Episodic Memory Performance Predicted by the 2bp Deletion in Exon 6 of the “Alpha 7-Like” Nicotinic Receptor Subunit Gene
Abstract
Objective: There is evidence of linkage between chromosome 15q14 and the P50 auditory evoked response, a heritable neuropsychological marker associated with increased risk of schizophrenia. Chromosome 15q14 harbors the alpha-7 nicotinic receptor subunit gene (CHRNA7) and a hybrid gene of unknown function (CHRFAM7A). CHRNA7 is involved in memory formation, a core dysfunction in schizophrenia. The authors set out to determine if this locus is associated with memory dysfunction in schizophrenia. Method: A 2bp deletion in exon 6 of CHRFAM7A, which disrupts the hybrid gene and has previously been associated with P50 deficit, was genotyped in 251 individuals from the Maudsley Family Study of schizophrenia. Episodic memory function was assessed using the Wechsler Memory Scale. Results: Significant associations were identified with delayed recall and percentage retained, with the presence of the deletion predicting worse performance. Conclusions: These observations indicate that episodic memory function is a schizophrenia endophenotype and implicate the CHRFAM7A/CHRNA7 locus in modulating its function.
Several linkage studies have implicated chromosome 15q13-q14 in schizophrenia (1 , 2) . Freedman et al. (3) used the P50 auditory evoked response, a measure of sensory gating, as a neurophysiological endophenotype for linkage analysis in schizophrenia families and identified linkage in close proximity to the CHRNA7 gene at 15q13-q14 (3) .
The CHRNA7 locus is complex, harboring a brain-expressed gene of unknown function (CHRFAM7A) about 1.5 Mb proximal to CHRNA7 itself. It consists of a duplication of exons 5–10 of CHRNA7, fused with five novel exons (4 , 5) . There is a common 2bp deletion in exon 6 of CHRFAM7A (exon6Δ2) that creates a premature stop codon and is hypothesized to cause exon 6 of the duplicated gene to be spliced out (6) . Raux et al. (7) found that at least one copy of the exon6Δ2 polymorphism was sufficient to predict a deficit in the P50 auditory evoked response, leading the authors to conclude that it may disrupt normal CHRNA7 function through a dominant negative effect.
Both human and animal work also suggest a function for CHRNA7 in more general aspects of learning and memory (8 , 9) and in the cellular process of long-term potentiation (10 , 11) . Working and episodic memory are traits that are impaired in patients with schizophrenia and their healthy relatives (12) , implying that they are core deficits and endophenotypes that may be useful for genetic studies (13) .
In the present study, we used subjects from the Maudsley Family Study to determine if the CHRFAM7A exon6Δ2 polymorphism would be associated with episodic memory (as measured by the Wechsler Memory Scale) and to test the suitability of episodic memory as an endophenotype for genetic studies of schizophrenia. We chose this approach because the CHRFAM7A locus has been implicated in schizophrenia, evidence supports a role for neural nicotinic receptors in memory formation, and episodic memory was a familial endophenotype in the Maudsley Family Study (12) .
Method
The study group consisted of 251 subjects (96 affected individuals, 116 unaffected relatives, and 39 healthy comparison subjects) and is described in detail elsewhere (12) . Genotyping was performed as described (7) ; since the genotyping assay cannot distinguish between individuals with one or two copies of exon6Δ2 because of high homology between the CHRFAM7A and the CHRNA7 genes, it was analyzed as a dominant allele. Memory function was assessed using the immediate/delayed recall and percentage retention portions of the Wechsler Memory Scale’s logical memory subtest.
The Wechsler Memory Scale measures were converted into z scores correcting for age, sex, and IQ. Regression analysis for the association between the neurocognitive measures and genotype was performed. Since the population comprised a large proportion of related individuals, linear regression was applied with robust standard errors using family ID as the cluster variable. We analyzed three measures of memory: immediate recall, delayed recall, and percentage retained; percentage retained is a more refined delayed measure that takes into consideration performance in immediate recall. We estimated the fraction of the variation (R 2 ) in the dependent variable accounted for by the exon6Δ2 polymorphism. Association between exon6Δ2 and schizophrenia was tested using the transmission disequilibrium test.
Results
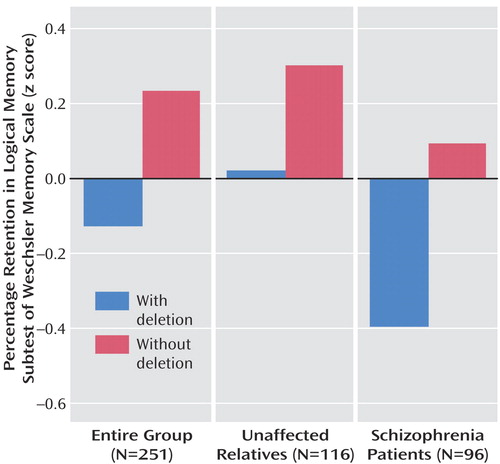
Consistent with previous work, no association was found with schizophrenia (exon6Δ2 frequency: 0.65 in schizophrenia group, 0.63 in unaffected relatives; transmission disequilibrium test: transmitted alleles=36, nontransmitted alleles=26 [χ 2 =0.94, df=1 p=0.20]). However, we found a significant relationship in the entire group between the presence of exon6Δ2 and both delayed recall (β=0.30, 95% CI=–0.28 to 0.60 [t=1.5, p<0.04; R 2 =0.02]) and percentage retained (β=–0.34, 95% CI=–0.58 to –0.10 [t=–2.82, p=0.006; R 2 =0.03]), with the absence of exon6Δ2 conferring superior performance ( Figure 1 ). No association was seen for immediate recall (β=0.10, 95% CI=–0.35 to 0.56 [t=0.47, p=0.64; R 2 =0.002]). When analyzed separately, a similar effect was found for the percentage retained measure in both the affected group (β=0.49, 95% CI=0.01 to 0.90 [t=2.02, p<0.05; R 2 =0.04]) and unaffected group (β=0.31, 95% CI=0.03 to 0.50 [t=2.22, p<0.03; R 2 =0.03]).
Discussion
We were able to ascertain that the presence of at least one copy of the exon6Δ2 polymorphism in the CHRFAM7A gene was associated with a deficit in episodic memory independently of disease status, indicating that this polymorphism is either associated with a true endophenotype of schizophrenia or is a general determinant of episodic memory in the population. Note that percentage retained scores are shifted downward slightly in the patient group. This is consistent with the memory measures in the study group, since schizophrenia patients on average perform worse than their unaffected relatives, as is expected for an endophenotype (12) . However the effect size is similar between the two groups.
Association between the deletion and episodic memory was seen in both delayed recall measures, both of which assess memory consolidation. No association was identified with immediate recall, which is a measure of the relative integrity of encoding of information. Thus, the presence of the exon6Δ2 polymorphism predicts a decrement in memory consolidation. The regression equation estimated that the amount of variance attributed to this genotype is 2%–3%. This is consistent with the general view that many genes of small effect contribute to variation in complex traits such as memory.
In summary, we have implicated the CHRFAM7A gene in regulation of episodic memory, which is associated with schizophrenia but appears to be independent of diagnosis, indicating it may be a true endophenotype. We hypothesize that either the full hybrid gene confers an advantage in episodic memory, which is lost when the reading frame is disrupted by exon6Δ2, or that the truncated CHRFAM7A protein has a dominant negative effect, perhaps on CHRNA7 function. Further work should concentrate on the ascertainment of the exact mechanism of the effect of this polymorphism and its relationship with both P50 and long-term episodic memory.
1. Riley BP, McGuffin P: Linkage and associated studies of schizophrenia. Am J Med Genet 2000; 97:23–44Google Scholar
2. Lewis CM, Levinson DF, Wise LH, DeLisi LE, Straub RE, Hovatta I, et al: Genome scan meta-analysis of schizophrenia and bipolar disorder, part II: schizophrenia. Am J Hum Genet 2003; 73:34–48Google Scholar
3. Freedman R, Coon H, Myles-Worsley M, Orr-Urtreger A, Olincy A, Davis A, et al: Linkage of a neurophysiological deficit in schizophrenia to a chromosome 15 locus. Proc Natl Acad Sci U S A 1997; 94:587–592Google Scholar
4. Gault J, Robinson M, Berger R, Drebing C, Logel J, Hopkins J, et al: Genomic organization and partial duplication of the human alpha7 neuronal nicotinic acetylcholine receptor gene (CHRNA7). Genomics 1998; 52:173–185Google Scholar
5. Riley B, Williamson M, Collier D, Wilkie H, Makoff A: A 3-Mb map of a large segmental duplication overlapping the alpha7-nicotinic acetylcholine receptor gene (CHRNA7) at human 15q13-q14. Genomics 2002; 79:197–209Google Scholar
6. Gault J, Hopkins J, Berger R, Drebing C, Logel J, Walton C, et al: Comparison of polymorphisms in the alpha7 nicotinic receptor gene and its partial duplication in schizophrenic and control subjects. Am J Med Genet 2003; 123B(1):39–49Google Scholar
7. Raux G, Bonnet-Brilhault F, Louchart S, Houy E, Gantier R, Levillain D, et al: The -2 bp deletion in exon 6 of the ‘alpha 7-like’ nicotinic receptor subunit gene is a risk factor for the P50 sensory gating deficit. Mol Psychiatry 2002; 7:1006–1011Google Scholar
8. Levin ED, Christopher NC, Weaver T, Moore J, Brucato F: Ventral hippocampal ibotenic acid lesions block chronic nicotine-induced spatial working memory improvement in rats. Brain Res Cogn Brain Res 1999; 7(3):405–410Google Scholar
9. Warburton DM, Rusted JM, Fowler J: A comparison of the attentional and consolidation hypotheses for the facilitation of memory by nicotine. Psychopharmacology (Berl) 1992; 108:443–447Google Scholar
10. Mansvelder HD, McGehee DS: Long-term potentiation of excitatory inputs to brain reward areas by nicotine. Neuron 2000; 27:349–357Google Scholar
11. Matsuyama S, Matsumoto A, Enomoto T, Nishizaki T: Activation of nicotinic acetylcholine receptors induces long-term potentiation in vivo in the intact mouse dentate gyrus. Eur J Neurosci 2000; 12:3741–3747Google Scholar
12. Toulopoulou T, Rabe-Hesketh S, King H, Murray RM, Morris RG: Episodic memory in schizophrenic patients and their relatives. Schizophr Res 2003; 63:261–271Google Scholar
13. Gottesman II, Gould TD: The endophenotype concept in psychiatry: etymology and strategic intentions. Am J Psychiatry 2003; 160:636–645Google Scholar



