Pharmacological Management of Lewy Body Dementia: A Systematic Review and Meta-Analysis
Abstract
Objective:
The authors examined research on effects, costs, and patient and caregiver views of pharmacological management strategies for Lewy body dementia.
Method:
Studies were identified through bibliographic databases, trials registers, gray literature, reference lists, and experts. The authors used the search terms “Lewy or parkinson” and “dementia” through March 2015 and used the following inclusion criteria: participants with diagnoses of Lewy body dementia, dementia with Lewy bodies, or Parkinson’s disease dementia (or participants’ caregivers); investigation of pharmacological management strategies; outcome measures and test scores reported. Data extraction and quality assessment were conducted by at least two authors. Meta-analyses were conducted, and when studies could not be combined, summaries were provided.
Results:
Forty-four studies examining 22 strategies were included in the review. Meta-analysis indicated beneficial effects of donepezil and rivastigmine for cognitive and psychiatric symptoms. Rivastigmine, but not donepezil, was associated with greater risk of adverse events. Meta-analysis of memantine suggested that it is well tolerated but with few benefits. Descriptive summaries provide some evidence of benefits for galantamine, modafinil, levodopa, rotigotine, clozapine, duloxetine, clonazepam, ramelteon, gabapentin, zonisamide, and yokukansan. Piracetam, amantadine, selegiline, olanzapine, quetiapine, risperidone, and citalopram do not appear to be effective.
Conclusions:
High-level evidence related to pharmacological strategies for managing Lewy body dementia is rare. Strategies for important areas of need in Lewy body dementia, such as autonomic symptoms and caregiver burden, have not been investigated, nor have the views of patients and caregivers about pharmacological strategies.
Lewy body dementia is a common cause of degenerative dementia in older people, accounting for some 3%–15% of cases (1, 2). It is characterized by impairments and fluctuations in cognition, recurrent visual hallucinations, and motor features of parkinsonism. Other significant features include sleep disorders, depression, delusions, and autonomic dysfunction (3, 4). The term “Lewy body dementia” is used here to include two related disorders: dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD). Comparisons suggest a broad overlap clinically, although executive impairment, delusions, and hallucinations may be more common in DLB (5, 6). The diagnosis of PDD is applied when motor symptoms occur at least 1 year before dementia, and the diagnosis of DLB is applied when dementia precedes or is closely followed by motor symptoms (4).
While a range of pharmacological strategies are used in an attempt to ameliorate the symptoms of Lewy body dementia, and while clinical guidelines have recommended cholinesterase inhibitors for cognitive and psychiatric symptoms, the current evidence for management options for the range of symptoms is limited, and there remains no unified evidence-based management care pathway (7–10).
To develop effective approaches to care, it is necessary to establish which strategies are effective and to determine whether DLB and PDD are amenable to the same treatments. In this article, we review pharmacological management strategies and address three questions: What are their benefits, harms, and costs in the disorders? What views do patients and caregivers have of these strategies? How, when, and where should these strategies be implemented?
Method
The review protocol is registered at PROSPERO (registration number CRD42014007180).
Search Strategy
Studies were identified through bibliographic databases, trials registers, and gray literature (see the registered protocol for full details). Reference lists of relevant studies and previous systematic reviews were also examined, and input was sought from experts on Lewy body dementia. We used the keywords “Lewy or parkinson” and “dementia,” conducting searches until March 2015, without restrictions on time or language.
Study Selection
Titles and abstracts were screened independently by four of the authors, with non-English language papers screened by native speakers. Discrepancies were resolved through discussion between screeners. Potentially relevant studies were obtained in full and examined in detail by the first author against the following criteria: 1) participants had a diagnosis of DLB, PDD, or Lewy body dementia (or were the caregivers of patients with these diagnoses); 2) studies examined pharmacological strategies; and 3) outcome measures and scores were specified. No restrictions were placed on study design, but opinion papers were excluded. (See Figure S1, the study flowchart, in the data supplement that accompanies the online edition of this article.)
Data Extraction
Data were extracted by two of the authors and recorded in an Excel spreadsheet. We collected information related to participant demographic characteristics, selection criteria, study design, management strategies, outcome measures and scores, adverse events, and withdrawals.
Data Synthesis
Studies were grouped and analyzed according to pharmacological strategy. For each strategy, studies of the highest level of evidence were included in the review. Classification of level of evidence was determined using guidelines from the Oxford Centre for Evidence-Based Medicine (11).
Methodological Quality
Methodological quality was assessed by three of the authors using the Quality Assessment Tool for Quantitative Studies (QATQS, www.ephpp.ca/tools.html) (12), which was developed to assess quality across study designs, aiding consistency and clarity of reporting. The QATQS examines selection bias, study design, confounders, blinding, data collection methods, withdrawals, and dropouts. Domains are rated as being of weak, moderate, or strong quality, which feed into an overall rating of study quality. The reliability and validity of the QATQS have been demonstrated (13).
Statistical Analysis
Meta-analysis was conducted using the Cochrane Collaboration’s RevMan, version 5.3 (www.tech.cochrane.org/revman), employing the inverse variance method. Heterogeneity was assessed using the chi-square and I2 statistics and considered significant with p values <0.10 for chi-square and >40% for I2. We employed random-effects models when there was significant study heterogeneity and fixed-effect models when heterogeneity was not significant. Missing data were sought from study authors; for data that were not obtainable, values were estimated using methods outlined in the Cochrane Handbook for Systematic Reviews of Interventions (14). We estimated risk ratio with 95% confidence intervals for dichotomous outcomes and weighted mean difference or standardized mean difference with 95% confidence intervals for continuous outcomes. Descriptive summaries were provided when studies could not be combined.
Results
The titles and abstracts of 28,568 records were screened; of these, 27,935 did not meet inclusion criteria. Assessment of 633 full articles was conducted; of these, 197 met our inclusion criteria, and 44 were included as having the highest level of evidence for 22 pharmacological strategies (see Table S1 in the online data supplement for the characteristics of the included studies).
Cholinesterase Inhibitors
Donepezil and rivastigmine.
Data from studies of donepezil and rivastigmine were combined and examined using meta-analysis to obtain an estimate of their combined and separate effects (no randomized controlled trials were identified for galantamine, which is discussed later). The highest level of evidence for donepezil was from randomized controlled trials, two for DLB and four for PDD (15–20); for rivastigmine, there was one randomized controlled trial each for DLB and PDD (21, 22). The highest level of evidence for donepezil in treating visual hallucinations (a key symptom of Lewy body dementia) was from an uncontrolled trial (23). The highest level of evidence for withdrawal of donepezil was an open-label study (24). We identified three ongoing studies examining donepezil for cognition, psychiatric symptoms, and functional ability (ClinicalTrials.gov identifiers, NCT00776347, NCT01014858; UMIN Clinical Trials Registry [umin.ac.jp] identifier, UMIN000010752), and one study to develop a predictive test to identify which cholinergic medication should be prescribed first (ClinicalTrials.gov identifier, NCT01944436).
Five studies examined clinician impression of change of global outcomes; four of them examined improvement (Figure 1A) (15–17, 22), three examined absence of deterioration (Figure 1B) (15, 17, 22), and four examined impression of change as a continuous outcome measure (16, 17, 19, 22) (see Figure S2A in the online data supplement). Scores were estimated for one trial (15). Analysis of improvement data indicated that participants who were treated with cholinesterase inhibitors were more frequently rated as improved than those receiving placebo (45% compared with 34%; risk ratio=1.37, 95% CI=1.15, 1.62). Subgroup analyses indicated that, compared with placebo, improvement was more likely with donepezil in DLB (64% compared with 33%; risk ratio=1.93, 95% CI=1.08, 3.43) and PDD (49% compared with 38%; risk ratio=1.29, 95% CI=1.02, 1.63), and rivastigmine in PDD (40% compared with 29%; risk ratio=1.37, 95% CI=1.05, 1.79). Analysis of data on absence of deterioration indicated that participants who were treated with cholinesterase inhibitors were more frequently rated as not deteriorating than those receiving placebo (71% compared with 62%; risk ratio=1.26, 95% CI=1.01, 1.57), but with considerable heterogeneity (I2=76%). Subgroup analyses indicated that absence of deterioration was more common in participants with DLB treated with donepezil (96% compared with 50%; risk ratio=1.93, 95% CI=1.34, 2.78), but with no between-group difference for donepezil or rivastigmine in participants with PDD. Analysis of continuous data indicated that mean change scores were 0.55 points lower (suggesting improvements) in participants treated with cholinesterase inhibitors (95% CI=−0.82, −0.29), but with significant heterogeneity (I2=52%). Subgroup analysis indicated benefits from donepezil in DLB (weighted mean difference=−1.13, 95% CI=−1.66, −0.60) and PDD (weighted mean difference=−0.37, 95% CI=−0.60, −0.14), and rivastigmine in PDD (weighted mean difference=−0.50, 95% CI=−0.77, −0.23).
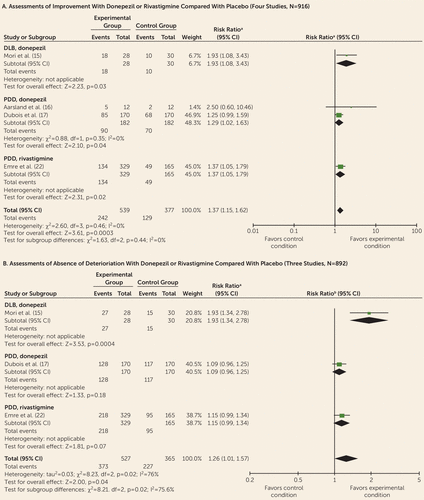
FIGURE 1. Forest Plots of Global Outcomes on the Alzheimer’s Disease Cooperative Study–Clinical Global Impression of Change (Assessing Improvement and Absence of Deterioration), Comparing Donepezil or Rivastigmine With Placebo, in the Treatment of Dementia With Lewy Bodies (DLB) and Parkinson’s Disease Dementia (PDD)
a Mantel-Haenszel method, fixed effect.
b Mantel-Haenszel method, random effects.
Eight studies assessed cognition using the Mini-Mental State Examination (MMSE) (15–22) (Figure 2). Standard deviations were estimated in three trials (17, 18, 21). Mean change scores were 1.26 points higher (suggesting improvements) in participants who received cholinesterase inhibitors (95% CI=0.66, 1.86). In a subgroup analysis, benefits were seen for donepezil in DLB (weighted mean difference=1.93, 95% CI=1.01, 2.85) and for rivastigmine in PDD (weighted mean difference=1.00, 95% CI=0.33, 1.67) but not for rivastigmine in DLB or donepezil in PDD.

FIGURE 2. Forest Plot of Cognitive Functioning on the Mini-Mental State Examination, Comparing Donepezil or Rivastigmine With Placebo, in the Treatment of Dementia With Lewy Bodies (DLB) and Parkinson’s Disease Dementia (PDD) (Eight Studies, N=1,202)
a Inverse variance method, random effects.
Six studies used the 10-item Neuropsychiatric Inventory (NPI-10) to assess psychiatric symptoms (15, 17, 18, 20–22) (Figure 3). Standard deviations were estimated in two trials (17, 18). There were no significant between-group differences, but there was significant heterogeneity (I2=79%). Subgroup analyses indicated benefits for total psychiatric symptoms in PDD from donepezil (weighted mean difference=−1.17, 95% CI=−2.26, −0.08) and rivastigmine (weighted mean difference=−2.00, 95% CI=−3.91, −0.09), but not in DLB from donepezil or rivastigmine. Two studies assessed psychiatric symptoms in DLB using the 4-item NPI (the sum of scores for apathy, delusions, depression, and hallucinations) (15, 21). A significant effect favoring cholinesterase inhibitors was observed (weighted mean difference=−3.36, 95% CI=−5.85, −0.87). Subgroup analysis indicated a benefit from donepezil (weighted mean difference=−4.80, 95% CI=−8.63, −0.97) but not rivastigmine (see Figure S2B in the online data supplement). Generally, subscale scores for the NPI have not been reported, although McKeith et al. (21) and Mori et al. (15) presented them graphically. From the confidence intervals, rivastigmine does not appear to have beneficial effects on subscale items in DLB, but donepezil may be beneficial for delusions, hallucinations, and cognitive fluctuations. In PDD, donepezil was not beneficial in the treatment of hallucinations, hostility, suspiciousness, or unusual thought content (19). In an uncontrolled trial examining donepezil for hallucinations in DLB (N=13), scores were 4.6 points lower on the Behavioral Pathology in Alzheimer’s Disease rating scale following treatment, suggesting improvements (23).
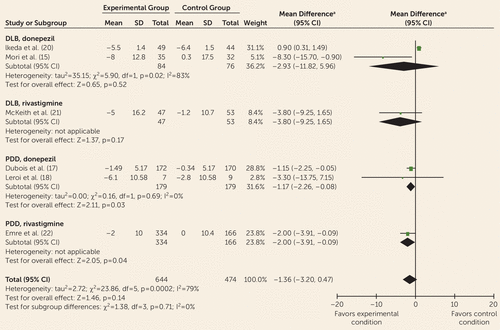
FIGURE 3. Forest Plot of Neuropsychiatric Symptoms on the 10-Item Neuropsychiatric Inventory, Comparing Donepezil or Rivastigmine With Placebo, in the Treatment of Dementia With Lewy Bodies (DLB) and Parkinson’s Disease Dementia (PDD) (Six Studies, N=1,118)
a Inverse variance method, random effects.
Three studies investigated activities of daily living in PDD using the Alzheimer’s Disease Cooperative Study–Activities of Daily Living Inventory, the Disability Assessment for Dementia, and the activities of daily living section of the Unified Parkinson’s Disease Rating Scale (17, 18, 22) (see Figure S2C in the data supplement). Standard deviations were estimated in two trials (17, 18). A significant effect of cholinesterase inhibitors was observed (standardized mean difference=0.2, 95% CI=0.07, 0.34), with subgroup analysis indicating a benefit of rivastigmine (standardized mean difference=0.21, 95% CI=0.02, 0.40) but not donepezil.
Eight studies reported adverse events and study withdrawals (15–22; see Figures S2D and S2E in the data supplement). Adverse events were more common in participants receiving cholinesterase inhibitors than those receiving placebos (80% compared with 70%; risk ratio=1.13, 95% CI=1.06, 1.21). Subgroup analysis indicated a greater risk ratio in DLB (92% compared with 75%; risk ratio=1.21, 95% CI=1.03, 1.43) and PDD (84% compared with 71%; risk ratio=1.18, 95% CI=1.06, 1.31) for rivastigmine, but not for donepezil in DLB or PDD. Adverse events that were reported more frequently in the treatment than the placebo groups were nausea (rivastigmine: DLB and PDD; donepezil: PDD), vomiting (rivastigmine: DLB and PDD), anorexia (rivastigmine: DLB), tremor (rivastigmine: PDD), somnolence (rivastigmine: DLB), dizziness (rivastigmine: PDD), and insomnia (donepezil: PDD). Overall, 25% of participants in the cholinesterase inhibitor treatments groups withdrew from studies, compared with 17% of those in placebo groups (risk ratio=1.47, 95% CI=1.16, 1.85). Subgroup analysis indicated that the risk of study withdrawal was significantly greater for participants with PDD treated with donepezil (23% compared with 15%; risk ratio=1.51, 95% CI=1.03, 2.23) and rivastigmine (27% compared with 18%; risk ratio=1.53, 95% CI=1.07, 2.18) than those receiving placebos, but there was no difference between placebo and donepezil or rivastigmine for participants with DLB. There were no between-group differences in motor function (weighted mean difference=−1.67, 95% CI=−4.02, 0.69), as assessed by four studies using the motor evaluation section of the Unified Parkinson’s Disease Rating Scale (15, 16, 18, 19) (see Figure S2F in the data supplement); standard deviations were estimated in one trial (18).
Results of an uncontrolled trial suggest that sudden withdrawal of cholinesterase inhibitors may be associated with deteriorations in cognition in DLB and PDD (−4.4 and −3.5 points, respectively, on the MMSE) and with increased psychiatric symptoms (NPI change, 22) for PDD (24).
Galantamine.
The highest level of evidence for galantamine was from an uncontrolled trial in DLB and a controlled trial in PDD (25, 26). In DLB (N=50), improvements were reported for cognitive fluctuations (1-day fluctuations scale change, −1.8), sleep disturbances (Pittsburgh Sleep Quality Index change, −3.2), and psychiatric symptoms (NPI change, −8.2) (25). Inconsistent results were reported for cognition. In PDD (galantamine, N=21; treatment as usual, N=20), improvements were reported for cognition (MMSE change difference=4.9), total psychiatric symptoms (NPI change difference=8.9), hallucinations (NPI hallucinations change difference=4.1), anxiety (NPI anxiety change difference=0.7), apathy (NPI apathy change difference=3.9), and sleep (NPI sleep change difference=1.4), that favored treatment with galantamine (26).
Discontinued drugs.
A small number of studies of metrifonate and tacrine were identified in our search. These drugs have been discontinued and are not discussed here.
Agents in Other Drug Classes
Memantine.
The highest level of evidence for memantine was one randomized controlled trial for PDD and two for mixed DLB and PDD samples (26–28). Three studies examined clinician impression of change; three examined improvement (Figure 4) (27–29), two absence of deterioration (27, 29) (see Figure S3A in the data supplement), and two continuous data (27, 29) (see Figure S3B in the data supplement). There were no significant between-group differences on improvement or absence of deterioration. Analysis of continuous data indicated that mean change scores were significantly lower (0.40 points) in participants treated with memantine (95% CI=−0.71, −0.10), suggesting improvements. Subgroup analysis indicated that this difference was significant for DLB (weighted mean difference=−0.60, 95% CI=−1.16, −0.04) but not for PDD.
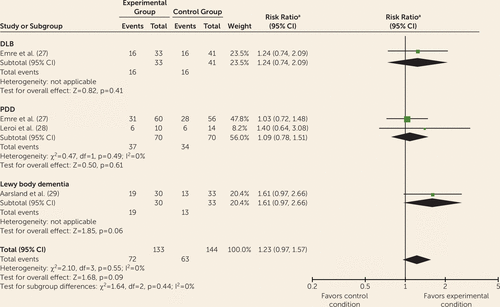
FIGURE 4. Forest Plot of Global Outcomes on the Alzheimer’s Disease Cooperative Study–Clinical Global Impression of Change (Assessing Improvement), Comparing Memantine With Placebo, in the Treatment of Dementia With Lewy Bodies (DLB) and Parkinson’s Disease Dementia (PDD) (Three Studies, N=277)
a Mantel-Haenszel method, fixed effect.
Two studies assessed cognition using the MMSE (28, 29) (see Figure S3C in the data supplement) and found no between-group differences. Three studies used the NPI to assess psychiatric symptoms (27–29) (see Figure S3D in the data supplement), with standard deviations estimated in two trials (27, 28). There were no between-group differences. The effect of memantine on activities of daily living was investigated in two studies, one using the Alzheimer’s Disease Cooperative Study–Activities of Daily Living Inventory, the other using the Disability Assessment for Dementia (see Figure S3E in the data supplement) (27, 28). There were no between-group differences. Three trials reported adverse events (see Figure S3F in the data supplement) and withdrawals (see Figure S3G in the data supplement) (27–29). There were no between-group differences on adverse events or withdrawals, nor on the motor evaluation section of the Unified Parkinson’s Disease Rating Scale (see Figure S3H in the data supplement).
Armodafinil/modafinil.
The highest level of evidence for armodafinil was from an uncontrolled trial (sleep) and a case series (global impression) in DLB, a case series (global impression) in PDD, and an uncontrolled trial (attention) in Lewy body dementia (30, 31). Results for DLB (N=17) suggest that treatment with armodafinil is associated with increased wakefulness (30). Data from self-reports and computer-based reaction tasks in Lewy body dementia (N=7) suggest improvements in subjective alertness and reflexive attention (31). In a retrospective review of treatment with modafinil or armodafinil (DLB, N=2; PDD, N=4), 50% of individuals were rated by clinicians as minimally improved, 33% as much improved, and 17% as not improved (31).
Piracetam.
The highest level of evidence for piracetam in PDD was from a randomized controlled trial (N=20) (32). No between-group differences were observed in cognition, motor function, or functional ability, except on a single subscale (engagement in recreational activities), which favored the piracetam group. No studies of piracetam for DLB were identified.
Antiparkinsonian Medications
Levodopa.
The highest level of evidence for levodopa in DLB and PDD was from four uncontrolled trials (33–36). The highest level of evidence for levodopa withdrawal in PDD was from a randomized controlled trial (37). Examinations of acute and chronic effects of levodopa suggest improvements in motor function and reductions in tremor for individuals with DLB and PDD (33–35). Beneficial effects of levodopa (i.e., 10% or more improvement in score on the motor evaluation section of the Unified Parkinson’s Disease Rating Scale) were more common in PDD (65%–70%) than in DLB (32%–50%) (33, 34, 36), although approximately one-third of those who derived motor benefits experienced increases in psychotic symptoms (34).
A randomized controlled trial in which withdrawal of levodopa in PDD was examined (N=11) showed that removing levodopa did not result in worsening cognition, motor function, or psychiatric symptoms (37).
Amantadine.
The highest level of evidence for amantadine in PDD was from an uncontrolled trial (PDD, N=10; Parkinson’s disease and cognitive impairment, N=15) (38). For participants with dementia, no significant effects of amantadine were observed on 13 of 15 cognitive tests. Statistically significant improvements were observed in total score on the Frontal Assessment Battery (3 points) and the inhibitory control subscale (0.3 points). No studies of amantadine for DLB were identified.
Rotigotine.
The highest levels of evidence for rotigotine in PDD were from one uncontrolled trial of Parkinson’s disease severity and one case series of anxiety (39, 40). Degree of disability due to Parkinson’s disease (N=9) and anxiety (N=2) were rated as less severe after treatment (Unified Parkinson’s Disease Rating Scale change, −12; Hamilton Anxiety Rating Scale change, −23). No studies of rotigotine for DLB were identified.
Selegiline.
The highest level of evidence for selegiline in PDD was from a cohort study (PDD, N=4; Parkinson’s disease without dementia, N=3) (41). No beneficial effects were observed for participants with dementia on measures of behavior, cognition, and motor function. No studies of selegiline for DLB were identified.
Antipsychotics
Clozapine.
The highest level of evidence for clozapine in PDD was a chart review (PDD, N=8; “other dementia,” N=8) (42). Scores on the Brief Agitation Rating Scale and the Cohen-Mansfield Agitation Inventory were significantly lower in the PDD group after treatment (−2.4 and −4.2, respectively), with 62.5% of patients rated as much improved. Side effects included drooling, sedation, tremors, constipation, and delirium. No studies of clozapine for DLB were identified.
Olanzapine.
The highest level of evidence for olanzapine was from a secondary analysis of a randomized controlled trial in Alzheimer’s disease in which participants were retrospectively identified as meeting DLB criteria and an uncontrolled trial in PDD (43, 44). In participants with possible DLB (N=29), those treated with 5 mg/day of olanzapine (N=10) showed greater reductions in scores on the NPI subscales for delusions (−3.8 points) and hallucinations (−5.9 points) than those receiving placebo (N=10). No significant differences were observed between the placebo group, the 10-mg group, and the 15-mg group (43). While no side effects were reported in that study, other authors have suggested that around 38% of patients with DLB do not tolerate olanzapine even at low dosages (2.5 mg/day) (45). In a sample in which three participants with PDD were treated with olanzapine (44), reductions were reported in delusions (NPI subscale, −3.2 points), hallucinations (NPI subscale, −0.97), and total psychiatric symptoms (NPI subscale, −15.1 points; Behavioral Pathology in Alzheimer’s Disease, −15 points). Worsening of motor function and psychiatric symptoms have been reported in 33%–80% of individuals with PDD following olanzapine treatment (44, 46).
Quetiapine.
The highest level of evidence for quetiapine was from a case series in DLB, a retrospective chart review in PDD, and a randomized controlled trial in Lewy body dementia (47–49). Reductions in psychiatric symptoms were reported for six of nine individuals with DLB following treatment with quetiapine (change in sum of NPI scores on the delusions, hallucinations, and agitation/aggression subscales, 7.7 points) (47). However, 33% of participants withdrew because of adverse events. For individuals with PDD and drug-induced psychosis, quetiapine was associated with worsening cognition and motor function without improvements to psychiatric status (48). A randomized placebo-controlled trial of quetiapine in Lewy body dementia (DLB, N=23; PDD, N=9; Alzheimer’s disease with parkinsonian features, N=8) revealed no between-group differences on measures of psychiatric symptoms, cognition, activities of daily living, motor function, or clinician’s impression of change (49).
Risperidone.
The highest level of evidence for risperidone was from a randomized trial in DLB and an uncontrolled trial in Parkinson’s disease dementia (50, 51). In participants with PDD and psychosis (N=9) significant reductions were observed in Brief Psychiatric Rating Scale score (−9.5 points) and Cohen-Mansfield Agitation Inventory score (−9.6 points), and improvements were seen in social, occupational, and psychological functioning (Global Assessment of Functioning change, 17 points) (50). No side effects were reported. In DLB, risperidone does not appear to be well tolerated; results of a randomized controlled trial (N=31) suggest deterioration in cognition (MMSE change, −2.3 points), worsening psychiatric symptoms (NPI change, 17.3 points), and study withdrawal (65%) (51).
Antidepressants
Citalopram/escitalopram, duloxetine, and trazodone.
The highest level of evidence for antidepressants was from a randomized trial of citalopram in DLB (51) and an unpublished trial of duloxetine, escitalopram, and trazodone in PDD (52). Citalopram does not appear to be efficacious or well tolerated in DLB, with 10 of 14 participants (71.4%) withdrawing because of adverse effects (51). In a trial of antidepressants for depression in PDD, the Montgomery-Åsberg Depression Rating Scale score was reduced (−18.6 points) in participants who were treated with duloxetine (N=8) (52). Individuals receiving escitalopram (N=7) and trazodone (N=8) were reported to have had reductions in depressive symptoms, but scores were not provided.
Sedatives
Clonazepam.
The highest level of evidence for clonazepam in DLB was from a case series in which two of three participants experienced reductions in the number of nights on which episodes of sleep disturbance occurred (53). No studies of clonazepam for PDD were identified.
Ramelteon.
The highest level of evidence for ramelteon in DLB was a case series (54). Descriptive statistics indicated reductions in neuropsychiatric symptoms, sleep disturbances, and self-reported caregiver burden, with no adverse effects. No studies of ramelteon for PDD were identified.
Anticonvulsants
Gabapentin.
The highest level of evidence for gabapentin was from case reports, one for DLB (55) and one for PDD (56). After treatment, reductions were reported in symptoms of restless leg syndrome in the patient with DLB and agitation in the patient with PDD.
Zonisamide.
The highest level of evidence for zonisamide was from case series of patients with DLB (57) and PDD (58). Descriptive reports of patients with DLB (N=3) suggest improvements in daily living skills, motor function, and caregiver burden in mild dementia and a reduction in psychiatric symptoms in severe dementia (57). Similar results were reported in a single case of PDD (58). Randomized controlled trials of zonisamide for motor function (ClinicalTrials.jp identifier, JapicCTI-122040) and for psychiatric symptoms (UMIN Clinical Trials Registry [umin.ac.jp] identifier, UMIN000010631) are under way.
Herbal Medicines
Yokukansan
The highest level of evidence for yokukansan was from a randomized crossover trial in DLB and an uncontrolled trial in PDD (59, 60). In participants with DLB randomized to receive yokukansan followed by no treatment (group A, N=9) or no treatment followed by yokukansan (group B, N=6), reductions were reported in psychiatric symptoms in both groups, although the change was statistically significant in group A only (NPI change: group A, −10.1 points; group B, −12.4 points) (59). In a trial of participants with PDD (N=7), there were significant differences in pre- and posttreatment NPI scores for total psychiatric symptoms (−6 points) and visual hallucinations (−2.6 points), but no differences on any other NPI subscales (60).
Patient and Caregiver Views of Management Strategies
No studies were identified that examined patient or caregiver views on pharmacological strategies for Lewy body dementia.
Cost-Effectiveness Analysis
Two studies reported economic data (61, 62). Estimates of cost per quality-adjusted life-year gained from treatment with cholinesterase inhibitors in Alzheimer’s disease and DLB suggested lower costs in the DLB group (61). However, in the DLB group there was considerable variability in the estimates of three models (microsimulation=£2,706; Markov=£35,922; Southampton Health Technology Assessment Centre=£46,794). In a study comparing rivastigmine with placebo for PDD, no significant between-group differences were reported for quality-adjusted life-days or total costs (62).
Discussion
We conducted a systematic review of pharmacological strategies for Lewy body dementia, identifying 28,568 potentially relevant papers. Forty-four papers examining 22 pharmacological strategies were included in our analyses. High-level evidence was rare, with only 17 randomized controlled trials. Methodological quality was rated as weak for 41% of included studies, moderate for 39%, and strong for 20%.
Data from controlled trials were available for donepezil, rivastigmine, galantamine, memantine, olanzapine, risperidone, piracetam, quetiapine, citalopram, and yokukansan. Meta-analyses indicated improvements with donepezil and rivastigmine for cognition, global psychiatric symptoms (in PDD only), hallucinations, delusions, and activities of daily living (without worsening motor symptoms of parkinsonism) but with adverse events. This is consistent with previous reviews of cholinesterase inhibitors for Lewy body dementia (7, 9). Evidence for galantamine suggests potential benefits for psychiatric symptoms and possibly for cognition, but the data are limited. Memantine appears to be well tolerated but provides few benefits to patients. Consistent with previous meta-analyses, memantine was superior to placebo only in terms of impression of change when analyzed as a continuous outcome measure; this advantage was not observed when analyzed as categorical data in terms of improvement or absence of deterioration. Recently published secondary analyses of memantine suggest some statistical advantages of memantine over placebo in relation to aspects of attention, sleep, caregiver burden, aspects of quality of life, and goal attainment (63–66). For olanzapine and quetiapine, reductions in psychiatric symptoms appear to be limited by high levels of adverse events. Citalopram, piracetam, and risperidone do not appear to be beneficial. Data are mixed on yokukansan for psychiatric symptoms.
There was weak evidence for potential efficacy of armodafinil/modafinil, levodopa, zonisamide, ramelteon, clonazepam, gabapentin, rotigotine, duloxetine, escitalopram, trazodone, and clozapine. These studies did not include controls, so we can conclude only that there could be an association between interventions and benefits to participants. Amantadine and selegiline do not appear to be effective for managing symptoms of Lewy body dementia, but data are available from only single trials or small samples. Overall, we must be cautious not to overstate the apparent effects, or lack of effects, given how few high-level studies are available for each strategy.
Limited data are available on costs of pharmacological strategies. We identified two studies indicating that donepezil and rivastigmine may not be cost-effective, as overall treatment costs were not significantly different compared with placebo groups, and estimates are typically above the threshold of £30,000 per quality-adjusted life-year for treatment, a cutoff often considered cost-effective. However, patents on these drugs have since expired, and cheaper generic versions are now available.
To date, only a small number of studies have investigated associations between treatment efficacy and participant characteristics. The results, which must be treated with caution, suggest a better response to levodopa among younger participants with DLB (36), a greater benefit from rivastigmine for cognition (global cognition, attention) in patients who have hallucinations (67, 68), and a greater benefit from rivastigmine for aspects of cognition and global neuropsychiatric symptoms among patients with elevated plasma homocysteine levels (69). In general, there is insufficient evidence on how, when, and where management strategies should be implemented.
Differing treatment effects between DLB and PDD have received little attention, but some differences are apparent. For example, quetiapine has some benefits for psychiatric symptoms among some patients with DLB but has shown a general lack of efficacy and adverse effects in PDD (47, 48), and levodopa appears to be more beneficial for PDD than DLB (36). Our meta-analyses suggest that the effects of donepezil and rivastigmine are comparable for DLB and PDD. Other studies suggest similar effects of donepezil in both groups (70). Overall, a lack of direct comparison hampers our ability to clarify differences in treatment effects. An additional caveat is the uncertainty about whether DLB and PDD are separate diseases; the 1-year rule for distinguishing the diagnoses is provided only as a guide for clinical practice (4).
A notable outcome from our review is a potential disconnect between research trials and the reality of clinical practice and the preferences of patients or caregivers. For example, patient-related outcomes and symptom-specific measures were rarely used, and apparent benefits of strategies were typically determined on the basis of statistical rather than clinical significance. Furthermore, no studies were identified on the views that patients with Lewy body dementia or their caregivers have of pharmacological management strategies. Research that focuses on areas of need reported by patients with Lewy body dementia and their families may provide useful information about which strategies to employ.
This review has a number of limitations. First, the evidence base is small. Even for the most well researched management strategies, there were few randomized controlled trials. Second, studies were affected by a variety of issues related to risk of bias and study quality, including open-label study designs, lack of control groups, small samples, and concurrent use of medications that may obscure the effects of study drugs. Furthermore, the accuracy of the diagnostic criteria for DLB and PDD is not clear, with diagnoses in some cases applied retrospectively (43). Third, research on pharmacological management strategies for Lewy body dementia has usually taken the form of efficacy studies. While these are important in establishing therapeutic effects under highly controlled circumstances, the results are not necessarily generalizable to clinical practice. For example, in research trials, samples are often homogeneous, interventions are standardized without scope for flexibility, concurrent treatments or comorbid conditions are not allowed, and patients with more severe difficulties may be less likely to be recruited or to participate. Effectiveness studies provide a way to address some of these concerns. Fifth, it was necessary to estimate missing information for our meta-analyses, as we were not able to obtain original data from study authors. Sixth, studies of management strategies that are used in clinical practice and have been recommended in expert opinion reviews were not included, because data for groups with DLB or PDD were not available. For example, in Parkinson’s disease, there is evidence from randomized controlled trials that pimavanserin and clozapine may be useful in treating psychotic symptoms in patients with lower cognition (71).
In summary, in this comprehensive review of pharmacological management strategies for DLB and PDD, we have identified the current best evidence for many key areas of need. There remain substantial gaps in our knowledge of patient and caregiver experiences, of cost-effectiveness, of how, when, and with whom strategies should be implemented, and of how meaningfully the results of studies translate to clinical practice.
1 : A systematic review of prevalence studies of dementia in Parkinson’s disease. Mov Disord 2005; 20:1255–1263Crossref, Medline, Google Scholar
2 : The prevalence and incidence of dementia with Lewy bodies: a systematic review of population and clinical studies. Psychol Med 2014; 44:673–683Crossref, Medline, Google Scholar
3 : Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 2007; 22:1689–1707Crossref, Medline, Google Scholar
4 : Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 2005; 65:1863–1872Crossref, Medline, Google Scholar
5 : Comparison of dementia with Lewy bodies to Alzheimer’s disease and Parkinson’s disease with dementia. Mov Disord 2004; 19:60–67Crossref, Medline, Google Scholar
6 : Intellectual, mnemonic, and frontal functions in dementia with Lewy bodies: a comparison with early and advanced Parkinson’s disease. Behav Neurol 1998; 11:173–183Crossref, Medline, Google Scholar
7 : Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson’s disease dementia, and cognitive impairment in Parkinson’s disease. Cochrane Database Syst Rev 2012; 3:CD006504Medline, Google Scholar
8 : Memantine for Lewy body disorders: systematic review and meta-analysis. Am J Geriatr Psychiatry 2015; 23:373–383Crossref, Medline, Google Scholar
9 : Efficacy and safety of cholinesterase inhibitors and memantine in cognitive impairment in Parkinson’s disease, Parkinson’s disease dementia, and dementia with Lewy bodies: systematic review with meta-analysis and trial sequential analysis. J Neurol Neurosurg Psychiatry 2015; 86:135–143Crossref, Medline, Google Scholar
10
11
12 : A process for systematically reviewing the literature: providing the research evidence for public health nursing interventions. Worldviews Evid Based Nurs 2004; 1:176–184Crossref, Medline, Google Scholar
13 : Assessment of study quality for systematic reviews: a comparison of the Cochrane Collaboration Risk of Bias Tool and the Effective Public Health Practice Project Quality Assessment Tool: methodological research. J Eval Clin Pract 2012; 18:12–18Crossref, Medline, Google Scholar
14 Selecting studies and collecting data, in Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0 (updated March 2011). Edited by Higgins JPT, Green S (www.cochrane-handbook.org)Google Scholar
15 : Donepezil for dementia with Lewy bodies: a randomized, placebo-controlled trial. Ann Neurol 2012; 72:41–52Crossref, Medline, Google Scholar
16 : Donepezil for cognitive impairment in Parkinson’s disease: a randomised controlled study. J Neurol Neurosurg Psychiatry 2002; 72:708–712Crossref, Medline, Google Scholar
17 : Donepezil in Parkinson’s disease dementia: a randomized, double-blind efficacy and safety study. Mov Disord 2012; 27:1230–1238Crossref, Medline, Google Scholar
18 : Randomized placebo-controlled trial of donepezil in cognitive impairment in Parkinson’s disease. Int J Geriatr Psychiatry 2004; 19:1–8Crossref, Medline, Google Scholar
19 : Donepezil for dementia in Parkinson’s disease: a randomised, double blind, placebo controlled, crossover study. J Neurol Neurosurg Psychiatry 2005; 76:934–939Crossref, Medline, Google Scholar
20 : Donepezil for dementia with Lewy bodies: a randomized, placebo-controlled, confirmatory phase III trial. Alzheimers Res Ther 2015; 7:4Crossref, Medline, Google Scholar
21 : Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet 2000; 356:2031–2036Crossref, Medline, Google Scholar
22 : Rivastigmine for dementia associated with Parkinson’s disease. N Engl J Med 2004; 351:2509–2518Crossref, Medline, Google Scholar
23 : Improved visual hallucination by donepezil and occipital glucose metabolism in dementia with Lewy bodies: the Osaki-Tajiri project. Eur Neurol 2010; 64:337–344Crossref, Medline, Google Scholar
24 : What happens when donepezil is suddenly withdrawn? An open label trial in dementia with Lewy bodies and Parkinson’s disease with dementia. Int J Geriatr Psychiatry 2003; 18:988–993Crossref, Medline, Google Scholar
25 : Efficacy and safety of galantamine in patients with dementia with Lewy bodies: a 24-week open-label study. Dement Geriatr Cogn Disord 2007; 23:401–405Crossref, Medline, Google Scholar
26 : [Efficacy and safety of galantamine (Reminyl) in the treatment of dementia in patients with Parkinson’s disease (open-label controlled trial)]. Zh Nevrol Psikhiatr Im S S Korsakova 2007; 107:25–33 (Russian)Medline, Google Scholar
27 : Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol 2010; 9:969–977Crossref, Medline, Google Scholar
28 : Randomized controlled trial of memantine in dementia associated with Parkinson’s disease. Mov Disord 2009; 24:1217–1221Crossref, Medline, Google Scholar
29 : Memantine in patients with Parkinson’s disease dementia or dementia with Lewy bodies: a double-blind, placebo-controlled, multicentre trial. Lancet Neurol 2009; 8:613–618Crossref, Medline, Google Scholar
30 Pilot study of armodafinil in patients with dementia with Lewy bodies. Clinicaltrials.gov identifier: NCT01023672 (http://www.clinicaltrials.gov/ct2/show/NCT01023672)Google Scholar
31 : Modafinil and armodafinil improve attention and global mental status in Lewy bodies disorders: preliminary evidence. Int J Geriatr Psychiatry 2013; 28:1095–1097Crossref, Medline, Google Scholar
32 : A controlled trial of piracetam in intellectually impaired patients with Parkinson’s disease. Mov Disord 1990; 5:230–234Crossref, Medline, Google Scholar
33 : L-dopa responsiveness in dementia with Lewy bodies, Parkinson disease with and without dementia. Neurology 2004; 63:376–378Crossref, Medline, Google Scholar
34 : Effects of dopaminergic medications on psychosis and motor function in dementia with Lewy bodies. Mov Disord 2008; 23:2248–2250Crossref, Medline, Google Scholar
35 : Cohort study of prevalence and phenomenology of tremor in dementia with Lewy bodies. J Neurol 2013; 260:1731–1742Crossref, Medline, Google Scholar
36 : The role of levodopa in the management of dementia with Lewy bodies. J Neurol Neurosurg Psychiatry 2005; 76:1200–1203Crossref, Medline, Google Scholar
37 : The effects of withdrawal of dopaminergic medication in nursing home patients with advanced parkinsonism. J Am Med Dir Assoc 2008; 9:670–675Crossref, Medline, Google Scholar
38 : Effects of amantadine sulfate on cognitive impairments in patients with Parkinson’s disease. Neurosci Behav Physiol 2011; 41:567–573Crossref, Google Scholar
39 : Rotigotine and demented Parkinson’s disease patients: a therapeutic option. Presented at the Movement Disorder Society’s 15th International Congress of Parkinson’s Disease and Movement Disorders, Toronto, June 5–9, 2011Google Scholar
40 : Rotigotine for anxiety during wearing-off in Parkinson’s disease with dementia. Aging Clin Exp Res 2013; 25:601–603Crossref, Medline, Google Scholar
41 : The effect of deprenyl (selegiline) on cognition and emotion in parkinsonian patients undergoing long-term levodopa treatment. Acta Neurol Scand Suppl 1983; 95:135–144Crossref, Medline, Google Scholar
42 : Clozapine for treatment-resistant agitation in dementia. J Geriatr Psychiatry Neurol 2007; 20:178–182Crossref, Medline, Google Scholar
43 : Efficacy of olanzapine in the treatment of psychosis in dementia with Lewy bodies. Dement Geriatr Cogn Disord 2002; 13:67–73Crossref, Medline, Google Scholar
44 : Olanzapine as a treatment of neuropsychiatric disorders of Alzheimer’s disease and other dementias: a 24-month follow-up of 68 patients. Am J Alzheimers Dis Other Demen 2003; 18:205–214Crossref, Medline, Google Scholar
45 : Olanzapine in dementia with Lewy bodies: a clinical study. Int J Geriatr Psychiatry 1999; 14:459–466Crossref, Medline, Google Scholar
46 : Olanzapine for the treatment of psychosis in patients with Parkinson’s disease and dementia. Psychosomatics 2001; 42:477–481Crossref, Medline, Google Scholar
47 : Quetiapine treatment of psychotic symptoms and aggressive behavior in patients with dementia with Lewy bodies: a case series. Prog Neuropsychopharmacol Biol Psychiatry 2003; 27:549–553Crossref, Medline, Google Scholar
48 : The effect of quetiapine in psychotic Parkinsonian patients with and without dementia: an open-labeled study utilizing a structured interview. J Neurol 2006; 253:171–175Crossref, Medline, Google Scholar
49 : Quetiapine for agitation or psychosis in patients with dementia and parkinsonism. Neurology 2007; 68:1356–1363Crossref, Medline, Google Scholar
50 : The use of risperidone for psychosis and agitation in demented patients with Parkinson’s disease. J Neuropsychiatry Clin Neurosci 1997; 9:594–597Crossref, Medline, Google Scholar
51 : Treating neuropsychiatric symptoms in dementia with Lewy bodies: a randomized controlled trial. Alzheimer Dis Assoc Disord 2010; 24:360–364Crossref, Medline, Google Scholar
52 : The efficacy of psychopharmacologic treatment in depressive disorders associated with Parkinson’s disease dementia. Presented at the 18th European Congress of Psychiatry, Munich, Feb 27 to March 2, 2010Google Scholar
53 : Drug treatment of REM sleep behavior disorders in dementia with Lewy bodies. Int Psychogeriatr 2003; 15:377–383Crossref, Medline, Google Scholar
54 : Effectiveness of ramelteon for treatment of visual hallucinations in dementia with Lewy bodies: a report of 4 cases. J Clin Psychopharmacol 2013; 33:581–583Crossref, Medline, Google Scholar
55 : Effects of gabapentin enacarbil on restless legs syndrome and leg pain in dementia with Lewy bodies. Psychogeriatrics 2014; 14:132–134Crossref, Medline, Google Scholar
56 : A retrospective chart review of gabapentin for the treatment of aggressive and agitated behavior in patients with dementias. Am J Geriatr Psychiatry 2000; 8:221–225Crossref, Medline, Google Scholar
57 : Administration of zonisamide in three cases of dementia with Lewy bodies. Psychiatry Clin Neurosci 2010; 64:327–329Crossref, Medline, Google Scholar
58 : Zonisamide for seizures in Parkinson’s disease with dementia. Seizure 2013; 22:324–325Crossref, Medline, Google Scholar
59 : A randomized cross-over study of a traditional Japanese medicine (kampo), yokukansan, in the treatment of the behavioural and psychological symptoms of dementia. Int J Neuropsychopharmacol 2009; 12:191–199Crossref, Medline, Google Scholar
60 : Successful treatment with yokukansan for behavioral and psychological symptoms of Parkinsonian dementia. Prog Neuropsychopharmacol Biol Psychiatry 2010; 34:284–287Crossref, Medline, Google Scholar
61 : Economic evaluation of cholinesterase inhibitor therapy for dementia: comparison of Alzheimer’s disease and dementia with Lewy bodies. Int J Geriatr Psychiatry 2009; 24:1072–1078Crossref, Medline, Google Scholar
62 : Economic evaluation of rivastigmine in patients with Parkinson’s disease dementia. Pharmacoeconomics 2006; 24:93–106Crossref, Medline, Google Scholar
63 : The effect of memantine on sleep behaviour in dementia with Lewy bodies and Parkinson’s disease dementia. Int J Geriatr Psychiatry 2010; 25:1030–1038Crossref, Medline, Google Scholar
64 : Quality of life and the effect of memantine in dementia with lewy bodies and Parkinson’s disease dementia. Dement Geriatr Cogn Disord 2011; 32:227–234Crossref, Medline, Google Scholar
65 : Memantine improves goal attainment and reduces caregiver burden in Parkinson’s disease with dementia. Int J Geriatr Psychiatry 2014; 29:899–905Crossref, Medline, Google Scholar
66 : Memantine improves attention and episodic memory in Parkinson’s disease dementia and dementia with Lewy bodies. Int J Geriatr Psychiatry 2015; 30:46–54 Crossref, Medline, Google Scholar
67 : Hallucinations predict attentional improvements with rivastigmine in dementia with lewy bodies. Dement Geriatr Cogn Disord 2004; 18:94–100Crossref, Medline, Google Scholar
68 : Effects of rivastigmine in patients with and without visual hallucinations in dementia associated with Parkinson’s disease. Mov Disord 2006; 21:1899–1907Crossref, Medline, Google Scholar
69 : Rivastigmine versus placebo in hyperhomocysteinemic Parkinson’s disease dementia patients. Mov Disord 2008; 23:1532–1540Crossref, Medline, Google Scholar
70 : A comparison of the efficacy of donepezil in Parkinson’s disease with dementia and dementia with Lewy bodies. Int J Geriatr Psychiatry 2005; 20:938–944Crossref, Medline, Google Scholar
71 : Pharmacological treatment of Parkinson disease: a review. JAMA 2014; 311:1670–1683Crossref, Medline, Google Scholar



