Changes in Regional Cerebral Blood Flow With Venlafaxine in the Treatment of Major Depression
Abstract
OBJECTIVE: Neuroimaging studies reveal abnormalities of regional cerebral blood flow (rCBF) in major depression. In this study the authors prospectively investigated rCBF and clinical response to venlafaxine, a novel antidepressant. METHOD: A trial of venlafaxine was performed with seven patients referred with ICD-10 major depression. At entry and 6-week follow-up, the Beck Depression Inventory and Hamilton Depression Rating Scale were administered and rCBF was measured by means of single photon emission computed tomography with [99mTc]hexamethylpropyleneamine oxime. Blood flow changes were explored with statistical parametric mapping. RESULTS: The subjects showed significant improvement after treatment. Statistical parametric mapping analysis revealed increased rCBF bilaterally in the thalamus and decreased rCBF in the left occipital lobe, right cerebellum, and temporal cortex bilaterally. CONCLUSIONS: These data confirm limbic cortical rCBF changes associated with effective antidepressant treatment.
Several theories postulate regional brain dysfunction in cortical and subcortical regions associated with affective disorders (1–3). Few studies have followed a patient group with imaging before and after treatment with a single antidepressant.
The imaging data thus far (1, 2) suggest that abnormalities in regional cerebral blood flow (rCBF) accompany depression and are altered by treatment. We used [99mTc]hexamethylpropyleneamine oxime single photon emission computed tomography ([99mTc]HMPAO SPECT) to test the hypothesis that treatment with the novel antidepressant venlafaxine is associated with rCBF changes in limbic and frontal regions. Venlafaxine has a main action at serotonergic and noradrenergic receptors (4).
Method
Permission for the study from the local research ethics committee and the U.K. Administration of Radioactive Substances Advisory Committee was obtained. After complete description of the study to the subjects, written informed consent was obtained.
The inclusion criteria were an ICD-10 diagnosis of depressive episode, an entry score on the Hamilton Depression Rating Scale (5) of 18–30, no previous history of depression, and medication-free status at study entry. The exclusion criteria were comorbid psychiatric disorder, physical disorder, substance misuse, and pregnancy or breast-feeding. Seven patients with major depression, two men and five women, were recruited.
Baseline and posttreatment SPECT scans were performed. The patients were medication free for at least 2 weeks before the baseline scan. After the baseline scan each subject received venlafaxine, 37.5 mg b.i.d., for 6 weeks. A second scan was then performed. The 21-item Beck Depression Inventory (6) and 17-item Hamilton depression scale (5) were applied at entry and 6-week follow-up by a single rater (J.D.).
Cerebral blood flow imaging was carried out by using 99mTc HMPAO SPECT. The scanning was performed with a Camstar XRT gamma camera (GE Medical Systems, Waukesha, Wis.) with Nuclear Diagnostics software (Nuclear Diagnostics AB, Stockholm) and a 128×128-pixel matrix (resolution full width at half maximum, 12 mm). The scans took 32 minutes and proceeded in a 360° circular arc of 64 positions from an anterior projection around the head, with 30 seconds per angle, commencing 2 minutes after intravenous injection of 500 MBq of [99mTc]HMPAO. The scanning was performed in a consistent, quiet environment at rest. The gamma emissions from the patient were subject to low-energy general-purpose collimation before detection by the gamma camera. Brain images were computed in the oblique transaxial, sagittal, and coronal planes.
The scans were transferred to a workstation and processed by using SPM 99 statistical parametric mapping image analysis software (Wellcome Department of Cognitive Neurology, London; http://www.fil.ion.bpmf.ac.uk/spm).
The scans were spatially normalized to the positron emission tomography (PET) CBF template provided with SPM 99 by using the option for linear spatial normalization. The spatially normalized scans were smoothed by using an isotropic gaussian spatial filter with a resolution of 12 mm full width at half maximum before the voxel-wise statistical analysis. Normalization of global voxel intensity was achieved by using proportional scaling.
The scans were statistically analyzed by using the appropriate SPM 99 module (7). The baseline scans were compared vowel-wise to the posttreatment scans by using the statistics option “multisubject, different conditions.”
Results
The seven subjects (two men, five women) were all right-handed. Their mean age was 43 years (SD=9, range=33–60). All but two of the patients had been drug free for at least 1 year. Their mean pretreatment scores on the Hamilton depression scale and Beck Depression Inventory were 25.00 (SD=4.24) and 24.71 (SD=1.38), respectively, and the posttreatment Hamilton and Beck scores were 6.29 (SD=2.50) and 3.28 (SD=0.76), respectively. The mean percentages of improvement in scores with treatment were 40% for the Hamilton depression scale and 52% for the Beck Depression Inventory (for each scale, z=–2.38, two-tailed p=0.02, Wilcoxon matched-pairs signed rank test). At 2-year follow-up no patient had relapsed.
The results of statistical parametric mapping analysis showed a significant increase in blood flow after treatment in the thalamus bilaterally (Figure 1). Significant rCBF decreases were also noted in the right posterior temporal lobe (Brodmann’s area 37), bilateral superior temporal gyrus (Brodmann’s area 21/22), left occipital lobe (Brodmann’s area 7), and right cerebellum. All results were thresholded at an uncorrected p<0.001 (for voxel height) and a corrected p<0.04 (for spatial extent).
Discussion
This is one of the first prospective studies confirming changes in cerebral blood flow after effective treatment with venlafaxine in drug-free, moderately ill subjects. Venlafaxine treatment resulted in clinical improvement in all cases. We have substantiated changes in the limbic system identified in previous studies (8, 9).
PET and SPECT studies (8, 10) have shown significantly lower baseline rCBF in patients than in healthy comparison subjects. In a PET study of venlafaxine for depressed outpatients (11), baseline prefrontal and paralimbic hypometabolism was associated with antidepressant efficacy. Attempts to understand data from this field are complicated by many differences between studies, e.g., patient characteristics (severity, duration, and stage of illness), medication, and scanning techniques and instruments.
One explanation for the lack of significant changes in the frontal cortex in this study may be a type II error (false negative). The image-averaging technique used in SPM 99 is a 12-parameter affine transform that, while accounting for translation, rotation, size, and shape of the whole brain, does not make corrections for differences in individual gyri. SPM 99 software can correct for detailed differences in anatomy, but the correction is not satisfactory for images of limited resolution (∼12 mm). False negative results may result from the small number of subjects in this study. Nevertheless, important differences were found in distributed limbic and thalamic networks, and the possibility that moderate illness is not in fact associated with frontal dysfunction warrants consideration.
Identifying rCBF changes after successful treatment provides useful biological markers to guide discovery of novel drugs. Further studies in this area, with larger patient cohorts, are needed to identify blood flow patterns that predict response to treatment.
Received Dec. 27, 2001; revision received June 6, 2002; accepted July 11, 2002. From the Mental Health Research Group, Peninsula Medical School; the Department of Nuclear Medicine, Derriford Hospital, Plymouth, U.K.; the Department of Clinical Physics, Southern General Hospital, Glasgow Research Unit, Glasgow, U.K.; and the Institute of Psychiatry, Kings College London. Address reprint requests to Dr. Lloyd, Mental Health Research Group, Peninsula Medical School, Wonford House Hospital, Exeter EX2 5AF, U.K.; [email protected] (e-mail). Supported by Wyeth Laboratories. Dr. Pilowsky is a U.K. Medical Research Council Senior Research Fellow. The authors thank Dr. Gillian Vivien, Dr. Robert Ward, and Professor S.D. Martin.
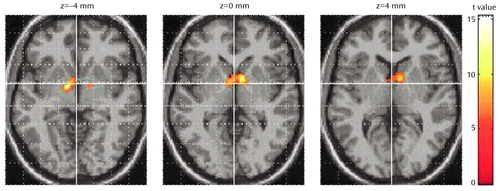
Figure 1. Statistical Parametric Maps of a Significant Increase in Regional Cerebral Blood Flow (rCBF) in the Thalamus Bilaterally of Seven Patients With Major Depression After Treatment With Venlafaxine
1. Post RM, DeLisi LE, Holcomb HH, Uhde TW, Cohen R, Buchsbaum MS: Glucose utilization in the temporal cortex of affectively ill patients: positron emission tomography. Biol Psychiatry 1987; 22:545-553Crossref, Medline, Google Scholar
2. Schlegel S, Aldenhoff JB, Eissner D, Lindner P, Nickel O: Regional cerebral blood flow in depression: association with psychopathology. J Affect Disord 1989; 17:211-218Crossref, Medline, Google Scholar
3. Bench CJ, Friston KJ, Brown RG, Scott LC, Frackowiak RSJ, Dolan RJ: The anatomy of melancholia—focal abnormalities of cerebral blood flow in major depression. Psychol Med 1992; 22:607-615Crossref, Medline, Google Scholar
4. Effexor—Venlafaxine Hydrochloride. Taplow, Maidenhead, Berkshire, UK, Wyeth Laboratories, January 1999Google Scholar
5. Hamilton M: Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol 1967; 6:278-296Crossref, Medline, Google Scholar
6. Beck AT, Rush AJ, Shaw BF, Emery G: Cognitive Therapy of Depression. Chichester, UK, John Wiley & Sons, 1979, pp 1-230Google Scholar
7. Ashburner J, Friston K: Multimodal image coregistration and partitioning—a unified framework. Neuroimage 1997; 6:209-217Crossref, Medline, Google Scholar
8. Martin SD, Martin E, Rai SS, Richardson MA, Royall R: Brain blood flow changes in depressed patients treated with interpersonal psychotherapy or venlafaxine hydrochloride: preliminary findings. Arch Gen Psychiatry 2001; 58:641-648Crossref, Medline, Google Scholar
9. Mayberg HS, Lewis PJ, Regenold W, Wagner HN Jr: Paralimbic hypoperfusion in unipolar depression. J Nucl Med 1994; 35:929-934Medline, Google Scholar
10. Sackeim HA, Prohovnik I, Mueller JR, Brown RP, Apter S, Prudic J, Devanand DP, Mukherjee S: Regional cerebral blood flow in mood disorders, I: comparison of major depressives and normal controls at rest. Arch Gen Psychiatry 1990; 47:60-70Crossref, Medline, Google Scholar
11. Little JT, Ketter TA, Kimbrell TA, Danielson A, Benson B, Willis MW, Post RM: Venlafaxine or bupropion responders but not nonresponders show baseline prefrontal and paralimbic hypometabolism compared with controls. Psychopharmacol Bull 1996; 32:629-635Medline, Google Scholar



