Antidepressant-Resistant Depression in Patients With Comorbid Subclinical Hypothyroidism or High-Normal TSH Levels
Case 1: A 35-year-old woman with a history of previous depression responsive to imipramine at 150 mg/day presented with a second depressive episode that met criteria for major depressive disorder. Despite a trial of imipramine at 300 mg/day (the highest dosage she could tolerate), residual symptoms included low mood, lack of interest, loss of appetite, and initial insomnia. Since her first episode of depression 10 years earlier, the patient had been treated surgically for a thyroid adenoma and was taking levothyroxine at 125 μg/day. Her thyroid measures were all in the normal laboratory range (serum T4, 8.7 μg/dL; free T4, 1.2 ng/dL; free T3 index, 160; and TSH, 3.0 μIU/mL). Her endocrinologist noted no signs of overt hypothyroidism. On a repeat TSH assessment, the level was 3.2 μIU/mL, and while the endocrinologist pointed out that this value remained in the normal range, after some discussion, we agreed to try an increase in levothyroxine to 175 μg/day. The patient’s depressive symptoms decreased substantially within a week with all other medications unchanged, and she remained depression free, even after her imipramine dosage was reduced to 150 mg/day, which dramatically reduced her complaints of dry mouth, constipation, and light-headedness. Her TSH level at 2 weeks of treatment was 1.9, and at 6 months, 1.6. No signs of hyperthyroidism, such as agitation or increased pulse, were noted.
Case 2: A 52-year-old woman had experienced episodes of depression in college and after the birth of her second child. These episodes responded well to fluoxetine and, later, escitalopram at 10 mg/day. Escitalopram was continued as maintenance treatment at 10 mg/day. At age 46, she developed an autoimmune thyroiditis and consequent hypothyroidism that was treated with levothyroxine at 100 μg per day. Recurrence of depressive symptoms at age 50 was addressed by an increase in the patient’s escitalopram dosage to 20 mg/day and then by trials of venlafaxine to 225 mg/day and sertraline to 250 mg/day, each for at least 6 weeks, but all with poor response. Thyroid parameters included serum T4 values between 8.0 and 10.0 μg/dL on repeated measures and TSH values between 2.9 and 3.6 μIU/mL. When an increase in thyroid hormone supplementation to 150 μg/day was instituted, the patient recovered well over a period of several weeks, and TSH levels were 1.0 to 1.4 μIU/mL. She had no notable side effects, but she and her endocrinologist were interested in reducing the supplement dosage some months later. All other medications remained unchanged, but a decrease of levothyroxine to 125 μg/day, with an associated serum TSH level of 2.9 μIU/mL, was followed by an increase in depressive symptom severity. Remission was restored by resuming treatment with levothyroxine at 150 μg/day, which was associated with a serum TSH level of 1.3 μIU/mL.
Thyroid and mood disorders are both common and may co-occur in the same person, and psychiatrists must optimize treatment for patients with these comorbid disorders. In many cases, thyroid disorder is clear or preexisting. In other cases, thyroid dysfunction is not obvious and is suggested only by marginal abnormalities in thyroid biomarkers or poor response to standard antidepressant treatment. Optimal intervention requires knowledge of thyroid pathology and attention to the possibility of subtle thyroid dysfunction in patients with affective disorders.
The first case described above, from 35 years ago, alerted us to the possibility that some patients with normal thyroid test results might nonetheless have subtle thyroid dysfunction, enough to affect treatment for depression. Since then, we have treated many patients already receiving thyroid hormone supplementation for preexisting hypothyroidism, as well as patients without clear hypothyroidism, in whom, because of some evidence of thyroid dysfunction, we tried either levothyroxine (T4) or liothyronine (T3) to augment antidepressant treatment response. Most commonly, these patients had thyroid-stimulating hormone (TSH) levels in the high normal range and persistent depression, with low mood and low energy.
These cases raise the questions of when to treat, whether to prescribe T4 or T3, and how to monitor treatment. To address these questions, we review here some background issues, including recent evidence on the “normal range” for thyroid function tests, and cite evidence and make suggestions on treatment options when there is a question of mild thyroid dysfunction contributing to inadequate antidepressant response.
Thyroid Disorders and Monitoring of thyroid state
The thyroid gland produces the iodinated tyrosine-based hormones T4 (thyroxine) and T3 (triiodothyronine). Although more T4 is released, T4 is predominantly a prohormone. It is taken into cells by active transport and converted to T3, which subsequently interacts with proteins that include nuclear receptors to modulate gene expression and numerous aspects of metabolism, including energy metabolism. Production of T4 is determined by levels of TSH, released from the pituitary gland, in response to thyrotropin-releasing hormone (TRH), released from the hypothalamus. T4 and T3 are largely protein bound in serum, being carried by several proteins, including thyroid-binding globulin, transthyretin, and albumin (1). The hypothalamus and pituitary monitor serum thyroid hormone levels, a process that provides a feedback mechanism for modulating TRH and TSH release and maintaining adequate amounts of thyroid hormone. Entry of thyroid hormone into the brain is mostly through capillary blood, although it also occurs through CSF (1). As with serum carriers of thyroid hormone, there are numerous proteins that actively transport thyroid hormone into cells, metabolize them, and carry them to receptors (2, 3).
Both excessive thyroid activity (hyperthyroidism) and inadequate thyroid hormone (hypothyroidism) are common, with hypothyroidism often associated with symptoms of depression (4, 5). The most common causes of hypothyroidism are autoimmune disorders (Hashimoto’s thyroiditis) and inadequate thyroid hormone production due to surgical removal of thyroid adenomas. Both major depression and thyroid disease are two to three times as common in women as in men (6), and there is evidence suggesting that hormone supplementation may be more effective in women (7). In psychiatry, a common cause of hypothyroidism is lithium treatment, and thyroid hormone status is routinely monitored in patients receiving lithium. Smoking and race have minimal associations with thyroid function. Dopaminergic agents, glucocorticoids, and a few other medications can cause modest reductions in TSH release (8). Antidepressants may be associated with small decreases in T4 levels, although the meaning of that association, against a background of mild thyroid abnormalities in depression, is unclear (9).
There are simple blood tests and good interventions to document and modulate thyroid state. However, it is important to note that while there are standard or normal laboratory result ranges, thyroid dysfunction, like many medical disorders, is a matter of degree, and the standard range cutoffs are guidelines (10). For example, the normal high end of the TSH range is often quoted as 4.5 μIU/mL, but that does not necessarily mean that a patient is physiologically normal at 4.4 but abnormal at 4.6.
The management of overt thyroid disease is best handled by specialists. Evaluation involves multiple measures, including serum thyroid hormone levels (both free and bound levels), but the core of monitoring is of serum TSH levels (8). This is because TSH is a reflection of adequate thyroid hormone in the periphery and the brain, as monitored in the pituitary and hypothalamus, respectively. The normal range of TSH has been the subject of ongoing debate, and some authorities, based on recent evidence, now define the upper limit of optimal TSH as being well below the long-quoted standard of 4–4.5 μIU/mL (8, 11, 12). While most people (97.5%, the usual percentage included in a lab range) have TSH levels below 4 or 4.5, in subjects screened for thyroid health (and having no evidence of thyroid antibodies), some 90%−95% of people have TSH levels below 2.5 (8, 11). In essence, the distribution of TSH levels in a healthy sample is not centered within the lab normal range (Figure 1); it is skewed, with a large peak around 1.5 (1.43 for men and 1.67 for women) (11). That is, most people have TSH levels toward the bottom of the TSH range (0.5–2.5), and there is a long “tail” of TSH values with a relatively small proportion of people with levels above 2.5 (8, 11). Therefore, a TSH level above 2.5 is not physiologically optimal or normal for the vast majority of people. In fact, those with high-normal TSH levels compared with those with levels below 2.5 may actually have mild hypothyroidism, prodromal to overt hypothyroidism in ensuing years (11, 13). These recent reassessments (13) suggest that TSH levels above the peak may represent “occult thyroid disease,” and current reviews suggest that the target for TSH with supplementation should be below 2.5 (14, 15) or even below 2 (10).
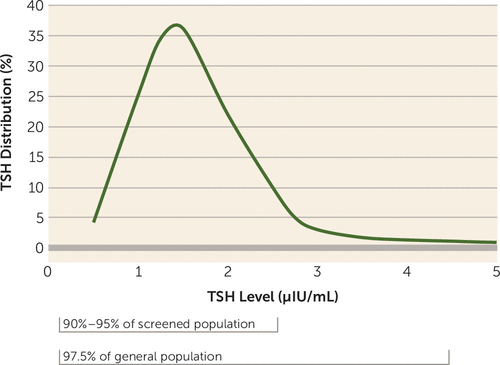
FIGURE 1. Distribution of TSH Levels in the Populationa
a The graph is a smoothed curve, combining the results of several studies (8, 11, 13), illustrating the distribution of laboratory values of TSH in an unselected (general) population. The curve is skewed, with a “tail” to the right and peaks at a TSH level of 1.5 μIU/mL. Laboratory normal limits are defined as including 97.5% of the population, which contains all values of TSH below approximately 4.5 μIU/mL. In a population screened to eliminate anyone with evidence of thyroid disease or elevated levels of thyroid antibodies, 90%−95% of TSH values are below 2.5 μIU/mL.
The benefits of thyroid supplementation in people with minimal elevations of TSH level (above 2 or 2.5 μIU/mL) have not been fully explored. Studies of subclinical hypothyroidism (defined as a T4 level in the normal range but a TSH level above 4–4.5 μIU/mL) are more common, and generally they have observed no short-term benefit of treatment in most patients with elevated TSH levels (16). However, the studies to date include heterogeneous populations in which most study subjects have no or minimal complaints, including any symptoms of affective disorders. In addition, supplementation was conservative, with additional thyroid hormone given at very modest dosages, so that TSH level was often only minimally reduced (17). Given the absence of complaints and such conservative treatment, benefits would not be expected. By comparison, several studies have suggested that there are short-term benefits from thyroid supplementation in patients with symptoms, often including complaints as simple as fatigue or low mood (14, 18, 19). In addition, the current consensus is that treatment of subclinical hypothyroidism improves lipid profile and the long-term risk of stroke and cardiovascular disease (14, 19).
Thyroid State and Mood Disorders
Thyroid hormones affect most aspects of cell development and function (20, 21), so it is not surprising that there are documented relationships between thyroid state and psychiatric disorders. Specifically, thyroid disorders may increase the risk of mood disorders, and the rate of depression is modestly elevated in those with hypothyroidism (4, 5). Also, when overt hypothyroidism is treated with thyroid supplements, symptoms of low mood often clear along with other features of thyroid hormone inadequacy (22). However, despite the general effects of thyroid hormones on most body processes and despite some overlap in symptoms, such as changes in energy and appetite shared by thyroid and mood disorders, most patients with major depression show evidence of neither overt nor subclinical hypothyroidism (23).
While depression is not strongly associated with overt thyroid disorder, it is associated with complex and subtle thyroid function irregularities, including modestly elevated TSH levels, modestly elevated T4 levels, a blunted TSH response to TRH, and altered circadian release of thyroid hormone (24). However, similar abnormalities of thyroid regulation have been observed in many illnesses as well as in response to stress (7, 25), and many hormones and processes, not just thyroid function, show circadian abnormalities in major depression. Thus, these thyroid alterations may be nonspecific, not causally or crucially related to the pathophysiology of depression. Immune system abnormalities have been observed both in thyroid and depressive disorders (26), but although the most common cause of thyroid disorders is an autoimmune disorder, dysregulated immune function, not autoimmunity, is more clearly documented in depression, as noted above. The relationships among immune, thyroid, and mood disorders remain matters for further research.
Overall, the evidence does not suggest that thyroid dysfunction underlies most depression or that substantial thyroid dysfunction is present in typical patients with major depression. Nonetheless, even a TSH level in the upper quartile of normal has been reported to be associated with a higher frequency of depressive episodes, more severe symptoms, and poorer response to treatment in people with major depression (27). Pae et al. (28) reported less severe but more treatment-resistant depression in euthyroid perimenopausal women with high-normal TSH levels. Corruble et al. (29) observed poorer antidepressant response, especially to selective serotonin reuptake inhibitors, in patients with major depression and higher TSH levels, with no correlation of response to serum T3 or T4 levels. Not all studies have found associations between TSH level and response (30), but given relatively small numbers of patients available for evaluation in any clinic, none of these studies has been large enough to be definitive.
In assessing thyroid state, there has been some debate as to whether serum TSH level always accurately reflects brain thyroid hormone levels, as transporters vary somewhat among different tissues and even in different cells within tissues, such as glia and neurons (31, 32). However, there are numerous uptake proteins, providing compensatory mechanisms for transport (32). Known abnormalities of brain uptake with clinically documented consequences are rare—for example, genetic disorders associated with peripheral and brain anomalies and with developmental disorders and substantial disability (32, 33). Of note in interpreting TSH levels is that uptake of thyroid hormone is best in the pituitary, which has the most active transporters (31). If thyroid hormone levels in the pituitary are low and TSH level consequently is high, levels are likely to be even more inadequate in brain. Thus, a high serum TSH level requires serious consideration for treatment.
TSH measures vary by time of day, from day to day, and from lab to lab, making it important to obtain repeated measurements to monitor supplementation (12, 13). Thyroid hormone supplementation that is adequate to relieve most signs and symptoms of hypothyroidism may not be adequate for treatment of patients with depression. A well-powered study (with 13,000 subjects enrolled) assessing this issue (34) observed a correlation between elevated TSH levels and depression, even with TSH levels within the usual laboratory range. Specifically, the depression rate was twice as high in women with TSH levels above 2.3 μIU/mL, again suggesting that the optimal TSH level is well below 4 μIU/mL.
The second case vignette presented above, a recent case, in which “on/off” treatment was prescribed, illustrates that for some patients, titrating thyroid hormone to a TSH level below 2.5 μIU/mL may be crucial in amelioration of symptoms.
Patients Without Diagnosed Thyroid Disease
In patients with thyroid dysfunction who are already receiving thyroid hormone supplementation, the task is to determine the adequacy of that treatment. The more difficult case is that of patients without diagnosed thyroid disease but with evidence of mild thyroid dysfunction. For the psychiatrist treating a patient with unremitted depression and a TSH level above 2.5 μIU/mL, the question arises as to whether or not to prescribe thyroid hormone, particularly if there is no history of frank thyroid disease and if the patient is not already being treated with supplementation. The literature on subclinical hypothyroidism partly addresses such cases (see reference 17), and there is evidence that a TSH level in the high normal range is specifically associated with poorer antidepressant response (35, 36).
Optimizing TSH level and prescribing thyroid hormone supplementation may apply in treating bipolar as well as unipolar depression (37, 38). High-normal TSH levels may be associated with poor response in patients with bipolar disorder, and supplementation may be beneficial, just as in major depressive disorder.
Clinical Intervention: Choice, Dosage, and Monitoring of thyroid Hormone Supplementation
There is a large literature dating back to the 1980s on the use of thyroid hormone supplementation in depression, either given alone or as augmentation to an antidepressant medication, even when there is no clear evidence of thyroid dysfunction. The results of those studies are reviewed in detail in Lasser and Baldessarini (7) and Jonklass et al. (25). Most studies suggest only modest benefit from thyroid hormone supplements given as the primary treatment for depression or even as augmentation of standard antidepressant treatment in patients with depression in the absence of thyroid dysfunction. However, some studies suggest that supplementation with either T4 (39) or T3 (40, 41) may exert effects comparable to lithium as augmentation for antidepressants (39, 40). Unfortunately, these studies have been too few and too small to be definitive (42).
By comparison, studies of thyroid hormone supplementation in patients with documented thyroid abnormalities show consistently positive effects (25). Questions have arisen about whether T3 or T4 might be preferable as thyroid supplementation in depressed patients. Most often, endocrinologists prescribe T4 (usually its synthetic version, levothyroxine, because supplements prepared from natural sources show high variability) (43), and studies have shown no advantages for T3 (also its synthetic version, liothyronine) (14). However, in the depression literature, there are more studies of T3. T3 may enter and equilibrate in tissue more rapidly than T4, and in one small study comparing the effects of T3 and T4, the results favored T3, although patients in each group appeared to respond to the hormone they received (44). The brain preferentially takes up T4, and Joffee (45) suggested that T3 supplementation may decrease serum T4 by feedback inhibition in the hypothalamus, thus decreasing brain T3. No confirmatory studies have been conducted to test this hypothesis. Notably, a study by Targum et al. (46) comparing T4 and T3 showed similar beneficial effects with either of these supplements.
In a related matter, it has been argued that abnormalities of transport or conversion of T4 to T3 may produce hypothyroidism only in the brain and not in peripheral tissues (47), but no evidence supports this speculation for depression. Uptake of T4 and T3 occurs by multiple mechanisms, and none of the transport or metabolic mechanisms is unique to the brain. Either T4 or T3 supplementation produces adequate active T3 in the brain, and variants of transport or metabolism of thyroid hormone leading to disease are rare and are not likely to produce specific effects on mood (48).
Thus, to date, few studies comparing T3 and T4 have been conducted, none of them adequately powered, and no compelling clinical or physiologic evidence is available to favor T3 or T4 as a treatment (25, 42). T3 is absorbed faster, but this can be a disadvantage, as this supplement may have to be taken more than once a day, and rapid absorption can lead to mild and transient hyperthyroidism after some doses. Moreover, thyroid hormone often must be taken a half hour before meals to avoid fluctuations in blood levels, making multiple doses per day unwieldy (49). There are no clinical features or markers that might be used to choose a particular thyroid hormone supplement (50). T4, as the most commonly prescribed supplement in internal medicine, is a reasonable choice. On the other hand, because most depression studies of thyroid hormone supplementation have used T3, it is also reasonable to start with that. If a patient is already receiving either T4 or T3, increasing the dosage of that supplement is the most straightforward choice. Although some clinicians have prescribed T3 for patients already receiving T4, there are reports but no definitive trials supporting that approach (51).
There are other clinical considerations in the prescription of thyroid hormone. If a patient has switched brands or preparations of thyroid supplement, lowered or increased tissue levels and effects may ensue (14). If a patient taking an increased dosage of T4 experiences no amelioration of depressive symptoms, a trial or addition of T3 is reasonable. Low dosages, starting with 50–100 μg/day for T4 (levothyroxine) and 25–50 μg/day for T3 (liothyronine) usually suffice (52), but it is important to track symptoms, side effects, and serum TSH levels.
Elevated thyroid hormone levels have been associated with a variety of side effects, including increased pulse rate, decreased appetite and sleep, gastrointestinal hyperactivity, and agitation, but at the dosages of thyroid hormone used to augment antidepressants, these side effects are usually mild at worst. The most common worrisome side effect of thyroid hormone treatment is atrial fibrillation, but that is rare and is usually seen only if serum TSH is driven very low, below 0.1 μIU/mL (14). Therefore, TSH level should be driven below 2 μIU/mL, unless side effects, such as agitation, intervene. In our own practices, unless response is seen at lower dosages, we often attempt to achieve enough thyroid hormone supplementation to drive the TSH to 1.0 μIU/mL or just below, in order to give supplementation an adequate trial. Beneficial effects often appear early, sometimes within a week, but an adequate trial requires 6 weeks, and full effects may require months (14).
Special Populations
Prescribing thyroid hormone to patients with high-normal TSH levels is not controversial in the pregnant population. Adequate thyroid hormone is essential for development of the fetus, and to the mother, who may have an increased risk of postpartum depression if the TSH level is in the high normal range (13, 53). In each of these cases, that is, throughout pregnancy and into the postpartum period, the recommended range for TSH level is below 2.5 μIU/mL, rather than below 4 or 4.5 μIU/mL (54, 55).
In the past, older patients with subclinical hypothyroidism and no overt endocrinologic symptoms were often not treated with thyroid hormone supplementation. In part, this was because average TSH level increases with age (12, 56), and such increases were interpreted as “normal.” However, that these changes happen on average does not mean that such increases are physiologically beneficial and that the target TSH level in older patients should be higher than in younger patients. In fact, not only TSH, but also antithyroid antibodies increase with age (8, 57). Most recent reviews suggest that even past age 80, the majority of healthy people have TSH levels below 2.5 μIU/mL and that the rise in TSH levels seen with age is an indication of thyroid dysfunction and should be evaluated for treatment. As in younger populations, properly monitored thyroid hormone supplementation in elderly patients is relatively safe (14).
Summary and Conclusions
Adequate thyroid hormone levels are necessary for most physiologic functions, including normal brain function and response to medications. Unfortunately, the literature is sparse on the specific topic of treating patients who have both high-normal TSH levels, suggesting some degree of thyroid inadequacy, and major depressive disorder. Most studies addressing the use of thyroid hormone supplementation in patients with mild thyroid dysfunction and depression have been small, often called “pilot” and “preliminary” studies by their authors. Such studies have not been followed by large-scale definitive studies, and despite the clinical importance of defining adequate thyroid function in refractory depression, it is unlikely that definitive large-scale studies will be conducted.
Likewise, the cases we cited here are not proof of best treatment, but rather they illustrate the clinical importance of optimizing TSH level in a manner consistent with the evidence summarized from the literature. Ultimately, one must make choices when standard treatment is not adequately effective.
Some conclusions can be drawn from the literature and clinical experience. Overt thyroid disease or subclinical hypothyroidism is not the cause of most depression, and thyroid hormone supplementation alone is not an effective treatment for most cases of depression. However, mild thyroid abnormalities or inadequate thyroid hormone supplementation are not rare in patients with depression, and a target TSH level below 4.5 μIU/mL, as previously recommended in the literature, would appear to be inadequate for most people, especially those with treatment-resistant depression. In monitoring supplementation, both blood tests and side effects are followed, but it is likely that the TSH level will need to be below 2.5 μIU/mL, or even below 2 μIU/mL, for most patients to achieve optimal outcomes.
The literature strongly suggests that patients with high-normal TSH levels or subclinical hypothyroidism and clinical complaints, including depression, benefit from additional thyroid hormone. Fortunately, thyroid supplementation comes with few side effects and can be adjusted easily, so the recommendation of increased supplementation in instances of high-normal TSH levels and poor antidepressant response is a low-risk intervention.
Guidelines for the evaluation and treatment of patients with known or suspected thyroid dysfunction and depression are summarized in Figure 2. Rosenthal et al. (58) have also provided useful considerations previously in these pages. Readers should note that the TSH levels we have quoted here are the most common laboratory ranges, but some laboratories may use different assays and have different normal ranges. In any case, results at the higher end of the laboratory normal range may indicate suboptimal thyroid hormone levels in the patient tested (12).

FIGURE 2. Guidelines for the Recognition and Treatment of Thyroid Inadequacy in Major Depression With Poor Antidepressant Response
1 : Transport of thyroid hormone in brain. Front Endocrinol (Lausanne) 2014; 5:98Crossref, Medline, Google Scholar
2 : Thyroid hormone transporters in the brain. Rev Neurosci 2010; 21:173–186Crossref, Medline, Google Scholar
3 : Transport of thyroid hormones via the choroid plexus into the brain: the roles of transthyretin and thyroid hormone transmembrane transporters. Front Neurosci 2015; 9:66Crossref, Medline, Google Scholar
4 : Psychiatric and cognitive manifestations of hypothyroidism. Curr Opin Endocrinol Diabetes Obes 2014; 21:377–383Crossref, Medline, Google Scholar
5 : Depression, anxiety, health-related quality of life, and disability in patients with overt and subclinical thyroid dysfunction. Arch Med Res 2006; 37:133–139Crossref, Medline, Google Scholar
6 : The association between depression and thyroid disorders in a regionally representative Canadian sample. Psychol Health Med 2012; 17:335–345Crossref, Medline, Google Scholar
7 : Thyroid hormones in depressive disorders: a reappraisal of clinical utility. Harv Rev Psychiatry 1997; 4:291–305Crossref, Medline, Google Scholar
8 : Laboratory medicine practice guidelines: laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid 2003; 13:3–126Crossref, Medline, Google Scholar
9 : Thyroid hormones, the brain, and affective disorders. Crit Rev Neurobiol 1994; 8:45–63Medline, Google Scholar
10 : Neuropsychiatric aspects of hypothyroidism and treatment reversibility. Minerva Endocrinol 2007; 32:49–65Medline, Google Scholar
11 : Thyrotropin levels in a population with no clinical, autoantibody, or ultrasonographic evidence of thyroid disease: implications for the diagnosis of subclinical hypothyroidism. J Clin Endocrinol Metab 2008; 93:1224–1230Crossref, Medline, Google Scholar
12 : Normal limits for serum thyrotropin vary greatly depending on method. Clin Endocrinol (Oxf) 2016; 85:110–115Crossref, Medline, Google Scholar
13 : Biologic variation is important for interpretation of thyroid function tests. Thyroid 2003; 13:1069–1078Crossref, Medline, Google Scholar
14 : Subclinical hypothyroidism. N Engl J Med 2017; 376:2556–2565Crossref, Medline, Google Scholar
15 : 2013 ETA Guideline: management of subclinical hypothyroidism. Eur Thyroid J 2013; 2:215–228Crossref, Medline, Google Scholar
16 : Thyroid hormone therapy for older adults with subclinical hypothyroidism. N Engl J Med 2017; 376:2534–2544Crossref, Medline, Google Scholar
17 : A 6-month randomized trial of thyroxine treatment in women with mild subclinical hypothyroidism. Am J Med 2002; 112:348–354Crossref, Medline, Google Scholar
18 : Major depression and subclinical (grade 2) hypothyroidism. Psychoneuroendocrinology 1992; 17:215–221Crossref, Medline, Google Scholar
19 : The beneficial effect of l-thyroxine on cardiovascular risk factors, endothelial function, and quality of life in subclinical hypothyroidism: randomized, crossover trial. J Clin Endocrinol Metab 2007; 92:1715–1723Crossref, Medline, Google Scholar
20 : Thyroid hormone receptors, cell growth, and differentiation. Biochim Biophys Acta 2013; 1830:3908–3916Crossref, Medline, Google Scholar
21 : Role of the type 2 iodothyronine deiodinase (D2) in the control of thyroid hormone signaling. Biochim Biophys Acta 2013; 1830:3956–3964Crossref, Medline, Google Scholar
22 : Disease-specific as well as generic quality of life is widely impacted in autoimmune hypothyroidism and improves during the first six months of levothyroxine therapy. PLoS One 2016; 11:e0156925Crossref, Medline, Google Scholar
23 : Hypothyroidism and hyperthyroidism in major depression revisited. J Clin Psychiatry 1995; 56:186–192Medline, Google Scholar
24 : Chronobiological hypothalamic-pituitary-thyroid axis status and antidepressant outcome in major depression. Psychoneuroendocrinology 2015; 59:71–80Crossref, Medline, Google Scholar
25 : Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid 2014; 24:1670–1751Crossref, Medline, Google Scholar
26 : A bidirectional relationship between depression and the autoimmune disorders: new perspectives from the National Child Development Study. PLoS One 2017; 12:e0173015Crossref, Medline, Google Scholar
27 : Serum thyroid-stimulating-hormone concentration as an index of severity of major depression. Int J Neuropsychopharmacol 1999; 2:105–110Crossref, Medline, Google Scholar
28 : Thyroid hormones affect recovery from depression during antidepressant treatment. Psychiatry Clin Neurosci 2009; 63:305–313Crossref, Medline, Google Scholar
29 : Should major depression with “high normal” thyroid-stimulating hormone be treated preferentially with tricyclics? Neuropsychobiology 2004; 50:144–146Crossref, Medline, Google Scholar
30 : Peripheral thyroid hormone levels in treatment resistant depression. Biol Psychiatry 1999; 45:1053–1055Crossref, Medline, Google Scholar
31 : The kinetics of thyroid hormone transporters and their role in non-thyroidal illness and starvation. Best Pract Res Clin Endocrinol Metab 2007; 21:323–338Crossref, Medline, Google Scholar
32 : Function of thyroid hormone transporters in the central nervous system. Biochim Biophys Acta 2013; 2013:3965–3973Crossref, Google Scholar
33 : Resistance to thyroid hormone mediated by defective thyroid hormone receptor alpha. Biochim Biophys Acta 2013; 2013:4004–4008Crossref, Google Scholar
34 : Relationship between thyroid-stimulating hormone levels and risk of depression among the general population with normal T4 levels. Psychoneuroendocrinology 2015; 58:114–119 Crossref, Medline, Google Scholar
35 : Occult thyroid dysfunction in patients with refractory depression. Am J Psychiatry 1988; 145:1012–1014Link, Google Scholar
36 : Subclinical hypothyroidism: a modifiable risk factor for depression? Am J Psychiatry 1993; 150:508–510Link, Google Scholar
37 : Slower treatment response in bipolar depression predicted by lower pretreatment thyroid function. Am J Psychiatry 2002; 159:116–121Link, Google Scholar
38 : Thyroid functions and bipolar affective disorder. J Thyroid Res 2011; 2011:306367Crossref, Medline, Google Scholar
39 : Should thyroid augmentation precede lithium augmentation? A pilot study. J Affect Disord 1998; 49:235–239Crossref, Medline, Google Scholar
40 : T3 augmentation of SSRI resistant depression. J Affect Disord 2006; 91:211–215Crossref, Medline, Google Scholar
41 : A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: a STAR*D report. Am J Psychiatry 2006; 163:1519–1530Link, Google Scholar
42 : Hormone treatment of depression. Dialogues Clin Neurosci 2011; 13:127–138Medline, Google Scholar
43 : Fine adjustment of thyroxine replacement dosage: comparison of the thyrotrophin releasing hormone test using a sensitive thyrotrophin assay with measurement of free thyroid hormones and clinical assessment. Clin Endocrinol (Oxf) 1988; 28:325–333Crossref, Medline, Google Scholar
44 : A comparison of triiodothyronine and thyroxine in the potentiation of tricyclic antidepressants. Psychiatry Res 1990; 32:241–251Crossref, Medline, Google Scholar
45 : A perspective on the thyroid and depression. Can J Psychiatry 1990; 35:754–758Crossref, Medline, Google Scholar
46 : Thyroid hormone and the TRH stimulation test in refractory depression. J Clin Psychiatry 1984; 45:345–346Medline, Google Scholar
47 : Does thyroid hormone have a role as adjunctive therapy in depression? Thyroid 1996; 6:63–67Crossref, Medline, Google Scholar
48 : The syndromes of reduced sensitivity to thyroid hormone. Biochim Biophys Acta 2013; 1830:3987–4003Crossref, Medline, Google Scholar
49 : Timing of levothyroxine administration affects serum thyrotropin concentration. J Clin Endocrinol Metab 2009; 94:3905–3912Crossref, Medline, Google Scholar
50 : The use of thyroid supplements to augment antidepressant medication. J Clin Psychiatry 1998; 59:26–29Medline, Google Scholar
51 : T3 augmentation of antidepressant treatment in T4-replaced thyroid patients. J Clin Psychiatry 1992; 53:16–18Medline, Google Scholar
52 : [Potentializing of tricyclics and serotoninergics by thyroid hormones in resistant depressive disorders]. Encephale 2004; 30:267–275 (French)Crossref, Medline, Google Scholar
53 : Thyroid function tests at delivery and risk for postpartum depressive symptoms. Psychoneuroendocrinology 2013; 38:1007–1013Crossref, Medline, Google Scholar
54 : Subclinical thyroid disease: scientific review and guidelines for diagnosis and management. JAMA 2004; 291:228–238Crossref, Medline, Google Scholar
55 : TSH ≥2.5 mIU/L is associated with the increased oxidative damage to membrane lipids in women of childbearing age with normal thyroid tests. Horm Metab Res 2017; 49:321–326Crossref, Medline, Google Scholar
56 : Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab 2007; 92:4575–4582Crossref, Medline, Google Scholar
57 : Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 2002; 87:489–499Crossref, Medline, Google Scholar
58 : T3 augmentation in major depressive disorder: safety considerations. Am J Psychiatry 2011; 168:1035–1040Link, Google Scholar



