Efficacy of Quetiapine Monotherapy in Posttraumatic Stress Disorder: A Randomized, Placebo-Controlled Trial
Abstract
Objective:
This was a 12-week randomized, placebo-controlled trial to assess the efficacy of quetiapine monotherapy in the treatment of posttraumatic stress disorder (PTSD).
Method:
Eighty patients were randomly assigned to treatment with either quetiapine or placebo. The primary outcome measure was the Clinician-Administered PTSD Scale (CAPS). Secondary efficacy measures included the CAPS subscales, the Davidson Trauma Scale, the Positive and Negative Syndrome Scale (PANSS), the Clinical Global Impressions (CGI) scales for severity of Illness and improvement, the Hamilton Depression Rating Scale (HAM-D), and the Hamilton Anxiety Rating Scale (HAM-A). Safety measurements included adverse events, vital signs, the Abnormal Involuntary Movement Scale, the Barnes Akathisia Scale, the Simpson-Angus Scale, and the Arizona Sexual Experiences Scale.
Results:
After a 1-week placebo run-in, quetiapine was started at a daily dosage of 25 mg and increased to a maximum of 800 mg; the average was 258 mg (range, 50–800 mg). Reductions in CAPS total, re-experiencing, and hyperarousal scores were significantly greater for the quetiapine group than for the placebo group. Greater improvements were also observed for quetiapine in scores on the Davidson Trauma Scale, CGI severity and improvement ratings, PANSS positive symptom and general psychopathology subscales, HAM-A, and HAM-D than for placebo. Adverse events were generally mild and expected based on prior studies of quetiapine in this and other patient population. There were no differences in safety measures between groups.
Conclusion:
Quetiapine monotherapy was efficacious in the treatment of PTSD. These findings suggest quetiapine as a single agent is effective in treating military PTSD.
Posttraumatic stress disorder (PTSD) is often a chronic, disabling illness. The estimated lifetime prevalence of PTSD in the United States is approximately 7.8% (1), but rates are much higher in specific trauma populations, such as combat veterans (2). PTSD is characterized by exposure to psychological trauma and the following symptom clusters: re-experiencing of the event, avoidance and numbing, changes in cognition, and hyperarousal (3).
There is extensive comorbidity between PTSD and anxiety, mood, and substance use disorders (1). Psychotic symptoms may be present in up to 40% of veterans with chronic PTSD (4, 5). In fact, the severity of positive, negative, and general psychopathology symptoms may approach that of chronic schizophrenia (6).
Antidepressant medications are considered a primary medication mainstay of treatment for PTSD. Two selective serotonin reuptake inhibitor antidepressants (SSRIs), sertraline and paroxetine, are the only agents approved by the U.S. Food and Drug Administration (FDA) for treatment of PTSD (7). However, many chronic PTSD patients, especially male combat veterans, have a limited or no response to antidepressants (8–11).
Second-generation or atypical antipsychotics have been utilized either as monotherapy or as adjunctive therapy in PTSD patients with or without psychotic features (12–14). Two double-blind, placebo-controlled studies found that olanzapine was effective in reducing PTSD symptoms (15, 16).
Several double-blind, placebo-controlled studies of risperidone as adjunctive treatment in PTSD reported benefit (reviewed in reference 17). However, a large trial in 296 military veterans with PTSD found that risperidone as an add-on to SSRIs was no better than placebo in improving PTSD severity (18), underscoring how difficult it is to treat this population.
Quetiapine is an atypical antipsychotic that is FDA-approved for the treatment of schizophrenia, bipolar disorder, and bipolar depression. Quetiapine is an antagonist of the serotonin 5-HT2 and dopamine D2 receptors (19); this profile probably mediates its antipsychotic properties. Quetiapine is also an antagonist of the 5-HT2A receptor, partial agonist of the 5-HT1A receptor, and antagonist of the histamine 1 (H1) receptor and the α1/α2-noradrenergic receptors (19). Quetiapine increases neuropeptide Y levels and lowers corticotropin-releasing hormone (CRH) in cerebrospinal fluid (CSF) (20). Furthermore, norquetiapine, the main quetiapine metabolite, is a norepinephrine reuptake inhibitor (reviewed in reference 21). This pharmacologic profile suggests that quetiapine has specific properties that can be helpful in the treatment of PTSD, particularly in re-experiencing and hyperarousal symptoms, as well as associated depressive and anxiety symptoms.
Insomnia and other sleep disturbances are often the most refractory symptoms in this population. Hamner and colleagues treated 20 combat veterans with PTSD and demonstrated good tolerability of quetiapine and a significant improvement in scores on the Clinician-Administered PTSD Scale (CAPS) (22) as well as improvement in sleep (23). Additional case reports and open trials have also supported the potential efficacy of quetiapine in PTSD (reviewed in 17). A retrospective chart review study found that quetiapine was as effective as prazosin in the short-term management of nighttime symptoms (24).
The objective of this randomized, placebo-controlled, double-blind study of quetiapine was to test the efficacy of this medication as monotherapy in the treatment of military PTSD. We hypothesized that quetiapine would reduce the severity of PTSD, particularly of re-experiencing and hyperarousal symptoms. Although in current practice quetiapine and other second-generation antipsychotics are recommended as adjunctive therapies (Veterans Administration/Department of Defense guidelines, 2010 [25]), we felt it would be useful to establish the role of monotherapy with one of these agents, particularly in the difficult-to-treat military population.
Method
Subject Population
Patients meeting DSM-IV criteria for chronic PTSD were recruited for the study between 2004 and 2008 from two sites: the Ralph H. Johnson VA Medical Center in Charleston, S.C., and the Raymond G. Murphy VA Medical Center in Albuquerque, N.M. The study was approved by the Research and Development Committees from both hospitals and the Institutional Review Boards of the Medical University of South Carolina and the University of New Mexico School of Medicine. After the participants signed informed consent statements, screening procedures were conducted. Patients were included in the study if they were 18 to 65 years of age and met DSM-IV criteria for PTSD, as established with the Clinician-Administered PTSD Scale Diagnostic Version for DSM-IV (CAPS-DX) (26). Patients were also assessed with the Structured Clinical Interview for DSM-IV Axis I Disorders, Patient Edition (SCID-I/P) (27). Veterans of both genders and any ethnic background were recruited. Participants had to have a score of at least 50 on the CAPS at baseline and be capable of giving informed consent. Female patients were required to use a medically approved contraceptive or otherwise not be of child-bearing potential. Patients did not take any psychotropic medications or herbal remedies within 1 week prior to randomization (2 weeks for fluoxetine) and during the course of the study except for rescue medications (see below). Medications for medical indications were held constant for 1 month prior to study entry. Exclusion criteria included history of sensitivity to quetiapine, use of psychotropic medications within 1 week prior to randomization and throughout the duration of the study (except for short-term use as rescue medication as specified in the Concomitant Medications section), and medical disorders that may preclude safe administration of quetiapine or exacerbate anxiety symptoms. Diabetes mellitus was not an exclusion criterion. Additional reasons for exclusion were alcohol or substance abuse or dependence within 1 month of study entry as defined by DSM-IV criteria; schizophrenia, schizoaffective disorder, bipolar disorder, or dementia; suicidal or homicidal ideation; current pursuit of compensation or an increase in compensation for the effects of trauma; and initiation or change in psychotherapy within 3 months of randomization.
Study Procedures
Efficacy measures.
The primary outcome measure was the total CAPS score. The re-experiencing, avoidance, and hyperarousal subscales of the CAPS were also analyzed as secondary efficacy measures. Other secondary efficacy measures included the Davidson Trauma Scale, the Positive and Negative Syndrome Scale (PANSS), the Clinical Global Impressions Scale (CGI) ratings for improvement and severity of illness, the Hamilton Depression Rating Scale (HAM-D), the Hamilton Anxiety Rating Scale (HAM-A), and the Pittsburgh Sleep Quality Index.
Study treatment.
During the screening visit, patients received a physical examination, medical and psychiatric histories were recorded, and measurements were made of weight, height, heart rate, respiratory rate, and sitting blood pressure. Clinical laboratory tests were done if indicated by the medical history; these included measurement of serum electrolytes, creatinine, blood urea nitrogen, and glucose; liver function tests; complete blood cell count; urinalysis; and urine drug screen. Eligible patients then received a 1-week, single-blind placebo lead-in. Placebo nonresponders (participants who had less than a 30% reduction in CAPS total score) were then randomly assigned to receive double-blind quetiapine or placebo tablets for 12 weeks. The subjects were randomly assigned to quetiapine or placebo in a 1:1 ratio by using a computer program for random number assignment. The participants were evaluated at weeks 1, 2, 4, 8, and 12. The tablets were administered in a flexible-dose titration based on tolerability and clinical response. The study medication was initiated at a dose of 25 mg at bedtime and gradually titrated to 400 mg daily by the end of week 2 as tolerated. Further dose increases or decreases were allowed up to a maximum of 800 mg daily of study medication and a minimum of 50 mg daily.
Safety measures.
Safety measures included assessment of adverse events and vital signs at each study visit. The following scales were completed at the baseline and last visits: Abnormal Involuntary Movement Scale (AIMS), Simpson-Angus Scale, Barnes Akathisia Scale, and Arizona Sexual Experiences Scale. Laboratory tests were completed only at baseline due to budgetary constraints.
Concomitant Medications
Additional rescue medications were allowed during the placebo lead-in phase and during the first 2 weeks of the randomized phase. These included chloral hydrate up to 1000 mg daily for insomnia or agitation and lorazepam up to 3 mg daily for anxiety or agitation. Patients requiring rescue medications beyond the first 2 weeks of the randomized phase were discontinued from the study. In addition, the use of rescue medications was discouraged and minimized.
Statistical Analyses
The changes in total CAPS score in the two treatment arms (quetiapine and placebo) were compared by means of analysis of covariance (ANCOVA) with an intent-to-treat approach and the last observation carried forward, as described below. The comparison of the secondary efficacy and safety measures between the two treatment arms was done by means of mixed-model analysis of variance (ANOVA) with drug as a fixed factor and visit (baseline and weeks 2, 4, 8, and 12) as a repeated factor, using SAS version 9.2 (SAS Institute, Cary, N.C.). All statistical tests are two-sided with a significance level of 0.05.
Results
Of the screened patients, 119 entered the single-blind phase; 39 dropped out for various reasons, and 80 (40 at each site) were randomly assigned to quetiapine or placebo (see Figure 1). Six patients required rescue medication during the first 2 weeks of double-blind treatment, but none were excluded because of this. Patient demographic characteristics are shown in Table 1. Forty-two subjects were randomly assigned to quetiapine and 38 to placebo. Thirteen patients (31%) dropped out of the quetiapine group, and 20 patients (53%) dropped out of the placebo group (Fisher’s exact test, p=0.07). The mean age of the participants was 52 years, with no significant difference between the quetiapine and placebo groups. The patients in the quetiapine group had slightly more education (mean=14.2 years, SD=2.4) than those in the placebo group (mean=13.07 years, SD=2.3) (t=–2.13, df=77.474, p=0.04). The majority of the patients were male combat veterans. There was no difference in the percentage of males between groups (see Table 1). Race distributions were also similar in the two groups. Twenty-one of the patients were African American, 17 were Native American, and 42 were Caucasian. It should be noted that more Hispanic and Native American patients were seen at the New Mexico site and more African-American patients were seen at the Charleston site.
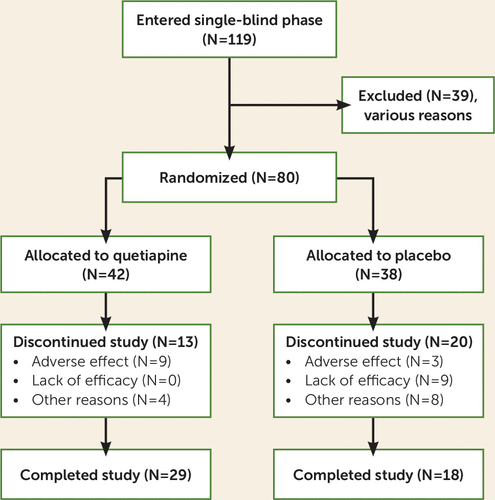
FIGURE 1. Patient Flow in Study of Quetiapine Monotherapy for PTSD
| Variable | Quetiapine (N=42) | Placebo (N=38) | Difference | ||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | pa | |
| Age (years) | 52 | 12 | 54 | 10 | 0.36 |
| Education (years) | 14.2 | 2.4 | 13.1 | 2.3 | 0.04 |
| N | % | N | % | p | |
| Gender | 0.20b | ||||
| Male | 38 | 90 | 37 | 97 | |
| Female | 4 | 10 | 1 | 3 | |
| Dropouts | 13 | 31 | 20 | 52 | 0.07c |
| Race | 0.68b | ||||
| Caucasian | 21 | 50 | 21 | 55 | |
| African American | 12 | 29 | 9 | 24 | |
| American Indian | 9 | 21 | 8 | 21 | |
TABLE 1. Demographic Variables for PTSD Patients in Study of Quetiapine
Efficacy Results
The main efficacy measures were conducted at baseline and at weeks 2, 4, 8, and 12. The mean total baseline CAPS scores were similar in the quetiapine and placebo groups, 75 (SD=16) versus 71 (SD=12) (t=−0.76, p=0.45). However, DSM-IV cluster B scores (re-experiencing) were higher in the quetiapine group (mean=21, SD=7) than in the placebo group (mean=17, SD=15) (t=2.4, p=0.02). For this reason, the main outcome analysis was ANCOVA utilizing the baseline CAPS score as a covariate, drug as a fixed factor, and visit as a repeated factor. We found a significant effect of the interaction of visit and treatment condition (F=2.88, df=4, 240, p=0.03), indicating that the quetiapine group had a greater drop in total CAPS score than the placebo group (see Figure 2). A logistic regression of the binary variable for dropouts was not significant (p=0.50), which is consistent with dropouts being “completely at random” and not related to the subjects’ last CAPS value, nor were dropouts related to which treatment group the subject was in (p=0.42). An intent-to-treat-analysis of the full repeated measures (ANCOVA) model gave a significant treatment-by-visit interaction (F=2.94, df=4, 312, p=0.02).
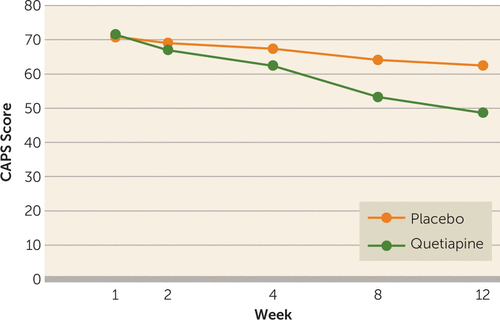
FIGURE 2. Clinician-Administered PTSD Scale (CAPS) Scores for PTSD Patients in Study of Quetiapinea
a Mean total CAPS score at each visit. Intent-to-treat analysis was used in the analysis of covariance with baseline CAPS score as a covariate, using the last observation carried forward. The interaction between treatment and visit was significant (F=2.94, df=4, 312, p=0.02).
The results of the secondary outcome measures were as follows. The quetiapine group had greater improvements than the placebo group on the CAPS re-experiencing subscale (F=12.7, df=1, 246, p=0.0004) and the hyperarousal subscale (F=7.43, df=1, 246, p=0.007) but not on the avoidance/numbing subscale (F=2.28, df=1, 246, p=0.13). Efficacy results are shown in Table 2.
| Quetiapine (N=42) | Placebo (N=38) | Effect Size (Cohen’s d)b | ||||
|---|---|---|---|---|---|---|
| Measure | Mean | SD | Mean | SD | p | |
| CAPS total | ||||||
| Baseline | 75.40 | 16.0 | 70.60 | 11.7 | ||
| Endpoint | 53.85 | 26.4 | 65.66 | 20.8 | 0.49 | 0.02c |
| CAPS re-experiencing | ||||||
| Baseline | 20.66 | 6.8 | 17.18 | 6.1 | ||
| Endpoint | 11.71 | 8.2 | 16.06 | 7.9 | 0.54 | 0.0004d |
| CAPS avoidance/numbing | ||||||
| Baseline | 30.02 | 7.4 | 29.55 | 7.5 | ||
| Endpoint | 25.54 | 12.2 | 28.30 | 10.5 | 0.24 | 0.13d |
| CAPS hyperarousal | ||||||
| Baseline | 24.71 | 5.3 | 23.86 | 4.7 | ||
| Endpoint | 16.60 | 9.7 | 21.30 | 6.8 | 0.56 | 0.007d |
| Davidson Trauma Scale | ||||||
| Baseline | 91.38 | 22.6 | 84.47 | 22.5 | ||
| Endpoint | 63.82 | 35.3 | 73.73 | 29.6 | 0.30 | 0.03e |
| HAM-D | ||||||
| Baseline | 20.40 | 5.2 | 19.76 | 6.4 | ||
| Endpoint | 13.40 | 7.1 | 18.00 | 7.3 | 0.64 | 0.02e |
| HAM-A | ||||||
| Baseline | 19.97 | 6.3 | 18.78 | 5.7 | ||
| Endpoint | 13.57 | 7.6 | 16.60 | 6.9 | 0.41 | 0.01e |
| Pittsburgh Sleep Quality Index | ||||||
| Baseline | 11.00 | 4.3 | 10.44 | 4.7 | ||
| Endpoint | 7.69 | 6.2 | 8.64 | 4.2 | 0.17 | 0.20e |
| PANSS, global psychopathology | ||||||
| Baseline | 34.81 | 6.4 | 34.68 | 7.5 | ||
| Endpoint | 28.97 | 5.7 | 32.46 | 7.7 | 0.50 | 0.005e |
| PANSS, positive symptoms | ||||||
| Baseline | 12.71 | 2.9 | 11.89 | 3.2 | ||
| Endpoint | 11.05 | 2.6 | 12.03 | 3.4 | 0.32 | 0.002e |
| PANSS, negative symptoms | ||||||
| Baseline | 14.28 | 4.1 | 13.73 | 4.0 | ||
| Endpoint | 13.11 | 4.0 | 13.40 | 3.8 | 0.07 | 0.16e |
| CGI severity | ||||||
| Baseline | 4.85 | 0.7 | 4.76 | 0.7 | ||
| Endpoint | 3.97 | 1.1 | 4.43 | 0.9 | 0.45 | 0.01e |
| CGI improvement | ||||||
| Baseline | ||||||
| Endpoint | 2.57 | 1.2 | 3.50 | 1.3 | 0.75 | <0.01f |
TABLE 2. Efficacy Measures for PTSD Patients in Study of Quetiapinea
We compared the Davidson Trauma Scale scores of the two groups with a mixed-model ANOVA with drug as a fixed factor and visit as a repeated factor. We found greater improvement in the quetiapine group (F=4.89, df=1, 246, p=0.03). A similar analysis for the CGI severity rating revealed greater improvement in the quetiapine group (F=3.35, df=4, 240, p=0.01). The CGI improvement scores were lower (better) at week 12 for quetiapine than for placebo (t=2.98, df=63, p<0.01) (see Table 2).
We also found a significant treatment-by-visit interaction for the HAM-D (F=2.88, df=4, 240, p=0.02) and the HAM-A (F=6.76, df=1, 63, p=0.01), indicating greater improvement in depression and anxiety scores in the quetiapine group (see Table 2).
The Pittsburgh Sleep Quality Index was administered at baseline and weeks 4, 8, and 12. We found a treatment effect (F=6.22, df=1, 77, p<0.05), but the visit-by-treatment interaction was nonsignificant (F=1.49, df=3, 1175, p=0.20) (see Figure 3).
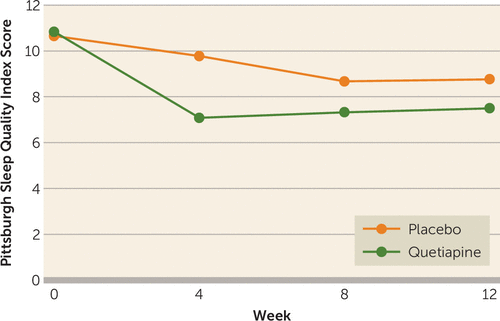
FIGURE 3. Pittsburgh Sleep Quality Index Scores for PTSD Patients in Study of Quetiapinea
a Mixed-model analysis of variance with drug as a fixed factor and visit as a repeated factor. The effect of treatment was significant (F=6.22, df=1, 77, p<0.05), but the visit-by-treatment interaction was not (F=1.49, df=3, 1175, p=0.20).
The PANSS was administered at weeks 0 and 12. The analysis of the PANSS global psychopathology and positive symptom subscales showed significant treatment-by-visit interactions (global: F=8.51, df=1, 63, p=0.005; positive symptoms: F=10.39, 4, 240, p=0.002, respectively), indicating greater improvement in the quetiapine group. However, we did not find a significant treatment-by-visit interaction for the negative symptom subscale (F=1.99, df=1, 63, p=0.16).
Safety Reports
The adverse events were generally mild and consistent with the known safety profile of quetiapine. The most common side effects in the quetiapine group were dry mouth (15.8%), somnolence (13.4%), and sedation (7.4%). Nine patients dropped out of the quetiapine group because of adverse effects, while three dropped out of the placebo group for that reason. There were no significant differences in weight, pulse, or blood pressure measurements between the quetiapine-treated patients and the placebo group (see Table 3). The safety scales were administered at weeks 0 and 12. There were no significant differences on these scales between quetiapine and placebo (see Table 3).
| Quetiapine (N=42) | Placebo (N=38) | ||||
|---|---|---|---|---|---|
| Variable | Mean | SD | Mean | SD | pb |
| Arizona Sexual Experiences Scale score | |||||
| Baseline | 19.66 | 0.92 | 18.97 | 0.95 | |
| Endpoint | 20.14 | 0.99 | 18.53 | 1.01 | 0.21 |
| AIMS score | |||||
| Baseline | 0.57 | 0.18 | 0.86 | 0.22 | |
| Endpoint | 0.65 | 0.19 | 0.67 | 0.18 | 0.56 |
| Barnes Akathisia Scale score | |||||
| Baseline | 1.76 | 0.32 | 1.78 | 0.34 | |
| Endpoint | 1.88 | 0.33 | 1.42 | 0.29 | 0.50 |
| Simpson-Angus Scale score | |||||
| Baseline | 2.14 | 0.33 | 1.31 | 0.40 | |
| Endpoint | 1.20 | 0.34 | 1.23 | 0.33 | 0.78 |
| Weight (lb) | |||||
| Baseline | 224.5 | 50.3 | 196.3 | 39.2 | |
| Endpoint | 220.7 | 44.6 | 197.7 | 38.2 | 0.25 |
| Systolic blood pressure (mm Hg) | |||||
| Baseline | 128.8 | 17.4 | 125.8 | 14.7 | |
| Endpoint | 125.2 | 17.9 | 128.4 | 14.9 | 0.67 |
| Diastolic blood pressure (mm Hg) | |||||
| Baseline | 75.95 | 10.38 | 75.63 | 10.10 | |
| Endpoint | 75.97 | 11.77 | 77.91 | 9.36 | 0.88 |
TABLE 3. Safety Measures for PTSD Patients in Study of Quetiapinea
Dose
The final dose range of quetiapine for all patients who had at least one efficacy assessment visit (N=41) was 50 to 800 mg daily. The average quetiapine dose was 258 mg daily. The final dose range of placebo in patients who had at least one efficacy assessment (N=38) was also 50 to 800 mg daily with an average dose of 463 mg daily. Since dose was considered an ordinal outcome and unlikely to be normally distributed, a nonparametric test (chi-square analysis) was utilized to compare doses of quetiapine and placebo. Based on this analysis, the dose of placebo was significantly higher than the quetiapine dose (χ2=13.75, df=1, p=0.0002). No patients dropped out of the quetiapine group because of lack of efficacy, and nine patients dropped out of the placebo group for that reason (see Figure 1).
Discussion
The primary outcome measure, the total CAPS score, demonstrated significantly greater improvement in the quetiapine-treated group than in the placebo group. That the dose of placebo was almost twice that of quetiapine confirms the significance of this finding, implying that clinicians continued to titrate the placebo to a higher dose due to lack of efficacy. Furthermore, while there were no dropouts in the quetiapine group due to lack of efficacy, nine patients dropped out of the placebo group for that reason. These results are remarkable, given the severity, chronicity, and difficult-to-treat nature of this population.
Total Davidson Trauma Scale scores and as hypothesized, CAPS re-experiencing and hyperarousal subscale scores, demonstrated significant improvement with quetiapine versus placebo. In contrast, and consistent with a review of atypicals in PTSD (17), scores on the CAPS avoidance/numbing subscale and the PANSS negative symptom subscale did not differ from those of the placebo group. In addition, the CGI improvement and severity, HAM-A, HAM-D, and PANSS positive symptom and general psychopathology subscale scores showed improvement in the active medication group. Of particular interest is the improvement in depression ratings (HAM-D), since the frequency of comorbid depression in PTSD is common and no antidepressant medication was used.
It is not surprising that in this study neither negative or avoidance symptoms responded during this relatively short-term trial. It may take longer for these symptoms to improve as patients adopt more healthy behaviors with improvement in re-experiencing and hyperarousal symptoms. Consistent with our findings, in a review of PTSD treatment with atypicals, re-experiencing and hyperarousal symptoms responded better to these medications (17).
A surprising finding was the Pittsburgh Sleep Quality Index results. We hypothesized improved sleep measures with quetiapine. We did find a treatment effect, but the treatment-by-visit interaction was not significant. The graph in Figure 3 suggests better sleep response in the quetiapine group by week 4 (visit 5) that was lost at subsequent visits. This needs to be further investigated.
This study was consistent with our earlier open trial and with several case reports and open trials of quetiapine in PTSD (22), reviewed by Ahearn et al. (17). These studies have largely investigated the use of quetiapine as an adjunctive therapy. To our knowledge, the present study is the first double-blind placebo-controlled trial of quetiapine as monotherapy in PTSD.
Interestingly, in spite of the paucity of evidence of quetiapine’s usefulness in PTSD, this medication and other atypicals are widely used to treat this condition. Studies in VA populations have shown high rates of atypical prescriptions for PTSD. Bauer and colleagues (28) analyzed data on 732,085 veterans with PTSD treated between 2003 and 2010 and found that 27.6% of them received an atypical for this condition. Hermes et al. conducted a survey of 2,613 VA providers and found that 13% of quetiapine prescriptions were given for a sole PTSD diagnosis (29). Furthermore, a retrospective chart review found that quetiapine was as effective as prazosin in treating nightmares, but patients taking quetiapine were more likely to discontinue medication due to side effects (24).
Other studies with quetiapine have also demonstrated a potentially unique effect in depression, particularly in patients with anxious features (30). In fact, quetiapine has FDA approval for treatment of bipolar depression. Moreover, there is increasing interest in treating comorbid psychiatric symptoms in PTSD (31), including psychotic symptoms (22), nightmares, (32), and depression, in addition to other comorbidities. The depressive symptoms are often secondary to PTSD or overlap with specific PTSD symptoms, e.g., sleep disturbances or anhedonia. Our findings indicate that quetiapine as a single agent significantly improved depressive symptoms in PTSD patients.
Quetiapine has a unique pharmacological profile, partly mediated by its metabolite norquetiapine, with a combination of effects on the serotonin, norepinephrine, and dopamine systems (19, 21). There is also evidence that quetiapine increases neuropeptide Y and lowers CRH in the CSF. This unique profile may explain the beneficial effects of quetiapine on PTSD, depression, and anxiety symptoms. The antihistaminergic effects may be desirable in this population to facilitate sleep. The α1-adrenergic antagonist properties may contribute to reduction in nightmares and associated sleep difficulties, as suggested by studies with prazosin, which is an α1-adrenergic antagonist (32). D2 receptor antagonist effects may affect a variety of symptoms, including intrusive memories, flashbacks, and comorbid psychotic symptoms. The higher ratio of HT2 receptor effects to D2 effects may contribute to the low extrapyramidal side effect profile. The relatively lower D2 receptor antagonist effects may also contribute to a lack of sustained prolactin elevation and associated side effects with quetiapine (33). Moreover, the rapid dissociation of quetiapine from the dopamine receptor (analogous to clozapine) may be sufficient to contribute to efficacy while yielding fewer extrapyramidal or prolactin-related side effects.
In summary, current practice guidelines support the use of atypical antipsychotics as one of several medication options in patients who are refractory or only partially responsive to antidepressants and psychotherapy, but they also note that data on use of these agents are limited (25, 34). Our findings suggest that quetiapine as a single agent is effective in the treatment of PTSD and associated depression and anxiety symptoms. The level of improvement observed with quetiapine suggests it may be superior for the treatment of PTSD over other antipsychotics, such as risperidone, which did not improve global PTSD symptoms in a large study with veterans (18).
Quetiapine was well tolerated. The main side effect was somnolence, which was not a significant problem, as no subjects discontinued the study on its account. There were no differences in vital signs, weight, or extrapyramidal symptoms between groups. However, a word of caution is in order because of increasing concerns about metabolic side effects of the new-generation antipsychotics (35). Diabetes mellitus was not an exclusion criterion; in fact, six subjects (15.8%) in the placebo group had diabetes and one (2.6%) had a history of hyperglycemia. In the quetiapine group, 10 subjects (23.8%) had a diabetes diagnosis and one (2.4%) had a history of hyperglycemia. Only one of the diabetic patients was not receiving diabetes treatment. Thirty-one patients were taking statins, and four were receiving gemfibrozil. Unfortunately, glucose and hemoglobin A1c were not monitored longitudinally, but the fact that we did not find weight gain in the quetiapine group indicates this medication was a safe short-term treatment. When quetiapine is used, weight, lipids, and hemoglobin A1c should be monitored closely. If metabolic changes are detected, the quetiapine dose should be lowered or switching to another medication should be considered.
Although improved, the patients receiving quetiapine remained symptomatic, with a mean CAPS score around 54. Therefore, in a clinical setting, additional psychopharmacological or psychotherapeutic interventions would need to be considered.
These initial results are encouraging because chronic military PTSD is often refractory to a variety of treatments, but the findings need replication. We hope that more studies will be conducted to better define the role of quetiapine and other atypical antipsychotics in patients suffering from PTSD. We would like to point out that we finished the study in 2008, but we feel our findings continue to be relevant since atypicals are often prescribed in PTSD and there are still relatively few studies testing their efficacy in this condition.
Patient Perspective
“Mr. Y” was a 54-year-old married Hispanic man with combat-related PTSD and alcohol use disorder in sustained remission. He worked as a salesperson but was having difficulties due to his PTSD symptoms. He was referred from the local veterans’ center by his therapist for further treatment. He was new to our system and had never tried medications. His more prominent symptoms were insomnia, nightmares, intrusive memories, and irritability. After his initial evaluation he was invited to participate in the study and he consented. He was started on study medication, titrated to 400 mg at bedtime. By week 2 Mr. Y reported he was sleeping better and stated, “I don’t know what is in those pills, but is working.” He also reported “a loss of strength on the intrusive memories…; they are easier to deal with.” The only side effect he reported was worsening of his chronic tinnitus. At the week 4 visit Mr. Y said he had had a bad week. He was having more sleep problems, intrusions, and irritability. However, by week 8 he was feeling better again. He reported that his memories of the war used to be 90% traumatic and 10% nontraumatic and now they were “50-50.” He was also noted to have more insight into his emotional reactions. For example, he reported that he had reacted angrily to a customer but later on realized this and apologized to the customer. This was unusual for him. Mr. Y’s improvement continued at week 12, the last study visit. After the study ended, it was revealed that Mr. Y had been in the group receiving quetiapine. He was particularly pleased with his improvement in sleep and asked to continue receiving the medication. Mr. Y was started on open-label quetiapine. He was again rapidly titrated to 400 mg at bedtime. Improvement continued at the 1-month poststudy visit, and the medication regimen was continued.
1 : Posttraumatic stress disorder in the National Comorbidity Survey. Arch Gen Psychiatry 1995; 52:1048–1060Crossref, Medline, Google Scholar
2 : Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med 2004; 351:13–22Crossref, Medline, Google Scholar
3 : Diagnostic and Statistic Manual for Mental Disorders, 5th ed. Washington, DC, APA, 2013Crossref, Google Scholar
4 : Psychotic features and combat-associated PTSD. Depress Anxiety 1997; 5:34–38Crossref, Medline, Google Scholar
5 : Psychotic symptoms in combat-related posttraumatic stress disorder. J Clin Psychiatry 1999; 60:29–32Crossref, Medline, Google Scholar
6 : Psychotic features in chronic posttraumatic stress disorder and schizophrenia: comparative severity. J Nerv Ment Dis 2000; 188:217–221Crossref, Medline, Google Scholar
7 : Effective Treatments for PTSD: Practice Guidelines From the International Society for Traumatic Stress Studies. New York, Guilford Press, 2008Google Scholar
8 : Fluoxetine in posttraumatic stress disorder. J Clin Psychiatry 1994; 55:517–522Medline, Google Scholar
9 : Lack of efficacy for fluoxetine in PTSD: a placebo controlled trial in combat veterans. Ann Clin Psychiatry 2000; 12:101–105Crossref, Medline, Google Scholar
10 : Double-blind placebo-controlled pilot study of sertraline in military veterans with posttraumatic stress disorder. J Clin Psychopharmacol 2002; 22:190–195Crossref, Medline, Google Scholar
11 : Bupropion treatment in veterans with posttraumatic stress disorder: an open study. J Clin Psychopharmacol 1998; 18:379–383Crossref, Medline, Google Scholar
12 : Adjunctive risperidone treatment in post-traumatic stress disorder: a preliminary controlled trial of effects on comorbid psychotic symptoms. Int Clin Psychopharmacol 2003; 18:1–8Crossref, Medline, Google Scholar
13 : Treatment-resistant posttraumatic stress disorder: strategies for intervention. CNS Spectr 2004; 9:740–752Crossref, Medline, Google Scholar
14 : Prospective study to evaluate the efficacy of aripiprazole as a monotherapy in patients with severe chronic posttraumatic stress disorder: an open trial. Psychopharmacol Bull 2007; 40:6–18Medline, Google Scholar
15 : Adjunctive olanzapine for SSRI-resistant combat-related PTSD: a double-blind, placebo-controlled study. Am J Psychiatry 2002; 159:1777–1779Link, Google Scholar
16 : Olanzapine monotherapy in posttraumatic stress disorder: efficacy in a randomized, double-blind, placebo-controlled study. Hum Psychopharmacol 2012; 27:386–391Crossref, Medline, Google Scholar
17 : A review of atypical antipsychotic medications for posttraumatic stress disorder. Int Clin Psychopharmacol 2011; 26:193–200Medline, Google Scholar
18 : Adjunctive risperidone treatment for antidepressant-resistant symptoms of chronic military service-related PTSD: a randomized trial. JAMA 2011; 306:493–502Crossref, Medline, Google Scholar
19 : Binding of antipsychotic drugs to human brain receptors focus on newer generation compounds. Life Sci 2000; 68:29–39Crossref, Medline, Google Scholar
20 : Quetiapine affects neuropeptide Y and corticotropin-releasing hormone in cerebrospinal fluid from schizophrenia patients: relationship to depression and anxiety symptoms and to treatment response. Int J Neuropsychopharmacol 2012; 15:1051–1061Crossref, Medline, Google Scholar
21 : Active metabolites as antidepressant drugs: the role of norquetiapine in the mechanism of action of quetiapine in the treatment of mood disorders. Front Psychiatry 2013; 4:102Crossref, Medline, Google Scholar
22 : Quetiapine treatment in patients with posttraumatic stress disorder: an open trial of adjunctive therapy. J Clin Psychopharmacol 2003; 23:15–20Crossref, Medline, Google Scholar
23 : Quetiapine improves sleep disturbances in combat veterans with PTSD: sleep data from a prospective, open-label study. J Clin Psychopharmacol 2005; 25:387–388Crossref, Medline, Google Scholar
24 : Prazosin versus quetiapine for nighttime posttraumatic stress disorder symptoms in veterans: an assessment of long-term comparative effectiveness and safety. J Clin Psychopharmacol 2010; 30:225–229Crossref, Medline, Google Scholar
25 : VA/DoD Clinical Practice Guideline for the Management of Post-Traumatic Stress, 2010 (http://www.healthquality.va.gov/guidelines/MH/ptsd/cpgPTSDFULL201011612c.pdf)Google Scholar
26 : The development of a Clinician-Administered PTSD Scale. J Trauma Stress 1995; 8:75–90Crossref, Medline, Google Scholar
27 : Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition. (SCID-I/P). New York, New York State Psychiatric Institute, Biometrics Research, 2002Google Scholar
28 : Off-label use of second generation antipsychotics for post-traumatic stress disorder in the Department of Veterans Affairs: time trends and sociodemographic, comorbidity, and regional correlates. Pharmacoepidemiol Drug Saf 2014; 23:77–86Crossref, Medline, Google Scholar
29 : The use of second generation antipsychotics for post-traumatic stress disorder in a US Veterans Health Administration Medical Center. Epidemiol Psychiatr Sci 2014; 23:281–288Crossref, Medline, Google Scholar
30 : Quetiapine monotherapy for bipolar depression. Neuropsychiatr Dis Treat 2008; 4:11–21Medline, Google Scholar
31 : Pharmacotherapy of PTSD in the U.S. Department of Veterans Affairs: diagnostic- and symptom-guided drug selection. J Clin Psychiatry 2008; 69:959–965Crossref, Medline, Google Scholar
32 : A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry 2013; 170:1003–1010Link, Google Scholar
33 : Plasma prolactin in schizophrenia subjects treated with Seroquel (ICI 204,636). Psychopharmacol Bull 1996; 32:107–110Medline, Google Scholar
34 Benedek DM, Friedman MJ, Zatzick D, Ursano RJ: Guideline Watch (March 2009): Practice Guideline for the Treatment of Patients With Acute Stress Disorder and Posttraumatic Stress Disorder. (http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/acutestressdisorderptsd-watch.pdf)Google Scholar
35 : Antipsychotic medications: metabolic and cardiovascular risk. J Clin Psychiatry 2007; 68(suppl 4):8–13Crossref, Medline, Google Scholar



