Computerized Cognitive Training in Older Adults With Mild Cognitive Impairment or Dementia: A Systematic Review and Meta-Analysis
Abstract
Objective:
Previous meta-analyses indicate that computerized cognitive training (CCT) is a safe and efficacious intervention for cognition in older adults. However, efficacy varies across populations and cognitive domains, and little is known about the efficacy of CCT in people with mild cognitive impairment or dementia.
Method:
The authors searched Medline, Embase, PsychINFO, CINAHL, and CENTRAL through July 1, 2016, for randomized controlled trials of CCT in older adults with mild cognitive impairment or dementia. Overall cognition, individual cognitive domains, psychosocial function, and activities of daily living were pooled separately for mild cognitive impairment and dementia trials.
Results:
The overall effect on cognition in mild cognitive impairment across 17 trials was moderate (Hedges’ g=0.35, 95% CI=0.20–0.51). There was no evidence of publication bias or difference between active- and passive-controlled trials. Small to moderate effects were found for global cognition, attention, working memory, learning, and memory, with the exception of nonverbal memory, and for psychosocial functioning, including depressive symptoms. In dementia, statistically significant effects were found on overall cognition (k=11, g=0.26, 95% CI=0.01–0.52) and visuospatial skills, but these were driven by three trials of virtual reality or Nintendo Wii.
Conclusions:
CCT is efficacious on global cognition, select cognitive domains, and psychosocial functioning in people with mild cognitive impairment. This intervention therefore warrants longer-term and larger-scale trials to examine effects on conversion to dementia. Conversely, evidence for efficacy in people with dementia is weak and limited to trials of immersive technologies.
Dementia is a progressive neurocognitive disorder characterized by insidious cognitive and functional decline until death. At present, the global prevalence of dementia is estimated at 5%−7% of people over 60 years (1). Mild cognitive impairment often precedes dementia and is characterized by largely intact everyday function despite objective evidence of cognitive decline (2). Mild cognitive impairment is a proximal risk factor for dementia (3), falls (4), and higher health expenditure (5), and risk increases proportionally with the number of impaired cognitive domains and symptom severity (3).
Conversion from mild cognitive impairment to dementia can be conservatively estimated at 5%−10% per year (3, 6, 7), and similar rates have been observed in the opposite direction (i.e., reversion from mild cognitive impairment to normal cognition) (7–9). Thus, mild cognitive impairment is an unstable cognitive state with potential to avert progression to dementia and attendant health and societal sequelae. To date, there is no systematic evidence for the effectiveness of any intervention on the cognitive and psychological symptoms of mild cognitive impairment (10). The current preferred medical treatment, cholinesterase inhibitors, only offer modest short-term cognitive benefits, and their clinical value continues to be debated given the risk of adverse events in clinical trials (11).
Computerized cognitive training (CCT) has generated considerable attention as a safe, relatively inexpensive and scalable intervention that aims to maintain cognition in older adults. CCT involves guided drill-and-practice on standardized tasks designed to load on specific cognitive processes, typically without explicit teaching of memory or problem-solving strategies, which distinguish CCT from other approaches for cognitive remediation (12). CCT can target single or multiple domains and usually adapts task difficulty to individual performance. Recent meta-analyses of randomized controlled trials of CCT have found moderate effect sizes on cognition in healthy older adults (13) and in Parkinson’s disease (14), as well as on symptom severity, daily functioning, and cognition in major depression (15).
While CCT is a frequent intervention in primary prevention trials (16), the extent to which CCT can benefit cognition in already diagnosed mild cognitive impairment or dementia is unclear. Systematic reviews of cognitive interventions in mild cognitive impairment or dementia have reported mixed results (17–25), but these interventions combined CCT with non-CCT interventions, such as cognitive stimulation or individual rehabilitation strategies, and mixed randomized controlled trials with other designs. We therefore aimed to conduct separate systematic reviews and meta-analyses of narrowly defined CCT in individuals with mild cognitive impairment or dementia in order to chart potential benefits on cognition and behavior across domains and diagnostic groups.
Method
This work adheres with PRISMA [Preferred Reporting Items for Systematic Reviews and Meta-Analyses] guidelines (26), was prospectively registered with PROSPERO (CRD42015023679), and follows our published methods for meta-analysis of CCT in older adults (13, 14).
Information Sources and Study Selection
We searched Medline, Embase, PsychINFO, CINAHL, and CENTRAL from inception to July 1, 2016 for randomized controlled trials examining the effects of CCT on one or more cognitive or behavioral outcomes in older adults with mild cognitive impairment or dementia (for the full search strategy, see Table S1 in the data supplement accompanying the online version of this article). We did not apply database limits, and non-English articles were translated. Additional articles were obtained by scanning reference lists of included studies and previous reviews. One reviewer (N.T.M.H. or V.L.C.) conducted initial eligibility screening based on title and abstract, followed by assessment of full-text versions by two independent reviewers (N.T.M.H., V.L.C., or an additional reviewer [see the Acknowledgments]). Disagreements were resolved by a senior reviewer (A.L.), who approved the final list of included studies. When eligibility was unclear, one reviewer (N.T.M.H.) contacted authors for additional information.
Eligibility Criteria
Types of participants.
The mean age of participants was ≥60 years old, with a diagnosis of mild cognitive impairment or dementia (of any etiology), confirmed by examining the inclusion criteria or baseline scores against standardized diagnostic criteria (2, 27).
Types of interventions.
At least 4 hours of drill and practice, with a clear cognitive rationale, videogames, or virtual reality, had to be completed. Studies combining CCT with other interventions were eligible if the control group received the same adjacent intervention. Studies were excluded if less than 50% of the cognitive intervention was CCT or not involving interaction with a computer (e.g., merely watching stimuli).
Types of controls.
Passive (no-contact, wait-list), active (e.g., sham CCT, psychoeducation), or pencil-and-paper cognitive training was required. Physical exercise as a sole control condition was excluded.
Types of outcomes.
Outcomes were change from baseline to posttraining in nontrained measures of cognition (global cognition, verbal or nonverbal learning, verbal or nonverbal memory, working memory, processing speed, attention, language, visuospatial skills, and executive function); activities of daily living; instrumental activities of daily living; or psychosocial functioning (neuropsychiatric symptoms, quality of life, and depression). All eligible outcomes per study and domain were included. Index scores were excluded if subdomain scores were available.
Data Collection and Coding
Coding of outcomes into cognitive domains and effect direction were performed independently by two reviewers (N.T.M.H. and L.M.) according to accepted neuropsychological categorization (28) or by consulting with a senior reviewer (A.L.) (for categorization of outcomes by domains, see Table S2 in the online data supplement). Outcomes were recorded as mean and standard deviations for each group at baseline and follow-up with the exception of standardized mean difference and 95% confidence interval (29) or mean change and standard deviation (30).
When studies included mixed cohorts, we asked primary authors for split data by diagnosis and group. Three studies for which split data were not available were coded as dementia according to baseline indications of functional impairment in >50% of the sample (31–33).
Risk of Bias in Individual Studies and Quality Appraisal
The Cochrane Collaboration’s risk of bias tool (34) was used to assess risk of bias in individual studies. Studies with high or unclear risk of bias for the blinding of assessors or incomplete outcome data categories were considered as high risk of bias. Methodological quality within studies was further assessed using the PEDro-P scale [Physiotherapy Evidence Database Rating Scale] (35). The original scale consists of 11 items. However, blinding of therapists and patients was not assessed due to nonfeasibility in CCT trials, and thus the maximum obtainable score (reflecting higher quality) was 9. Assessments were conducted by two independent reviewers (N.T.M.H., V.L.C. or an additional reviewer [see Acknowledgments]). A senior reviewer (A.L.) established consensus scores and resolved disagreements.
Data Analysis
We calculated standardized mean differences as Hedges’ g and 95% confidence interval of change in outcome measures between the CCT and control groups from baseline to posttraining and each follow-up. A positive standardized mean difference indicates a therapeutic effect of CCT over and above the control. Pooling of standardized mean differences across studies was performed using a random-effects model. Analogous to Cohen’s d (36), Hedges’ g estimates of <0.30, ≥0.30 and <0.60, and ≥0.60 were considered small, moderate, and large, respectively. Analyses were performed for overall cognitive outcomes, as well as for each cognitive or behavioral domain separately. When studies provided more than one outcome per domain for analysis, their standardized mean difference and variance were combined into a single study-level estimate. Finally, standardized mean differences from each arm (CCT and control) were split at the study level and pooled across studies in order to investigate nonspecific effects among control groups and likewise to investigate whether CCT genuinely enhances cognition.
Heterogeneity across studies was quantified using the I2 statistic, considered as low, moderate, or large when at 25%, 50%, or 75%, respectively (37). Small study effect (publication bias) was assessed by visually inspecting funnel plots of standardized mean differences against standard error for asymmetry (38). When at least 10 studies were available for analysis, Egger’s test of the intercepts (39) was used to formally test asymmetry, and the Duval and Tweedie trim and fill (40) was used to quantify the magnitude of small study effect. When less than 10 studies were available and potential asymmetry was found, a sensitivity analysis was performed by recalculating effect size after removal of outliers. A planned series of subgroup analyses based on key study design features (13) was not performed due to null statistical heterogeneity among mild cognitive impairment outcomes (I2=0% and τ2=0.001 for the most powered analysis, Figure 1), making redundant tests for further between-study variance. All analyses were performed using Comprehensive Meta-analysis version 3.0.
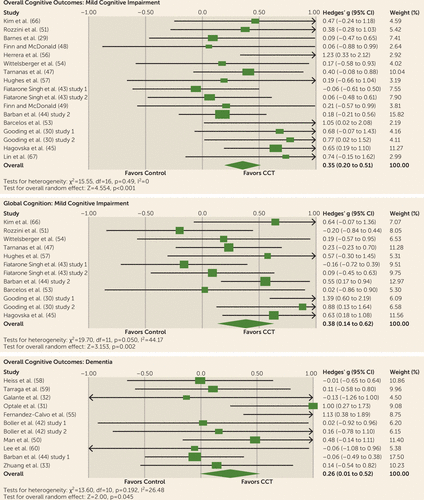
FIGURE 1. Meta-Analyses of Overall and Global Cognition Outcomesa
a Studies are sorted by publication year. CCT=computerized cognitive training.
Results
Study Selection
The initial search provided a total of 22,276 records. After removing duplicates, 14,961 articles were screened based on titles and abstracts, of which 660 full-text versions were assessed for inclusion. Twenty-six studies were eligible for inclusion in the review, of which one was excluded because the summary data were not provided in the original report (41) and could not be obtained from the authors. Four articles (30, 42–44) were split into two studies each, and two articles reporting outcomes from the same trial (45, 46) were combined into one study. Finally, one additional study was obtained from a book chapter (47), resulting in a final data set of 29 independent comparisons (mild cognitive impairment: k=17, dementia: k=12) (Figure 2). We requested additional data from authors of 18 reports, of which six provided data (29, 31, 44, 48–50).
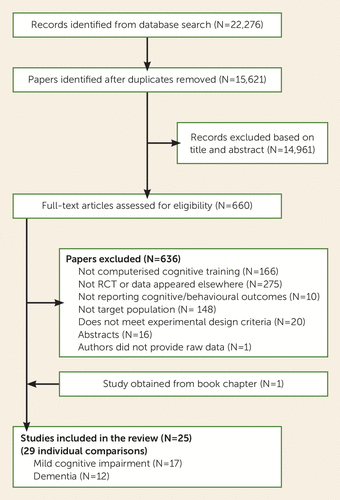
FIGURE 2. Summary of Trial Identification and Selectiona
a RCT=randomized controlled trial.
Characteristics of Included Studies
Mild cognitive impairment.
The 17 included studies encompassed 686 participants (CCT: N=351, mean group size: N=21; control: N=335, mean group size: N=20) (Table 1). Mean age ranged between 67 and 81 years old, and 51.88% of participants were female. One study (51) did not report gender ratios. Active control was provided in 11/17 studies. The mean PEDro-P score was 7.2/9 (SD=1.03), and 14/17 studies had high or unclear risk of bias (for risk of bias assessments, see Table S3 in the online data supplement). The majority of studies (15/17) administered supervised training.
| Study | N | Population Diagnosis | Mean Age (Years)b | Sex (% Female) | Mean Mini-Mental State Examination or Equivalent | Delivery | Program and Targeted Domains | Dosec | Number of Sessionsd | Session Lengthe | Session/Weekf | Control | Risk of Biasg | PEDro-P Scale |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Kim et al. (66) | 30 (CCT, N=15; control, N=15) | Mild cognitive impairment | 78.7 | 70 | 26.7 | Supervised | Virtual reality simulating household tasks | 6 | 12 | 30 | 3 | Active | High | 7 |
| Rozzini et al. (51) | 37 (CCT, N=15; control, N=22) | Mild cognitive impairment | 26.2 | Supervised | Neuropsychological training: | 60 | 60 | 60 | 5 | Active | Low | 8 | ||
| Memory, attention, language, executive function, visuospatial processing | ||||||||||||||
| Barnes et al. (29) | 47 (CCT, N=22; control, N=25) | Mild cognitive impairment | 74 | 40 | Home-based | Posit science brain fitness | 50 | 30 | 100 | 5 | Active | High | 8 | |
| Speed, verbal memory, working memory | ||||||||||||||
| Finn et al. (48) | 16 (CCT, N=8; control, N=8) | Mild cognitive impairment | 72.69 | 50 | 27.76 | Home-based | Lumosity | 10 | 30 | 20 | 3–5 | Passive | High | 7 |
| Attention, speed, nonverbal memory, executive functions | ||||||||||||||
| Herrera et al. (56) | 22 (CCT, N=11; control, N=11) | Mild cognitive impairment | 76.63 | 50 | 27.27 | Supervised | In-house program | 24 | 24 | 60 | 2 | Active | High | 8 |
| Verbal memory, nonverbal memory, verbal learning, non- verbal learning, attention, speed | ||||||||||||||
| Tarnanas et al. (47) | 71 (CCT, N=32; control, N=39) | Mild cognitive impairment | 70.05 | 60.5 | 26.5 | Supervised | Virtual reality museum task | 60 | 40 | 90 | 2 | Active | High | 7 |
| Wittelsberger et al. (54) | 27 (CCT, N=17; control, N=10) | Mild cognitive impairment | 70.07 | 48.14 | 22.88 | Supervised | Nintendo Wii bowling | 12 | 12 | 60 | 2 | Passive | High | 5 |
| Finn et al. (49) | 24 (CCT, N=12; control, N=12) | Mild cognitive impairment | 73.95 | 29.16 | 27.79 | Supervised | Repetition lag training | 9 | 6 | 90 | 2 | Passive | High | 6 |
| Verbal learning, verbal memory | ||||||||||||||
| Hughes et al. (57) | 20 (CCT, N=10; control, N=10) | Mild cognitive impairment | 77.4 | 70 | 27.1 | Supervised | Nintendo Wii | 36 | 24 | 90 | 1 | Active | High | 7 |
| Fiatarone Singh et al. (43) (study 1 [CCT + exercise vs. sham CCT + exercise]) | 49 (CCT, N=27; control, N=22) | Mild cognitive impairment | 70.1 | 68 | 27 | Supervised | COGPACK | 78 | 52 | 90 | 2 | Active | Low | 9 |
| Verbal memory, nonverbal memory, executive functions, attention, speed | ||||||||||||||
| Fiatrone Singh et al. (43) (study 2 [CCT + sham exercise vs. sham CCT + sham exercise]) | 51 (CCT, N=24; control, N=27) | Mild cognitive impairment | 70.1 | 68 | 27 | Supervised | COGPACK | 78 | 52 | 90 | 2 | Active | Low | 9 |
| Verbal memory, nonverbal memory, executive functions, attention, speed | ||||||||||||||
| Barban et al. (44) (study 2, mild cognitive impairment) | 106 (CCT, N=46; control, N=60) | Mild cognitive impairment | 73.54 | 47.16 | 27.74 | Supervised | Sociable | 24 | 24 | 60 | 2 | Passive | High | 8 |
| Verbal memory, nonverbal memory, executive functions, language, attention, visuospatial processing | ||||||||||||||
| Hagovska et al. (45, 46) | 78 (CCT, N=40; control, N=38) | Mild cognitive impairment | 66.97 | 48.75 | 26.33 | Supervised | CogniPlus | 10 | 20 | 30 | 2 | Passive | High | 7 |
| Verbal memory, nonverbal memory, verbal learning, nonverbal learning, working memory, attention, executive functions, visuospatial processing | ||||||||||||||
| Barcelos et al. (53) | 17 (CCT, N=8; control, N=9) | Mild cognitive impairment | 80.6 | 56 | 20.8h | Supervised | In-house virtual reality enhanced recumbent stationary bike coin and dragon collection | 18 | 24 | 20–45 | 2 | Active | High | 6 |
| Visuospatial processing, executive functions, attention | ||||||||||||||
| Gooding et al. (30) (study 1 [CCT and ACG]) | 41 (CCT, N=31; control, N=10) | Mild cognitive impairment | 75.59j | 61.9j | 50.62i | Supervised | BrainFitness by Posit Science | 30 | 32 | 60 | 2 | Active | High | 5 |
| Memory, attention, executive functions | ||||||||||||||
| Gooding (30) (study 2 [CVT and ACG]) | 33 (CCT, N=23; CTL, N=10) | Mild cognitive impairment | 75.59j | 61.9j | 50.84i | Supervised | BrainFitness by Posit Science | 30 | 32 | 60 | 2 | Active | High | 5 |
| Memory, attention, executive functions | ||||||||||||||
| Lin et al. (67) | 21 (CCT, N=10; control, N=11) | Mild cognitive impairment | 73.0 | 47.62 | 25.02h | Home-based | Posit Science InSight. | 24 | 24 | 60 | 4 | Active | High | 7 |
| Processing speed, visuospatial, attention, executive functions | ||||||||||||||
| Optale et al. (31) | 31 (CCT, N=15; control, N=16) | Mixedk | 80.96 | 67.74 | 21.91 | Supervised | Virtual reality Virtools platform | 18 | 36 | 30 | 3 | Active | High | 8 |
| Nonverbal learning, nonverbal memory, attention, visuospatial processing | ||||||||||||||
| Galante et al. (32) | 11 (CCT, N=7; control, N=4) | Mixedk | 75.51 | Not reported | 22.9 | Supervised | Neuropsychological training | 12 | 12 | 60 | 3 | Active | High | 9 |
| Memory (domain unspecified), working memory, language, attention, executive functions, visuospatial processing | ||||||||||||||
| Zhuang et al. (33) | 33 (CCT, N=19; control, N=14) | Mixedk | 78.07 | 75.75 | 10.16 | Supervised | In-house program | 108 | 72 | 90 | 3 | Not Specified | High | 8 |
| Nonverbal learning, Nonverbal memory, executive functions, visuospatial, processing | ||||||||||||||
| Heiss et al. (58) | 35 (CCT, N=18; control, N=17) | Dementia | 66.29 | 45.71 | 21.1 | Supervised | Rigling Reha-Service | 48 | 48 | 60 | 2 | Active | High | 6 |
| Memory, executive functions, visuospatial | ||||||||||||||
| Lowenstein et al. (52) | 44 (CCT, N=19, control, N=25) | Dementia | 76.43 | 34.09 | 23.96 | Supervised + home-based | Commercial games | 18 | 24 | 45 | 2 | Active | High | 7 |
| Language, executive functions, nonverbal learning, nonverbal memory, verbal memory, nonverbal memory, attention | ||||||||||||||
| Tarraga et al. (59) | 31 (CCT, N=15; control, N=16) | Dementia | 76.54 | 87.09 | 21.55 | Supervised | Smart Brain | 24 | 72 | 20 | 3 | Active | High | 7 |
| Memory, attention, language, visuospatial processing, working memory, executive functions | ||||||||||||||
| Fernandez-Calvo et al. (55) | 30 (CCT, N=15; control, N=15) | Dementia | 75.7 | 43.33 | 19.66 | Supervised | Nintendo Wii Big Brain Academy | 36 | 36 | 60 | 3 | Passive | Low | 8 |
| Nonverbal memory, working memory, executive functions, visuospatial processing | ||||||||||||||
| Boller et al. (42) (study 1 [recollection + control]) | 18 (CCT, N=12; control, N=6) | Dementia | 80.82 | 55.5 | 24.59 | Supervised + home-based | Repetition lag training | 6 | 30 | 20 | 15 | Passive | High | 9 |
| Verbal learning, verbal memory | ||||||||||||||
| Boller et al. (42) (study 2 [recognition + control]) | 18 (CCT, N=12; control, N=6) | Dementia | 81.5 | 55.5 | 25.51 | Supervised + home-based | Repetition lag training | 6 | 30 | 20 | 15 | Passive | High | 9 |
| Verbal learning, verbal memory | ||||||||||||||
| Lee et al. (60) | 13 (CCT, N=7; control, N=6) | Dementia | 77.7 | 69.23 | 16.07 | Supervised | In-house computerized errorless learning program | 6 | 12 | 60 | 2 | Active | High | 6 |
| Verbal memory, nonverbal memory, working memory, verbal learning, nonverbal learning, attention, executive functions, global cognition | ||||||||||||||
| Man et al. (50) | 44 (CCT, N=20; control, N=24) | Dementia | 80.29 | 85 | 22.03 | Supervised | Virtual reality home and shop simulation | 5 | 10 | 30 | 2–3 | Active | Low | 7 |
| Barban et al. (44) (study 1) | 81 (CTT, N=42; control, N=39) | Dementia | 76.79 | 70.37 | 23.4 | Supervised | Sociable | 24 | 24 | 60 | 2 | Passive | High | 8 |
| Verbal memory, nonverbal memory, executive functions, language, attention, visuospatial processing |
TABLE 1. Characteristics of Included Studiesa
Dementia.
The 12 included studies encompassed a total of 389 participants (CCT: N=201, mean group size: N=17; control: N=188, mean group size: N=16) (Table 1). Mean age ranged between 66 and 81 years old, and 63.5% of participants were female. One study (32) did not report gender ratios. Active control was confirmed in 7/12 studies. The mean PEDro-P score was 7.7/9 (SD=1.25), and 10/12 studies had high or unclear risk of bias (see Table S3 in the online data supplement). The majority of studies provided supervised training (9/12). One study (52) reported only behavioral outcomes.
Meta-Analysis of Mild Cognitive Impairment Outcomes
Overall efficacy on cognitive outcomes.
The overall effect size was moderate and statistically significant (k=17, g=0.35, 95% confidence interval [CI]=0.20–0.51, p<0.001, I2=0%) (Figure 1). The funnel plot did not reveal significant asymmetry (Egger’s intercept=1.39, p=0.11 [see Figure S1 in the data supplement). After splitting arms, CCT groups revealed a statistically significant improvement (g=0.32, 95% CI=0.20–0.44, I2=28.47%), compared with no change across control groups (g=0.02, 95% CI=–0.08 to 0.11, I2=0%). The effect size across active-controlled trials (k=11, g=0.40, 95% CI=0.17–0.63, I2=18.95%) was comparable to that of trials with passive control groups (k=6, g=0.32, 95% CI=0.09–0.55, I2=0%). Domain-specific effect sizes are summarized in Figure 3.
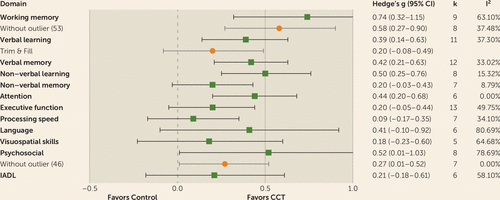
FIGURE 3. Efficacy of Computerized Cognitive Training (CCT) in Mild Cognitive Impairment Within Individual Domainsa
a IADL=Instrumental activities of daily living.
Global cognition.
The global cognition effect size was moderate and statistically significant (k=12, g=0.38, 95% CI=0.14–0.62, p=0.002, I2=44.17%) (Figure 1). The funnel plot did not reveal asymmetry (Egger’s intercept=0.33, p=0.83) (see Figure S1 in the data supplement). Once again, the pooled effect size across CCT groups was significant (g=0.28, 95% CI=0.05–0.51), compared with no change in the controls (g=–0.02, 95% CI=–0.16 to 0.12), and there was no difference between the effect across active (k=8, g=0.41, 95% CI=0.03–0.75) and passive (k=4, g=0.37, 95% CI=0.02–0.72) controlled trials.
Verbal learning.
The verbal learning effect size was moderate and statistically significant (k=11, g=0.39, 95% CI=0.14–0.63, p=0.002, I2=37.3%) (see Figure S2 in the data supplement). The funnel plot revealed significant asymmetry (Egger’s intercept=3.95, p=0.04) (see Figure S3 in the data supplement). A trim and fill analysis imputed three studies; the adjusted effect size was small and statistically nonsignificant (g=0.20, 95% CI=–0.08 to 0.49).
Verbal memory.
The verbal memory effect size was moderate and statistically significant (k=12, g=0.42, 95% CI=0.21–0.63, p<0.001, I2=33.02%) (see Figure S2 in the data supplement). The funnel plot revealed significant asymmetry (Egger’s intercept=2.5, p=0.06) (see Figure S3 in the data supplement). A trim and fill analysis did not impute additional studies.
Nonverbal learning.
The nonverbal learning effect size was moderate and statistically significant (k=8, g=0.50, 95% CI=0.25–0.76, p<0.001, I2=15.32%) (see Figure S2 in the data supplement). The funnel plot did not reveal asymmetry (see Figure S3 in the data supplement).
Working memory.
The working memory effect size was large and statistically significant (k=9, g=0.74, 95% CI=0.32–1.15, p<0.001, I2=63.1%) (see Figure S4 in the data supplement). The funnel plot revealed one outlier (53) (see Figure S3 in the data supplement). A sensitivity analysis after removal of the outlier revealed a moderate and statistically significant effect (g=0.58, 95% CI=0.27–0.90).
Attention.
The attention effect size was moderate and statistically significant (k=6, g=0.44, 95% CI=0.20–0.68, p<0.001, I2=0%) (see Figure S4 in the data supplement). The funnel plot revealed potential asymmetry (see Figure S3 in the data supplement), but asymmetry was not formally assessed due to an insufficient number of studies.
Psychosocial functioning.
The psychosocial functioning effect size was moderate and statistically significant (k=8, g=0.52, 95% CI=0.01–1.03, p=0.045, I2=78.69%) (see Figure S4 in the data supplement). The funnel plot revealed one study outside of the funnel (46), but this was a relatively large study and the rest of the funnel plot did not suggest small-study effect (see Figure S3 in the data supplement). A sensitivity analysis after removal of the outlier revealed a small and statistically significant effect with no evidence of heterogeneity (g=0.27, 95% CI=0.01–0.52, p=0.04, I2=0%).
Other domains.
Statistically nonsignificant results were found for nonverbal memory (k=7, g=0.20, 95% CI=–0.03 to 0.43, I2=8.79%), executive function (k=13, g=0.20, 95% CI=–0.05 to 0.44, I2=49.75%), processing speed (k=7, g=0.09, 95% CI=–0.17 to 0.35, I2=34.1%), visuospatial skills (k=5, g=0.18, 95% CI=–0.23 to 0.60, I2=64.68%), language (k=6, g=0.41, 95% CI=–0.10–0.92, I2=80.69%), or instrumental activities of daily living (k=6, g=0.21, 95% CI=–0.18 to 0.61) (see Figure 3). Analysis of activities of daily living outcomes was not performed because only one study (54) was available for analysis.
Meta-Analysis of Dementia Outcomes
Overall efficacy on cognitive outcomes.
The overall effect was small and statistically significant (k=11, g=0.26, 95% CI=0.01–0.52, p=0.045, I2=26.48%) (Figure 1). The funnel plot did not reveal significant asymmetry (Egger’s intercept=0.61, p=0.67) (see Figure S3 in the data supplement). However, the summary effect was driven by two studies with a g value ≥1.0 (31, 55). Removal of any of these resulted in statistically nonsignificant effect sizes (after removal of Optale et al. [31]: g=0.17, 95% CI=–0.05 to 0.40, I2=1.61%; after removal of Fernandez-Calvo et al. [55]: g=0.17, 95% CI=–0.06 to 0.39], I2=0%). A mixed-effects analysis revealed that separating these two studies from the other studies in the analysis created two homogenous subgroups with statistically significant difference in effect sizes (outliers: g=1.07, 95% CI=0.54–1.59, I2=0%; remaining: g=0.08, 95% CI=–0.15 to 0.31, I2=0%; Q-between=11.23, df=1, p=0.001).
Efficacy on individual cognitive domains.
The effect size on global cognition was moderate and statistically nonsignificant (k=7, g=0.31, 95% CI=–0.11 to 0.72, p=0.15, I2=56.66%). A moderate and statistically significant effect size was found on visuospatial skills (k=3, g=0.54, 95% CI=0.07–1.01), but this was once more driven by the Optale et al. study (31) (see Figure S4 in the data supplement). There were no other statistically significant effects on any other domain (see Table S4 in the data supplement).
Long-Term Outcomes
Four mild cognitive impairment studies from three articles (43, 56, 57) and four dementia studies (32, 58–60) reported outcomes beyond the first follow-up (see Table S5 in the data supplement). Results were not pooled due to an insufficient number of studies and variability of follow-up times, but individual study results indicated a substantial waning of training benefits after training cessation.
Discussion
Based on results from 17 randomized controlled trials of moderate quality, we conclude that CCT is a viable intervention for enhancing cognition in people with mild cognitive impairment. The overall effect size on cognition (g=0.35) is larger than effect sizes previously reported for healthy older adults (g=0.22) (13) and for Parkinson’s disease (g=0.23) (14). This effect was corroborated by a moderate effect size on common clinical measures of global cognition (mainly the Mini-Mental State Examination and Alzheimer’s Disease Assessment Scale-cognitive subscale). Participants in CCT groups improved significantly over the intervention period, while controls did not show any cognitive change, immune to retest effects or nonspecific factors. Most of the trials (70%) used an active control condition, and the effects across active- and passive-controlled trials were comparable. The results of the mild cognitive impairment analysis are therefore robust and indicate a beneficial therapeutic role for CCT in this population. Our analysis updates the benefits on global cognition and memory found in a previous meta-analysis of cognitive training in mild cognitive impairment (17) and is the first, to our knowledge, to focus specifically on randomized trials of CCT.
Moderate effect sizes on most memory and learning domains are encouraging, as amnestic symptoms are the most common presentation of Alzheimer’s disease (27), and amnestic mild cognitive impairment profiles are at higher risk for dementia conversion (3). On the other hand, consistent with findings of previous meta-analyses of CCT (13, 15), we report lack of efficacy on executive function, a key predictor of functional decline (61). Since cognitive training gains typically reflect training content (13, 62), this result may be due to insufficient training on executive processes (mainly fluid intelligence, inhibitory control, and reasoning) within studies. Future studies should consider dedicating more time to executive tasks. More surprising is the null effect on processing speed, since CCT exercises are typically timed and this domain was among the most responsive in prior meta-analyses in other populations (13, 14). In healthy older adults, effects on speed are driven by–and limited to–trials of processing speed training (13), and so again training content may help explain this result.
Depression is associated with mild cognitive impairment (63), as well as conversion to dementia (64). It is therefore notable that we found moderate effect sizes on psychosocial functioning (depression, quality of life, and neuropsychiatric symptoms) in mild cognitive impairment, consistent with prior studies (15) and suggestive that CCT may generalize to benefit mood. On the other hand, reliable effects were not seen on instrumental activities of daily living outcomes. A limitation in this area is the prevalent use of subjective measures that are insensitive to naturalistic or intervention-related change.
Conversely, the pattern of results in individuals with dementia was weak and driven by two studies. Importantly, clinically meaningful effect sizes were found only for studies that used nontraditional approaches to CCT, namely virtual reality (31, 50) and Nintendo Wii (55) (see Figure 1). It is conceivable that these methods are more stimulating and personally engaging than traditional CCT, an idea that merits further research. Overall, there is no robust evidence that CCT can benefit cognition or function in dementia, in keeping with prior meta-analyses in the field (22, 23).
Limitations
To our knowledge, this is the first meta-analysis focusing exclusively on randomized trials of CCT in people with mild cognitive impairment or dementia. Yet since most trials have focused on short-term cognitive outcomes, we had insufficient data to evaluate the durability of CCT effects and whether these may reduce conversion to dementia. Similarly, functional outcomes were measured mainly using proxy measures that are prone to multiple-source bias, typically requiring long-term follow-up and large samples to detect subtle effects on function.
Methodological differences across mild cognitive impairment studies did not translate into statistically meaningful heterogeneity and thus did not warrant planned moderator analysis such as delivery mode and dose. These factors are critical to CCT outcomes but have yet to be thoroughly investigated in primary studies (13, 65). Notably, while methodological quality across the literature has improved since prior reviews, sample sizes continue to be small. Given an effect size of g=0.36, 80% power, and controlling α at 0.05, the minimal intention-to-treat sample size for CCT trials in mild cognitive impairment is about 64. By contrast, only three studies would have met this criterion (44, 46, 47), and the median sample size across studies was 33.
It is noteworthy that we compared effect size estimates and precision in active- and passive-controlled trials because it has been argued that CCT benefits may be limited to passive-controlled studies (18, 20). As in healthy older adults (13), we did not find any systematic difference in effect sizes. However, since only five of the 17 studies employed a passive-control design, a formal subgroup analysis was underpowered and warrants caution.
Conclusions
In mild cognitive impairment, CCT is efficacious on global cognition, memory, working memory, and attention and helps improve psychosocial functioning, including depressive symptoms. Effects on other domains such as executive function and processing speed are negligible. Conversely, CCT is not likely to be beneficial for people with dementia, but immersive technologies may be more useful. Future trials should include larger sample sizes and directly compare CCT alternatives in order to optimize outcomes. Finally, there is insufficient data to determine whether training gains can be maintained over the long-term without further training, and thus study of efficient booster regimens is needed in order to examine whether CCT can indeed delay or prevent progression of mild cognitive impairment to dementia.
1 The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement 2013; 9:63–75 e62Crossref, Medline, Google Scholar
2 : The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7:270–279Crossref, Medline, Google Scholar
3 : Spectrum of cognition short of dementia: Framingham Heart Study and Mayo Clinic Study of Aging. Neurology 2015; 85:1712–1721Crossref, Medline, Google Scholar
4 : The role of cognitive impairment in fall risk among older adults: a systematic review and meta-analysis. Age Ageing 2012; 41:299–308Crossref, Medline, Google Scholar
5 : Associations between mild cognitive impairment and hospitalization and readmission. J Am Geriatr Soc 2015; 63:1880–1885Crossref, Medline, Google Scholar
6 : Rate of progression of mild cognitive impairment to dementia—meta-analysis of 41 robust inception cohort studies. Acta Psychiatr Scand 2009; 119:252–265Crossref, Medline, Google Scholar
7 : Higher risk of progression to dementia in mild cognitive impairment cases who revert to normal. Neurology 2014; 82:317–325Crossref, Medline, Google Scholar
8 : Reversion from mild cognitive impairment to normal or near-normal cognition: risk factors and prognosis. Neurology 2012; 79:1591–1598Crossref, Medline, Google Scholar
9 : Incidence of mild cognitive impairment in the Pittsburgh Cardiovascular Health Study-Cognition Study. Neurology 2012; 79:1599–1606Crossref, Medline, Google Scholar
10 : Treatment for mild cognitive impairment: systematic review. Br J Psychiatry 2013; 203:255–264Crossref, Medline, Google Scholar
11 : Efficacy and safety of cognitive enhancers for patients with mild cognitive impairment: a systematic review and meta-analysis. CMAJ 2013; 185:1393–1401Crossref, Medline, Google Scholar
12 : Aging, training, and the brain: a review and future directions. Neuropsychol Rev 2009; 19:504–522Crossref, Medline, Google Scholar
13 : Computerized cognitive training in cognitively healthy older adults: a systematic review and meta-analysis of effect modifiers. PLoS Med 2014; 11:e1001756Crossref, Medline, Google Scholar
14 : Cognitive training in Parkinson disease: a systematic review and meta-analysis. Neurology 2015; 85:1843–1851Crossref, Medline, Google Scholar
15 : Computerized cognitive training and functional recovery in major depressive disorder: a meta-analysis. J Affect Disord 2016; 189:184–191Crossref, Medline, Google Scholar
16 : Prevention of sporadic Alzheimer’s disease: lessons learned from clinical trials and future directions. Lancet Neurol 2015; 14:926–944Crossref, Medline, Google Scholar
17 : Cognitive intervention for persons with mild cognitive impairment: A meta-analysis. Ageing Res Rev 2011; 10:285–296Crossref, Medline, Google Scholar
18 : Cognition-based interventions for healthy older people and people with mild cognitive impairment. Cochrane Database Syst Rev 2011; (1):CD006220Medline, Google Scholar
19 : Cognitive intervention in amnestic mild cognitive impairment: a systematic review. Neurosci Biobehav Rev 2012; 36:1163–1178Crossref, Medline, Google Scholar
20 : Cognitive interventions in healthy older adults and people with mild cognitive impairment: a systematic review. Ageing Res Rev 2013; 12:263–275Crossref, Medline, Google Scholar
21 : Cognitive stimulation for dementia: a systematic review of the evidence of effectiveness from randomised controlled trials. Ageing Res Rev 2013; 12:253–262Crossref, Medline, Google Scholar
22 : Cognitive training and cognitive rehabilitation for mild to moderate Alzheimer’s disease and vascular dementia. Cochrane Database Syst Rev 2013; 6:CD003260Google Scholar
23 : Do cognitive interventions improve general cognition in dementia? A meta-analysis and meta-regression. BMJ Open 2015; 5:e005247–e005247Crossref, Medline, Google Scholar
24 : Computerized and virtual reality cognitive training for individuals at high risk of cognitive decline: systematic review of the literature. Am J Geriatr Psychiatry 2015; 23:335–359Crossref, Medline, Google Scholar
25 : Computer-based cognitive interventions for people living with dementia: a systematic literature review and meta-analysis. Aging Ment Health (Epub ahead of print, January 25, 2016)Google Scholar
26 : The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 2009; 339:b2700Crossref, Medline, Google Scholar
27 : The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7:263–269Crossref, Medline, Google Scholar
28 : A Compendium of Neuropsychological Tests: Administration, Norms and Commentary. Oxford, United Kingdom, Oxford University Press, 2006Google Scholar
29 : Computer-based cognitive training for mild cognitive impairment: results from a pilot randomized, controlled trial. Alzheimer Dis Assoc Disord 2009; 23:205–210Crossref, Medline, Google Scholar
30 : Comparing three methods of computerized cognitive training for older adults with subclinical cognitive decline Neuropsychol Rehabil 2016; 26:810–821Crossref, Medline, Google Scholar
31 : Controlling memory impairment in elderly adults using virtual reality memory training: a randomized controlled pilot study. Neurorehabil Neural Repair 2010; 24:348–357Crossref, Medline, Google Scholar
32 : Computer-based cognitive intervention for dementia: preliminary results of a randomized clinical trial. G Ital Med Lav Ergon 2007; 29(suppl B):B26–B32Medline, Google Scholar
33 : The impact of human-computer interaction-based comprehensive training on the cognitive functions of cognitive impairment elderly individuals in a nursing home. J Alzheimers Dis 2013; 36:245–251Crossref, Medline, Google Scholar
34 : The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011; 343:d5928Crossref, Medline, Google Scholar
35 : Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther 2003; 83:713–721Medline, Google Scholar
36 : Statistical Power Analysis for the Behavioral Sciences. Oxfordshire, United Kingdom, Taylor and Francis, 2013Crossref, Google Scholar
37 : Measuring inconsistency in meta-analyses. BMJ 2003; 327:557–560Crossref, Medline, Google Scholar
38 : Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011; 343:d4002Crossref, Medline, Google Scholar
39 : Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315:629–634Crossref, Medline, Google Scholar
40 : Trim and fill: a simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000; 56:455–463Crossref, Medline, Google Scholar
41 : Efficacy of an adjunctive computer-based cognitive training program in amnestic mild cognitive impairment and Alzheimer’s disease: a single-blind, randomized clinical trial. Int J Geriatr Psychiatry 2013; 28:91–99Crossref, Medline, Google Scholar
42 : Recollection training and transfer effects in Alzheimer’s disease: effectiveness of the repetition-lag procedure. Brain Cogn 2012; 78:169–177Crossref, Medline, Google Scholar
43 : The Study of Mental and Resistance Training (SMART) study—resistance training and/or cognitive training in mild cognitive impairment: a randomized, double-blind, double-sham controlled trial. J Am Med Dir Assoc 2014; 15:873–880Crossref, Medline, Google Scholar
44 : Protecting cognition from aging and Alzheimer’s disease: a computerized cognitive training combined with reminiscence therapy. Int J Geriatr Psychiatry 2016; 31:340–348Crossref, Medline, Google Scholar
45 : Effect of a combining cognitive and balanced training on the cognitive, postural and functional status of seniors with a mild cognitive deficit in a randomized, controlled trial. Eur J Phys Rehabil Med 2016; 52:101–109Medline, Google Scholar
46 : Impact of the combination of cognitive and balance training on gait, fear and risk of falling and quality of life in seniors with mild cognitive impairment. Geriatr Gerontol Int 2016; 16:1043–1050Crossref, Medline, Google Scholar
47 : Assessing Virtual Reality Environments as Cognitive Stimulation Method for Patients with MCI, in Technologies of Inclusive Well-Being. Edited by Brooks AL, Brahnam S, Jain LC. Berlin, Springer, 2014, pp 39–74Crossref, Google Scholar
48 : Computerised cognitive training for older persons with mild cognitive impairment: A pilot study using a randomised controlled trial design. Brain Impair 2011; 12:187–199Crossref, Google Scholar
49 : Repetition-lag training to improve recollection memory in older people with amnestic mild cognitive impairment. A randomized controlled trial. Neuropsychol Dev Cogn B Aging Neuropsychol Cogn 2015; 22:244–258Crossref, Medline, Google Scholar
50 : Evaluation of a virtual reality-based memory training programme for Hong Kong Chinese older adults with questionable dementia: a pilot study. Int J Geriatr Psychiatry 2012; 27:513–520Crossref, Medline, Google Scholar
51 : Efficacy of cognitive rehabilitation in patients with mild cognitive impairment treated with cholinesterase inhibitors. Int J Geriatr Psychiatry 2007; 22:356–360Crossref, Medline, Google Scholar
52 : Cognitive rehabilitation of mildly impaired Alzheimer disease patients on cholinesterase inhibitors. Am J Geriatr Psychiatry 2004; 12:395–402Crossref, Medline, Google Scholar
53 : Aerobic and cognitive exercise (ACE) pilot study for older adults: executive function improves with cognitive challenge while exergaming. J Int Neuropsychol Soc 2015; 21:768–779Crossref, Medline, Google Scholar
54 : The influence of Nintendo-Wii bowling upon residents of retirement homes. Z Gerontol Geriatr 2013; 46:425–430Crossref, Medline, Google Scholar
55 : Efficacy of cognitive training programs based on new software technologies in patients with Alzheimer-type dementia. Psicothema 2011; 23:44–50Medline, Google Scholar
56 : Positive effects of computer-based cognitive training in adults with mild cognitive impairment. Neuropsychologia 2012; 50:1871–1881Crossref, Medline, Google Scholar
57 : Interactive video gaming compared with health education in older adults with mild cognitive impairment: a feasibility study. Int J Geriatr Psychiatry 2014; 29:890–898Crossref, Medline, Google Scholar
58 : Long-term effects of phosphatidylserine, pyritinol, and cognitive training in Alzheimer’s disease: a neuropsychological, EEG, and PET investigation. Dementia 1994; 5:88–98Medline, Google Scholar
59 : A randomised pilot study to assess the efficacy of an interactive, multimedia tool of cognitive stimulation in Alzheimer’s disease. J Neurol Neurosurg Psychiatry 2006; 77:1116–1121Crossref, Medline, Google Scholar
60 : Evaluation of a computer-assisted errorless learning-based memory training program for patients with early Alzheimer’s disease in Hong Kong: a pilot study. Clin Interv Aging 2013; 8:623–633Medline, Google Scholar
61 : Executive function and instrumental activities of daily living in mild cognitive impairment and Alzheimer’s disease. Alzheimers Dement 2011; 7:300–308Crossref, Medline, Google Scholar
62 : Effects of cognitive training interventions with older adults: a randomized controlled trial. JAMA 2002; 288:2271–2281Crossref, Medline, Google Scholar
63 : Late-life depression, mild cognitive impairment, and dementia. JAMA Neurol 2013; 70:374–382Crossref, Medline, Google Scholar
64 : Modifiable predictors of dementia in mild cognitive impairment: a systematic review and meta-analysis. Am J Psychiatry 2015; 172:323–334Link, Google Scholar
65 : Cognitive rehabilitation of memory for mild cognitive impairment: a methodological review and model for future research. J Int Neuropsychol Soc 2014; 20:135–151Crossref, Medline, Google Scholar
66 : Effectiveness of cognitive training based on virtual reality for the elderly. J Korean Acad Rehabil Med. 2005; 29:424–433Google Scholar
67 : Cognitive and neural effects of vision-based speed-of-processing training in older adults with amnestic mild cognitive impairment: a pilot study. J Am Geriatr Soc 2016; 64:1293–1298Crossref, Medline, Google Scholar



