Decreased Presynaptic Dopamine Function in the Left Caudate of Depressed Patients With Affective Flattening and Psychomotor Retardation
Abstract
OBJECTIVE: The study assessed striatal presynaptic dopamine function in patients with different subtypes of depression. METHOD: Magnetic resonance imaging and positron emission tomography with [18F]fluorodopa ([18F]DOPA) were used to compare six depressed patients with marked affective flattening and psychomotor retardation, six depressed patients with marked impulsivity and anxiety, and 10 healthy comparison subjects. Depressed patient groups were matched for severity of depression. RESULTS: [18F]DOPA uptake Ki values in the left caudate were significantly lower in patients with psychomotor retardation than in patients with high impulsivity and in comparison subjects. CONCLUSIONS: These results suggest that left caudate dopamine function differs between depressed patients with psychomotor retardation and those with impulsivity and provide direct evidence of a link between dopamine hypofunction and psychomotor retardation in depression.
Several studies have reported that the concentration of the dopamine metabolite homovanillic acid in cerebrospinal fluid is lower in depressed patients than in healthy comparison subjects and that this difference is particularly pronounced in patients with marked psychomotor retardation (e.g., reference 1). These findings suggest that central dopamine function might be impaired in these patients. Furthermore, antidepressant drugs that enhance dopamine transmission might improve depression in patients with psychomotor retardation (2). Although such preliminary observations could have treatment implications, no study that we are aware of has directly investigated presynaptic dopamine function in patients with different clinical subtypes of depression. We assessed presynaptic dopamine function by using positron emission tomography (PET) and 6-[18F]fluorodopa ([18F]DOPA) in depressed patients with psychomotor retardation, in depressed patients with high impulsivity, and in healthy comparison subjects.
Method
Twelve right-handed inpatients with a major depressive disorder diagnosed according to DSM-IV criteria and with a depression score >25 on the Montgomery-Åsberg Depression Rating Scale (3) were recruited from consecutive admissions at three teaching hospitals. Only depressed patients with either marked depressive retardation (score >18 on the Depressive Retardation Rating Scale [4]) or marked impulsivity (scores >10 on the loss-of-control subscale of the Depressive Mood Scale [5]) and >10 on the Tyrer anxiety scale [6]) were included. Thus, two groups with clearcut depression subtypes were defined: 1) six depressed patients with psychomotor retardation, who had high affective flattening and low loss-of-control scores on the Depressive Mood Scale subscales (two male patients and four female patients; mean age=51.8 years, SD=7.6; mean Montgomery-Åsberg Depression Rating Scale score=30.5, SD=3.9; mean Depressive Retardation Rating Scale score=22.0, SD=3.2; mean Depressive Mood Scale affective flattening subscale score=15.5, SD=5.4; mean Depressive Mood Scale loss-of-control subscale score=6.0, SD=3.6; and mean Tyrer anxiety scale score=13.7, SD=4.6) and 2) six depressed patients with high impulsivity and low retardation scores (one male patient and five female patients; mean age=40.2 years, SD=8.7; mean Montgomery-Åsberg Depression Rating Scale score=27.7, SD=3.1; mean Depressive Retardation Rating Scale score=11.7, SD=4.2; mean Depressive Mood Scale affective flattening subscale score=7.7, SD=7.4; mean Depressive Mood Scale loss-of-control subscale score=14.5, SD=3.3; and mean Tyrer anxiety scale score=20.3, SD=3.1). Although both groups had comparable mean scores on the Montgomery-Åsberg Depression Rating Scale (Mann-Whitney U test, U=9, p=0.15), clinical differences were significant on all other scales: Depressive Retardation Rating Scale (U=1, p=0.006), Depressive Mood Scale affective flattening subscale (U=5, p=0.04), Depressive Mood Scale loss-of-control subscale (U=36, p=0.004), and Tyrer anxiety scale (U=3, p=0.02).
Only drug-free patients (N=6, three in each group) and patients taking selective serotonin reuptake inhibitors (SSRIs) (N=6, three in each group) were included. Patients who had been treated in the previous month with drugs known to directly modulate dopamine neurotransmission were excluded. Other exclusion criteria were neurological and general medical conditions, pregnancy, and DSM-IV axis I comorbidity.
Ten right-handed healthy comparison subjects (four male subjects and six female subjects; mean age=42.2 years, SD=12.1) were selected from a group of community volunteers. The comparison group was similar to the patient group in age (Kruskal-Wallis test, H=3.5, df=2, p=0.17) and gender (χ2=0.95, df=1, p=0.62). The comparison subjects had no history of neurological or psychiatric conditions.
After complete description of the study to the subjects, written informed consent was obtained.
Anatomic magnetic resonance imaging (MRI) and [18F]DOPA PET brain scans were performed (7). Briefly, in the MRI protocol, 124 T1-weighted images (1.5-mm thickness) were acquired with a 1.5-T Signa scanner (General Electric, Milwaukee). After a transmission scan for attenuation correction and injection of 2–5.6 mCi of [18F]DOPA (prepared as previously described [8]; mean=3.4 mCi, SD=0.9, in comparison subjects; mean=3.6 mCi, SD=1.2, in depressed patients with impulsivity; and mean=3.7 mCi, SD=0.8, in depressed patients with psychomotor retardation, Kruskal-Wallis test, H=0.27, df=2, p=0.87), 63 slices (2.5-mm thick) were obtained with an ECAT-HR+ tomograph (Siemens, Knoxville, Tenn.). Data for nine time frames of 10 minutes each were collected. Attenuation- and decay-corrected PET images were summed and aligned with the MRI images. Investigators who were blind to the clinical ratings determined regions of interest on the four or five aligned MRI slices in which the caudate and putamen were readily visible, and the regions were copied onto the PET images. Occipital cortex regions of interest, in which [18F]DOPA uptake was assumed to be negligible, were used as reference regions. Regional [18F]DOPA uptake Ki values were computed from the regional time-activity curves obtained between 30 and 90 minutes by using the Patlak graphical analysis method (9).
Group comparisons for quantitative variables were performed by using the Kruskal-Wallis nonparametric test. Pairwise comparisons were performed with the Mann-Whitney U test. Significance was set at 0.05, two-tailed.
Results
The mean uptake Ki values in the patients with psychomotor retardation, the patients with impulsivity, and the comparison subjects were, respectively, 0.0126 (SD=0.0008), 0.0123 (SD=0.0007), and 0.0126 (SD=0.0004) in the right caudate (Kruskal-Wallis test, H=0.85, df=2, p=0.65); 0.0112 (SD=0.0008), 0.0127 (SD=0.0007), and 0.0127 (SD=0.0007) in the left caudate (H=10.5, df=2, p=0.005); 0.0129 (SD=0.0010), 0.0123 (SD=0.0007), and 0.0130 (SD=0.0009) in the right putamen (H=2.1, df=2, p=0.34); and 0.0124 (SD=0.0009), 0.0123 (SD=0.0004), and 0.0126 (SD=0.0008) in the left putamen (H=1.4, df=2, p=0.49).
For the left caudate, comparisons showed that the mean Ki value in the patients with psychomotor retardation was significantly lower than that in the impulsive patients (U=2, p=0.01) and in the comparison subjects (U=2, p=0.002). The mean Ki values in the left caudate of the impulsive patients and the comparison subjects were not significantly different (U=28, p=0.83) (Figure 1).
There was no effect of age on Ki values in either the caudate (r=0.17, N=22, z=–0.77, p=0.26) or the putamen (r=0.27, N=22, z=1.21, p=0.62).
Discussion
Decreased presynaptic dopamine function was found in the left caudate of depressed patients with affective flattening and psychomotor retardation. The caudate is part of a loop involving basal ganglia, anterior cingulate, thalamus, and prefrontal cortex that has been hypothesized to modulate mood (10), and lesions of the left caudate have been associated with a higher frequency and severity of depression (11). Our results are consistent with findings of a negative correlation between depressive symptoms and left striatal [18F]DOPA uptake in patients with schizophrenia (12), decreased caudate glucose metabolism in depressed patients (13), and a negative correlation between psychomotor retardation and regional cerebral blood flow in the left prefrontal cortex in depressed patients (14).
Although previous treatment with SSRIs in some patients might have had an indirect effect on dopamine transmission, it is unlikely that such an effect would involve the left caudate only and appear only in depressives with psychomotor retardation.
Thus, although the number of patients in this study was small, the results suggest a link between dopamine hypofunction in left caudate pathways and depressive psychomotor retardation with affective flattening. Further studies are needed to examine the role of the caudate in depressive disorders and to determine whether antidepressants that directly enhance dopamine transmission would be useful in treating these patients.
Presented at the 55th annual meeting of the Society of Biological Psychiatry, Chicago, May 11–13, 2000, and at the sixth annual meeting of the Organization for Human Brain Mapping, San Antonio, Tex., June 12–16, 2000. Received Jan. 12, 2000; revision received Aug. 1, 2000; accepted Aug. 31, 2000. From Unité 334, Institut National de la Santé et de la Recherche Médicale, Service Hospitalier Frédéric Joliot, Division des Sciences du Vivant, Département de Recherche Medicale, Commissariat à l’Énergie Atomique, Orsay, France; Centre National de la Recherche Scientifique, Unité Mixte de Recherche 7593, Paris; and the Departments of Psychiatry at Hôpital de la Salpêtrière; Hôpital Albert Chenevier, Créteil, France; and Hôpital de Bicêtre, Le Kremlin-Bicêtre, France. Address reprint requests to Dr. Paillère Martinot, Service de Psychiatrie, Hôpital de la Salpêtrière, 47 Boulevard de l’Hôpital, 75013 Paris, France; [email protected] (e-mail).
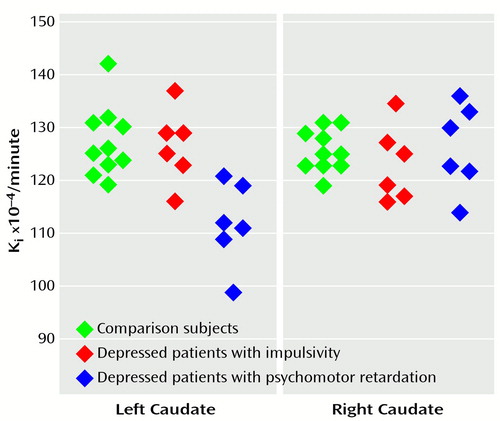
Figure 1. [18F]Fluorodopa Uptake Ki Values in the Left and Right Caudate of Healthy Subjects (N=10), Depressed Patients With Impulsivity (N=6), and Depressed Patients With Psychomotor Retardation (N=6)
1. van Praag HM, Korf J: Retarded depression and the dopamine metabolism. Psychopharmacologia 1971; 19:199–203Crossref, Medline, Google Scholar
2. Rampello L, Nicoletti G, Raffaele R: Dopaminergic hypothesis for retarded depression: a symptom profile for predicting therapeutical responses. Acta Psychiatr Scand 1991; 84:552–554Crossref, Medline, Google Scholar
3. Montgomery SA, Åsberg M: A new depression scale designed to be sensitive to change. Br J Psychiatry 1979; 134:382–389Crossref, Medline, Google Scholar
4. Widlöcher D, Ghozlan A: The measurement of retardation in depression, in Human Psychopharmacology: Measures and Methods, vol 2. Edited by Hindmarch I, Stonier PD. New York, John Wiley & Sons, 1989, pp 1–22Google Scholar
5. Jouvent R, Vindreau C, Montreuil M, Bungener C, Widlöcher D: Polydimensional rating scale of depressive mood. Psychiatrie & Psychobiologie 1988; 3:245–253Google Scholar
6. Tyrer P, Owen RT, Crochetti DV: The brief scale for anxiety: a subdivision of the comprehensive psychopathological rating scale. J Neurol Neurosurg Psychiatry 1984; 47:970–975Crossref, Medline, Google Scholar
7. Dao-Castellana MH, Paillère Martinot ML, Hantraye P, Attar-Lévy D, Rémy P, Crouzel C, Artiges E, Féline A, Syrota A, Martinot JL: Presynaptic dopaminergic function in the striatum of schizophrenic patients. Schizophr Res 1997; 23:167–174Crossref, Medline, Google Scholar
8. Dollé F, Demphel S, Hinnen F, Fournier D, Vaufrey F, Crouzel C:6-[18F]Fluoro-l-DOPA by radiofluorodestannylation: a short and simple synthesis of a new labelling precursor. J Labelled Compounds & Radiopharmaceuticals 1998; 41:105–114Google Scholar
9. Patlak CS, Blasberg RG: Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data: generalizations. J Cereb Blood Flow Metab 1985; 5:584–590Crossref, Medline, Google Scholar
10. Swerdlow NR, Koob GF: Dopamine, schizophrenia, mania, and depression: toward a unified hypothesis of cortico-striato-pallido-thalamic function. Behav Brain Sci 1987; 10:197–245Crossref, Google Scholar
11. Starkstein SE, Robinson RG, Berthier ML, Parikh RM, Price TR: Differential mood changes following basal ganglia vs thalamic lesions. Arch Neurol 1988; 45:725–730Crossref, Medline, Google Scholar
12. Hietala J, Syvälahti E, Vilkman H, Vuorio K, Räkköläinen V, Bergman J, Haaparanta M, Solin O, Kuoppamäki M, Eronen E, Ruotsalainen U, Salokangas RK: Depressive symptoms and presynaptic dopamine function in neuroleptic-naive schizophrenia. Schizophr Res 1999; 35:41–50Crossref, Medline, Google Scholar
13. Baxter LR, Phelps ME, Mazziotta JC, Schwartz JM, Gerner RH, Selin CE, Sumida RM: Cerebral metabolic rates for glucose in mood disorders: studies with positron emission tomography and fluorodeoxyglucose F 18. Arch Gen Psychiatry 1985; 42:441–447Crossref, Medline, Google Scholar
14. Bench CJ, Friston KJ, Brown RG, Frackowiak RS, Dolan RJ: Regional cerebral blood flow in depression measured by positron emission tomography: the relationship with clinical dimensions. Psychol Med 1993; 23:579–590Crossref, Medline, Google Scholar



