out after he had one seizure. Results of Helen’s neurological examination were essentially normal. She had a 24-hour EEG with no findings.
Diagnostic Formulation and Early Intervention Services
At the time of Helen’s 15-month assessment, the diagnostic impression by all the clinicians was autism. Unlike some very young children, Helen also met criteria for autism on the Autism Diagnostic Observation Schedule and on the Autism Diagnostic Interview. The program of early intervention prescribed included an intensive and highly structured and individualized therapeutic schedule consisting of speech- and language-based functional activities delivered by specialized educators and communication therapists as well as direct family training and support. Although a substantial body of work exists on intervention programs for slightly older children (1), information on intervention for infants is limited; therefore, we adapted principles for intervention used in slightly older children for Helen in the light of her particular pattern of strengths and weaknesses.
Goals for her program included the following: 1) to increase Helen’s rate of spontaneous and intentional communication by designing the environment to provide opportunities for her to independently initiate communicative acts (e.g., communicative “temptations,” reinforcement of eye contact and gestures directed at adults, use of predictable carrier phrases), 2) to increase Helen’s capacity to communicate for behavior regulation (e.g., to request actions and access to desired objects, to protest), 3) to increase her capacity to communicate for engaging in social interaction (e.g., calling attention to oneself, requesting continuation of a social routine, requesting comfort, showing off, greeting others), 4) to increase Helen’s capacity to bring attention to objects or events for the purpose of joint attention (e.g., commenting and requesting information), 5) to increase Helen’s repertoire of gestural communication (e.g., pairing of giving and pointing gestures with gaze and conventional gestures such as waving bye-bye and “all done”), 6) to increase Helen’s single-word vocabulary through the use of a photo/picture communication system, 7) to facilitate comprehension of language by repeated exposure to one- or two-word phrases corresponding to common actions (e.g., “wash hands,” “put in,” “all done”) as well as labels of common objects (e.g., puzzle, book), and 8) to facilitate engagement in symbolic play by adult-directed use of miniatures to mirror Helen’s own real-life, familiar, and repetitive events. Specific recommendations were also made for enhancing Helen’s adaptive level of functioning in the areas of communication, daily living skills, and socialization.
Follow-Up Assessments at 23 and 34 Months
By 18 months, Helen had pressure-equalization tubes placed because of repeated ear infections. Following that, she was reported to produce a wider range of sounds. Therapy had begun with a speech and language therapist, but only once a week for 1 hour. Two additional hours of therapy were provided by a special education teacher under the supervision of the communication specialist. Helen also joined two small play groups, but her participation was minimal because of her self-isolation. Helen’s parents brought her to a brief consultation for a diagnostic play session 2 weeks after the pressure-equalization tubes were placed. Helen’s presentation was virtually unchanged since the evaluation at the age of 15 months.
At the age of 23 months, Helen completed a reevaluation consisting of developmental assessment (Mullen Scales of Early Learning), diagnostic play (Autism Diagnostic Observation Schedule), and measurement of adaptive behaviors (Vineland Adaptive Behavior Scales). Her profile of strengths and deficits on developmental assessment (Figure 1) was unchanged relative to her evaluation at the age of 15 months, with encouraging gains in nonverbal cognitive functioning (going from the 14-month level to the 23-month level), fine motor skills (from 15 months to 22 months), and gross motor skills (from 16 months to 20 months), but only minimal gains in receptive language (from about 5 months to 8 months) and in expressive language (from about 9 months to 13 months). Helen’s Autism Diagnostic Observation Schedule scores were virtually unchanged, indicating a highly stable level of symptoms.
Absolute progress in the acquisition of adaptive skills in all areas of the Vineland Adaptive Behavior Scales was minimal, with the resultant drop in standard scores. At the age of 23 months, Helen’s age equivalent scores in the communication and socialization domain were at the 10-month levels (Figure 2). These results prompted a renewed and more concerted effort to secure intensive, appropriate, and specialized interventions, adding a core element of “learning to learn” skills (e.g., facilitating more active engagement in adult-led activities following highly routinized and behaviorally based learning tasks). Additionally, it was evident that a significant increase in the number of hours of her intervention program was warranted (her service providers had been unsure of the diagnosis and the need for intensive services).
Results of the 34-month assessment were less encouraging in terms of progress made. In contrast to the previous year, the absolute gains on the Mullen Scales of Early Learning were minimal (Figure 1). In the 11-month interim period, Helen gained only 1 month in nonverbal cognitive functioning (24-month level), 3 months in receptive language (11-month level), 1 month in expressive language (14-month level), and no change in fine motor skills (22-month level). Helen’s gross motor skills were not assessed because she was unable to attend to or imitate most of the gross motor probes.
The diagnostic play session (Autism Diagnostic Observation Schedule) revealed a pattern of scores that was virtually unchanged relative to the two previous assessments. Helen’s acquisition of adaptive skills was also stunted. She gained approximately 4 months in the 11-month period in the area of communication, her daily living skills advanced 1 month during that time, and there were no gains in the area of socialization. The most growth was noted in motor skills (6 months) (Figure 2).
Discussion
Helen’s case suggests that the diagnosis of autism can be made as early as 15 months of age. It demonstrates the utility of several standardized instruments that may aid in patient characterization and diagnostic formulation. Helen’s developmental profile on the Mullen Scales of Early Learning, which showed higher nonverbal cognitive functioning and motor skills and lower receptive and expressive language, followed the same scattered profile obtained for a large number of 2- to 3-year-old children with autism (unpublished paper by K. Chawarska et al.). Helen’s profile on the Communication and Symbolic Behavior Scales Developmental Profile was also consistent with available data for older children with autism on this instrument (26, 47) and was helpful in specifying a plan of intervention in this core area of disability. Her profile on adaptive skills revealed the familiar Vineland Adaptive Behavior Scales profile for children with autism, consisting of higher levels of ability in motor and daily living skills and delays in the communication and, particularly, socialization domains (51).
Despite the well-known limitations of the Autism Diagnostic Interview and Autism Diagnostic Observation Schedule to make a diagnosis of autism in very young (<2 years) and developmentally delayed children (18, 52), Helen met criteria for autism on both instruments. Often, young children fail to meet criteria because symptoms are not observed in the area of “narrow and repetitive interests and behaviors.” Helen showed a range of stereotypic behaviors that were more pronounced in the areas of response to sensory stimuli (e.g., focusing on lights and shiny surfaces, seeking to manipulate specific textures) and perseveration on repetitive toys and routines (e.g., she could play for long periods of time with cause-effect toys if left to her own devices) rather than stereotypic movements (e.g., hand flapping, body rocking), although she did exhibit some of these behaviors too.
Follow-up assessments 8 and 19 months later revealed an unchanged profile of relative strengths and substantial deficits in developmental abilities as measured with the Mullen Scales of Early Learning (more progress in nonverbal cognitive functioning and motor skills and very modest progress in receptive and expressive language), very little absolute progress in the acquisition of adaptive skills (and the consequent decrease in standard scores) as measured with the Vineland Adaptive Behavior Scales, and a remarkably stable level of psychopathology on the Autism Diagnostic Observation Schedule.
It is of great concern that the considerable progress in nonverbal problem-solving skills and (to a lesser extent) in fine motor skills documented at 23 months was not evidenced at 34 months. There is no doubt that Helen’s developmental trajectory reflects a complex interplay between her genetic and neurobiological makeup and experiential factors. Her social disability and attentional difficulties limit significantly the range of experiences typically available to nonaffected infants and toddlers. Helen tends to isolate herself and responds with active avoidance to purely social overtures. Her extremely limited motor and vocal imitation skills severely restrict her ability to learn through observation.
Over time, Helen’s tendency to explore novel toys has declined and her attention has become more fleeting. Her spontaneous activities consist frequently of seemingly aimless wandering, climbing, mouthing objects, or engaging with toys producing immediate effects (e.g., pushing buttons). She appears more restless and more perturbed by changes in the environment and the demands placed on her. Her self-stimulatory and repetitive behaviors have intensified, and the range of her interests has become narrower.
All of these factors are likely to have an impact on the aspects of social, cognitive, and communicative development that depend heavily on the access to appropriate experiences during periods of greatest sensitivity. Moreover, a transition into the third year of life is usually marked by the development of representational ability, involving significant improvement in representational accuracy, durability, and flexibility as well as the development of verbal and nonverbal categorization. This conceptual shift posits great difficulties for Helen, and this is reflected in her declining standard scores on developmental testing.
Although recommendations were made for a intensive and comprehensive treatment program at 15 months, it was not until the follow-up assessment at 23 months that Helen’s parents were able to attain an appropriate program of intervention for her. Several factors appeared to delay this process, including the fact that service providers did not believe that Helen in fact had autism or required a very intensive (in terms of number of hours of intervention), comprehensive (in terms of range of target areas), and specialized (in terms of preparedness of the interventionists, i.e., knowledge of autism and experience working with very young children) program.
Helen’s very early referral to a specialized clinic (at 15 months rather than 40 months, the epidemiologic mean age for a diagnosis of autism [5]) was the result of her pediatrician’s alertness and the fact that her parents were sensitized (and anxious) about genetic liability following the diagnosis of Helen’s older brother. Despite the pediatric follow-up that Helen had during the first year of life, the extent to which she suffered a very meaningful regression at the age of 12 to 14 months is still not clear. It is also unclear whether detectable social symptoms could have been identified in the first year of life by using methods that are not practical in routine pediatric visits. It is unequivocal, however, that Helen lost some words that she previously had.
There are several possibilities that could account for Helen’s regression: 1) a meaningful regressive phase characterizes the onset of autism in a group of children, not unlike what is observed in children with childhood disintegrative disorder early in life (53); 2) the parental perception of regression is an artifact of the greater visibility of the disability in the second year of life (i.e., earlier development is seen as “normal”) because environmental demands and expectations are greater (e.g., one expects toddlers to begin to talk) (54); and 3) a “pseudo-regressive phase” takes places (e.g., a child loses some words), but this process is part of the natural course of autism—onset of the condition does not coincide with the “regression” phase but precedes it, and words lost are results of a maturational process of speech acquisition that is not an integral part of the development of communication and, therefore, is not reinforced by the typical natural predisposition to seek and communicate with others. This would be a process not unlike the emergence of babbling in congenitally deaf children, which subsequently fades away (55). The issue of regression in autism is far from resolved despite an ongoing concerted effort to elucidate it (56).
Finally, the only neurobiological finding in Helen’s evaluation was the accelerated head growth; her head circumference measures went from the 50th percentile at 6 months to the 95th percentile at 9 months. This finding maps well on recently available data on head growth in children with autism (57). Interestingly, her brother also exhibited abruptly accelerated head growth, going from below the 50th percentile to the 95th percentile by 2 months of age. Although the neurophysiological meaning of accelerated head growth in autism is still debated (e.g., involving irregularities in genes associated with brain growth [58]), it is possible that the earlier onset of this abrupt shift in Helen’s brother could be associated with the fact that he was also more cognitively impaired.
These issues are further complicated by the fact that measurements of general weight in both children also shifted abruptly (although this coincided with accelerated head growth only in Helen’s brother; her weight gain occurred before 6 months), and macrocephaly appeared to be a familial trait.
Our unequivocal conclusion is that the diagnosis of autism can be made in some cases very early in the second year of life and that this diagnostic formulation may be stable over the course of early childhood. The extent to which this finding applies to children with autism with variable developmental profiles (including those who do not show language delays) should be an important area of research in the future.
Received July 23, 2004; accepted July 26, 2004. From the Yale Child Study Center and the Department of Genetics, Yale University School of Medicine, New Haven, Conn.; the Department of Communication Disorders, Southern Connecticut State University, New Haven, Conn.; and Communication Crossroads, Carmel, Calif. Address reprint requests to Dr. Klin, Yale Child Study Center, P.O. Box 207900, New Haven CT 06520; [email protected] (e-mail). Supported by National Institute of Child Health and Human Development Collaborative Programs of Excellence in Autism project grant 1PO1 HD-35482-01, grant 5-P01-HD-03008, and grant R01-HD-042127-02 and by NIMH Studies to Advance Autism Research and Treatment program project grant U54-MH-066494, the Yale Children’s Clinical Research Center, the National Alliance for Autism Research, Cure Autism Now, the Doris Duke Charitable Foundation, and the Simons Foundation. The authors thank “Helen” and her parents for their assistance.
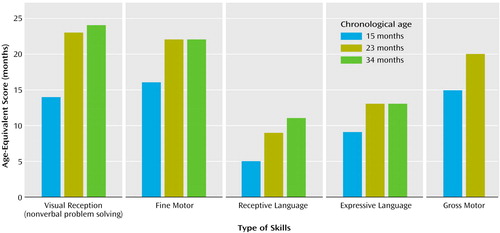
Figure 1. Age-Equivalent Scores of a Child With Autism on the Mullen Scales of Early Learning at 15-, 23-, and 34-Month Assessmentsa
aGross motor skills were not assessed at 34 months because the child was not able to attend to or imitate most of the gross motor probes.
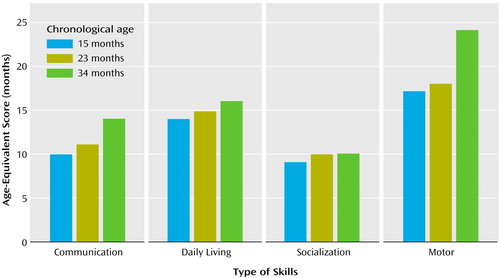
Figure 2. Age-Equivalent Scores of a Child With Autism on the Vineland Adaptive Behavior Scales at 15-, 23-, and 34-Month Assessments
1. Volkmar FR, Lord C, Bailey A, Schultz RT, Klin A: Autism and pervasive developmental disorders. J Child Psychol Psychiatry 2004; 45:1–36Crossref, Google Scholar
2. Volkmar FR, Nelson DS: Seizure disorders in autism. J Am Acad Child Adolesc Psychiatry 1990; 29:127–129Crossref, Medline, Google Scholar
3. Schultz RT, Klin A: The neural foundations of autism. J Am Acad Child Adolesc Psychiatry 2002; 41:1259–1262Crossref, Medline, Google Scholar
4. Rutter M: Genetic studies of autism: from the 1970s into the millennium. J Child Psychol Psychiatry 2000; 28:3–14Google Scholar
5. Chakrabarti S, Fombonne E: Pervasive developmental disorders in preschool children. JAMA 2001; 285:3093–3099Crossref, Medline, Google Scholar
6. Volkmar FR, Stier DM, Cohen DJ: Age of recognition of pervasive developmental disorder. Am J Psychiatry 1985; 142:1450–1452Link, Google Scholar
7. Siegel B, Piner C, Eschler J, Elliott GR: How children with autism are diagnosed: difficulties in identification of children with multiple developmental delays. J Dev Behav Pediatr 1988; 9:199–204Crossref, Medline, Google Scholar
8. Rogers SJ, DiLalla DL: Age of symptom onset in young children with pervasive developmental disorders. J Am Acad Child Adolesc Psychiatry 1990; 29:863–872Crossref, Medline, Google Scholar
9. Stone WL, Lee EB, Ashford L, Brissie J, Hepburn SL, Coonrod EE, Weiss BH: Can autism be diagnosed accurately in children under 3 years? J Child Psychol Psychiatry 1999; 40:219–226Crossref, Medline, Google Scholar
10. Baird G, Charman T, Baron-Cohen S, Cox A, Swettenham J, Wheelwright S, Drew A: A screening instrument for autism at 18 months of age: a 6-year follow-up study. J Am Acad Child Adolesc Psychiatry 2000; 39:694–702Crossref, Medline, Google Scholar
11. Briggs-Gowan MJ, Carter AS, Moye Skuban E, McCue Horwitz S: Prevalence of social-emotional and behavioral problems in a community sample of 1- and 2-year-old children. J Am Acad Child Adolesc Psychiatry 2001; 40:811–819Crossref, Medline, Google Scholar
12. Robins DL, Fein D, Barton ML, Green JA: The Modified Checklist for Autism in Toddlers: an initial study investigating the early detection of autism and pervasive developmental disorders. J Autism Dev Disord 2001; 31:131–144Crossref, Medline, Google Scholar
13. Rogers SJ: Empirically supported comprehensive treatments for young children with autism. J Clin Child Psychol 1998; 27:168–179Crossref, Medline, Google Scholar
14. Filipek P, Accardo PJ, Baranek G, Cook EH Jr, Dawson G, Gordon B, Gravel JS, Johnson CP, Kallen RJ, Levy SE, Minshew NJ, Ozonoff S, Prizant BM, Rapin I, Rogers SJ, Stone WL, Teplin S, Tuchman RF, Volkmar FR: The screening and diagnosis of autistic spectrum disorders. J Autism Dev Disord 1999; 29:439–484Crossref, Medline, Google Scholar
15. National Research Council Committee on Educational Interventions for Children With Autism, Division of Behavioral and Social Sciences and Education: Educating Children With Autism. Washington, DC, National Academy Press, 2001Google Scholar
16. Dawson G, Ashman SB, Carver LJ: The role of early experience in shaping behavioral and brain development and its implications for social policy. Dev Psychopathol 2000; 12:695–712Crossref, Medline, Google Scholar
17. Johnson M: Functional brain development in humans. Nat Rev Neurosci 2001; 2:475–483Crossref, Medline, Google Scholar
18. Lord C: Follow-up of two-year-olds referred for possible autism. J Child Psychol Psychiatry 1995; 36:1365–1382Crossref, Medline, Google Scholar
19. Stone WL, Ousley OY, Hepburn SL, Hogan KL, Hepburn SL: Nonverbal communication in two- and three-year-old children with autism. J Autism Dev Disord 1997; 27:677–696Crossref, Medline, Google Scholar
20. Rogers SJ, Hepburn SL, Stackhouse T, Wehner E: Imitation performance in toddlers with autism and those with other developmental disorders. J Child Psychol Psychiatry 2003; 44:763–781Crossref, Medline, Google Scholar
21. Charman T, Baron-Cohen S, Swettenham J, Baird G, Cox A, Drew A: Testing joint attention, imitation, and play as infancy precursors to language and theory of mind. Cognitive Development 2001; 15:481–498Crossref, Google Scholar
22. Charman T, Swettenham J, Baron-Cohen S, Cox A, Baird G, Drew A: Infants with autism: an investigation of empathy, pretend play, joint attention, and imitation. Dev Psychol 1997; 33:781–789Crossref, Medline, Google Scholar
23. Losche G: Sensorimotor and action development in autistic children from infancy to early childhood. J Child Psychol Psychiatry 1990, 31:749–761Google Scholar
24. Stone WL, Hoffman EL, Lewis SE, Ousley OY: Early recognition of autism. Arch Pediatr Adolesc Med 1994; 148:174–179Crossref, Medline, Google Scholar
25. Sigman M, Ruskin E, Arbeile S, Corona R, Dissanayake C, Espinosa M, Kim N, Lopez A, Zierhut C: Continuity and change in the social competence of children with autism, Down syndrome, and developmental delays. Monogr Soc Res Child Dev 1999; 64:1–114Crossref, Medline, Google Scholar
26. Wetherby A, Prizant B, Hutchinson T: Communicative, social-affective, and symbolic profiles of young children with autism and pervasive developmental disorder. Am J Speech Lang Pathol 1998; 7:79–91Crossref, Google Scholar
27. Volkmar FR, Chawarska K, Klin A: Autism in infancy and early childhood. Annu Rev Psychol (in press)Google Scholar
28. Adrien JL, Lenoir P, Martineau J, Perrot A, Hameury L, Larmande C, Sauvage D: Blind ratings of early symptoms of autism based upon family home movies. J Am Acad Child Adolesc Psychiatry 1993; 32:617–626Crossref, Medline, Google Scholar
29. Baranek GT: Autism during infancy: a retrospective video analysis of sensory-motor and social behaviors at 9–12 months of age. J Autism Dev Disord 1999; 29:213–224Crossref, Medline, Google Scholar
30. Osterling J, Dawson G: Early recognition of children with autism: a study of first birthday home videotapes. J Autism Dev Disord 1994; 24:247–257Crossref, Medline, Google Scholar
31. Osterling J, Dawson G, Munson J: Early recognition of 1-year-old infants with autism spectrum disorder versus mental retardation. Dev Psychopathol 2002; 14:239–251Crossref, Medline, Google Scholar
32. Werner E, Dawson G, Osterling J, Dinno H: Recognition of autism spectrum disorder before one year of age: a retrospective study based on home videotapes. J Autism Dev Disord 2000; 30:157–162Crossref, Medline, Google Scholar
33. Adrien JL, Perrot A, Sauvage D, Leddet I, Larmande C, Hameury L, Barthelemy C: Early symptoms in autism from family home movies: evaluation and comparison between 1st and 2nd year of life using IBSE Scale. Acta Paedopsychiatr 1992; 55:71–75Medline, Google Scholar
34. Dawson G, Osterling J, Meltzoff AN, Kuhl P: Case study of the development of an infant with autism from birth to two years of age. J Appl Dev Psychol 2000; 21:299–313Crossref, Medline, Google Scholar
35. Gillberg C, Ehlers S, Schaumann H, Jakobsson G, Dahlgren SO, Lindblom R, Bagenholm A, Tjuus T, Blidner E: Autism under age 3 years: a clinical study of 28 cases referred for autistic symptoms in infancy. J Child Psychol Psychiatry 1990; 31:921–934Crossref, Medline, Google Scholar
36. Green G, Brennan LC, Fein D: Intensive behavioral treatment for a toddler at high risk for autism. Behav Modif 2002; 26:69–102Crossref, Medline, Google Scholar
37. Zeanah CH, Davis S, Silverman M: The question of autism in an atypical infant. Am J Psychother 1988; 42:135–150Crossref, Medline, Google Scholar
38. Chawarska K, Klin A, Volkmar FR: Automatic attention cueing through eye movement in 2-year-old children with autism. Child Dev 2003; 74:1108–1122Crossref, Medline, Google Scholar
39. Klin A, Jones W, Schultz R, Volkmar F: The enactive mind, or from actions to cognition: lessons from autism. Philos Trans R Soc Lond B Biol Sci 2003; 358:345–360Crossref, Medline, Google Scholar
40. Swettenham J, Baron-Cohen S, Charman T, Cox A, Baird G, Drew A, Rees L, Wheelwright S: The frequency and distribution of spontaneous attention shifts between social and nonsocial stimuli in autistic, typically developing, and nonautistic developmentally delayed infants. J Child Psychol Psychiatry 1998; 39:747–753Crossref, Medline, Google Scholar
41. Volkmar FR, Klin A, Siegel B, Szatmari P, Lord C, Campbell M, Freeman BJ, Cicchetti DV, Rutter M, Kline W: Field trial for autistic disorder in DSM-IV. Am J Psychiatry 1994; 151:1361–1367Link, Google Scholar
42. Klin A, Chawarska K, Rubin E, Volkmar FR: Clinical assessment of toddlers at risk of autism, in Handbook of Infant and Toddler Mental Health Assessment. Edited by DelCarmen-Wiggins R, Carter A. Oxford, UK, Oxford University Press, 2004, pp 311–336Google Scholar
43. Rogers SJ: Diagnosis of autism before the age of 3, in International Review of Research in Mental Retardation: Autism. Edited by Glidden LM. San Diego, Academic Press, 2001, pp 1–31Google Scholar
44. Chawarska K, Volkmar FR: Autism in infancy and early childhood, in Handbook of Autism and Pervasive Developmental Disorders, 3rd ed. Edited by Volkmar FR, Klin A, Paul R. New York, John Wiley & Sons (in press)Google Scholar
45. Mullen EM: Mullen Scales of Early Learning, AGS ed. Circle Pines, Minn, AGS Publishing, 1995Google Scholar
46. Fenson L: The MacArthur Communicative Development Inventory: Infant and Toddler Versions. San Diego, San Diego State University, 1989Google Scholar
47. Wetherby A, Prizant B: Communication and Symbolic Behavior Scales Developmental Profile (CSBS-DP). Baltimore, Paul H Brookes, 1993Google Scholar
48. Lord C, Rutter M, DiLavore PC, Risi S: Autism Diagnostic Observation Schedule Manual. Los Angeles, Western Psychological Services, 2001Google Scholar
49. Sparrow SS, Balla D, Cicchetti D: Vineland Adaptive Behavior Scales, expanded ed. Circle Pines, Minn, American Guidance Service, 1984Google Scholar
50. Lord C, LeCouteur A, Rutter M: Autism Diagnostic Interview Toddler Version. Chicago, University of Chicago, 1991Google Scholar
51. Carter A, Volkmar FR, Sparrow SS, Wang JJ, Lord C, Dawson G, Fombonne E, Loveland K, Mesibov G, Schopler E: The Vineland Adaptive Behavior Scales: supplementary norms for individuals with autism. J Autism Dev Disord 1998; 28:287–302Crossref, Medline, Google Scholar
52. Cox A, Klein K, Charman T, Baird G, Baron-Cohen S, Swettenham J, Drew A, Wheelwright S: Autism spectrum disorders at 20 and 42 months of age: stability of clinical and ADI-R diagnosis. J Child Psychol Psychiatry 1999; 40:719–732Crossref, Medline, Google Scholar
53. Siperstein R, Volkmar FR: Parental reporting of regression in children with pervasive developmental disorders. J Autism Dev Disord 2004; 34:731–734Crossref, Medline, Google Scholar
54. Richler J, Luyster R, Risi S, Wan-Ling H, Dawson G, Bernier R, Dunn M, Hyman SL, McMahon WM, Goudie J, Minshew N, Rogers S, Sigman M, Spence MA, Tager-Flusberg H, Volkmar FR, Lord C: Is there a regressive phenotype of autism spectrum disorder associated with the measles-mumps-rubella vaccine? a CPEA study. J Autism Dev Disord (in press)Google Scholar
55. Lenneberg EH: Biological Foundations of Language. New York, John Wiley & Sons, 1967Google Scholar
56. Lord C, Shulman C, DiLavore P: Regression and word loss in autism spectrum disorders. J Child Psychol Psychiatry 2004, 45:1–21Google Scholar
57. Courchesne E, Carper R, Akshoomoff N: Evidence of brain overgrowth in the first year of life in autism. JAMA 2003, 290:393–394Google Scholar
58. Laumonnier F, Bonnet-Brilhault F, Gomot M, Blanc R, David A, Moizard MP, Raynaud M, Ronce N, Lemonnier E, Calvas P, Laudier B, Chelly J, Fryns JP, Ropers HH, Hamel BC, Andres C, Barthelemy C, Moraine C, Briault S: X-linked mental retardation and autism are associated with a mutation in the NLGN4 gene, a member of the neuroligin family. Am J Hum Genet 2004; 74:552–557Crossref, Medline, Google Scholar



